Advanced Search
Muscle Fiber Types in Human Vastus Lateralis muscle by Oxidative Stains
Last updated date: Oct 8, 2022 Views: 351 Forks: 0
Abstract
Enzymes are proteins which are highly sensitive to change in pH, temperature and other physiochemical factors.Most of the enzyme activity is lost when a tissue is subjected to standard histological processing and thus it is impossible to correlate accurately morphology with enzyme localisation and activity in the same biopsy specimen. The most useful oxidative enzymes studied in muscle are reduced nicotinamide adenine dinucleotide-tetrazolium reductase (NADH-TR),succinate dehydrogenase (SDH) and cytochrome c oxidase (COX).The principle of the histochemical technique for NADH-TR and SDH is to employ a colourless, soluble tetrazolium salt which is reduced to a dark insoluble formazan product by acting as an electron acceptor.Cytocyhrome oxidase (COX) is an oxidative enzyme in the election transport chain,which uses diaminobenzidine to produce a brown end product.The combination of SDH and COX highlights the COX deficient fibers as blue suggesting defect in respiratory chain complex.
Keywords
Oxidative Stains, NADH-TR, COX, SDH, muscle
Background
Enzyme histochemistry is an important diagnostic technique which is based on metabolism of a substrate provided to tissue enzyme in its orthotopic site. 1It complements standard morphological studies in histopathology2 and is indispensible in assessing the muscle biopsies of patients suffering from neuromuscular disorders .All steps in standard processing procedures, from formalin fixation to wax embedding, contribute to the loss of enzyme activity. In order to maintain the activity of enzymes , fixation and wax embedding should be avoided completely .Freeze drying maintains the tissue components , particularly the enzymes in their native state and are necessary for enzyme histochemistry .
Oxidative stains show the different biochemical properties of different fibre types and their selective involvement in disease processes.3They may show structural changes in the muscle which cannot be appreciated with routine histological stains, such as cores in core myopathies, ‘lobulated’ fibres, and abnormalities in the distribution of mitochondria(ragged blue fibres).
Tetrazolium compounds function as the hydrogen acceptor when diaphorases are being demonstrated histochemically, and the product of the reduction is the water-insoluble formazan pigment .4 Enzymatic activity releases hydrogen from the substrate, and the released hydrogen is transferred to the tetrazolium which is converted to purple-blue formazan pigment, marks the site of enzyme activity.
Succinic dehydrogenase (SDH) is a soluble iron flavoprotein .The enzyme catalyzes the oxidation of succinic acid to fumaric acid.
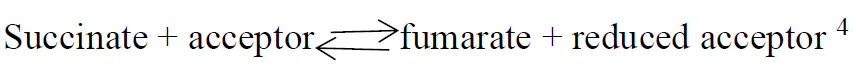
The histochemical demonstration of the enzyme is demonstrated by incubating fresh frozen sections with a succinate substrate in the presence of a tetrazolium compound.
Cytochromes are hemoproteins which transfer electrons in respiratory chain. 4 Cytochrome oxidase is considered to be a characteristic enzyme of the mitochondrial membrane.4 Diaminobenzidine in the presence of COX acts as an electron donor, Oxidative polymerization and cyclization of diaminobenzidine occurs and produces a brown end product that can be enhanced by osmium.3 It is also used to demonstrate fibres devoid of activity due to mutations in mitochondrial DNA.3The combination of SDH and COX highlights the COX deficient fibers but retain SDH activity,and these fibres appear blue .
Materials and Reagents
Gelatin – HIMEDIA GRM 019 – 500gm, Hygroscopic, store at room temperature
Glycerol – LOBA CHEMIE:00155 – 500ml, store at room temperature
Nitro blue tetrazolium – HIMEDIA MB107 – 100mg, store desiccated at 0-5°C
Phenol – RANKEM P0135 – 500gm, Store at room temperature
Succinic acid, disodium salt – Sigma Aldrich 14170 – 100gm, store at room temperature
NADH - Sigma N 8129, store at 0-5°C, Toxic by inhalation
TRIS – Finar 21890SG – 500gm store at room temperature
Catalase (C 9322)- Avoid contact and inhalation
Cytochrome C (SIGMA C 2506 )
3’diaminoberzidine tetrahydrochloride (HIMEDIA RM1440 – 05gm), store at -20°C
Sodium dibasic phosphate (Na2HPO4) dihydrate, Finar 21611SG – 500gm; Store at room temperature
Sucrose – RANKEM S2610 – 500gm, store at room temperature.
Tris EDTA buffer (50X Concentrated PathnSitu Biotechnologies, Cat no-PS009, 500ml)
Low Profile microtome blades 819 (Leica Biosystems, Germany, catalog number: 14035838925)
Microscope glass slides (Rohem , India, Frosted F.2 ISO8037/IS3099)
Camel hair brushes
Elite Pap pen (Diagnostics BioSystems, USA catalog number: CA94588)
Freshly dissected human vastus lateralis muscle
Isopentane (CDH010361, Batch number:040813)
Liquid Nitrogen
Stop watch
15 ml Falcon tubes (Tarsons graduated centrifuge tube, sterile, model-546021)
Leica tissue freezing medium (125ml, REF 14020108926)
Moist chamber
Equipment
Rotary Cryostat Microtome (Thermoscientific MICROM HM 525)
Vortex (Hitech Instrument,Delhi,India, Model No HI-223)
Olympus CX 21i microscope
Cryostat (Thermoscientific 3000265086 HM 525)
Incubator (Sanjeev Scientific Udyog,Haryana,India CE/06343/0418)
Ph meter (LABTRONICS Model-LT-11)
Procedure of tissue processing(Snap freezing)

Staining Procedure (COX):
Dissolve the COX incubating solution in 4.5ml of 50 mM phosphate buffer at pH 7.4
Incubate cryostat sections with incubating solution for 1-2 hours at 37°C incubator in moist chamber .
Wash briefly in distilled water, 3 times
Air dry and mount in gelatin.
Staining Procedure(NADH-TR):
1. NADH Incubating solution
NADH Stock incubating solution - 10.0 ml
NADH - 2.0 mg or pinch
2. Incubate cryostat sections in the NADH incubating solution for 1hour in the incubator at 37°C in moist chamber .
3. Rinse in distilled water, 3 times
4. Air dry and mount in gelatin
Procedure(SDH):
1. SDH Incubating solution
Stock incubating solution - 10.0ml
Disodium succinate - 25 mg
2. Incubate cryostat sections in the SDH incubating solution in the moist chamber for 2hour in a moist at 37°C in the incubator
3. Rinse in distilled water, 3 times.
4. Air dry and mount in gelatin
Procedure(COX-SDH):
Incubate cryostat sections in cytochrome oxidase incubating solution in the moist chamber for 2hour at 37°C in the incubator for 2 hour.
Incubate the same section with SDH incubating solution at 37°C in an incubator for 2 hours in a moistchamber.
Wash well in distilled water
Air dry and mount in glycerin jelly.
Recipes
1. COX Incubating Solution:
3'-3' diaminobenzidineterahydrochloride (DAB) - 4.5mg
Catalase C - 20 mg
Cytochrome C - 75 mg
Sucrose - 400 mg
Dissolve the COX incubating solution in 4.5ml of 50 mM phosphate buffer pH 7.4
2. 50 mM phosphatase buffer pH 7.4
Solution A: Sodium dihydrogen orthophosphate- 180 mg in 25ml of distilled water
Solution B: Disodium hydrogen orthophosphate- 190mg in 25ml of distilled water
Adjust pH to 7.4 with either solution A or solution B
3. 5 mM magnesium chloride (MgCl2)
50mg of magnesium chloride in 50ml of distilled water.
4. NADH /SDH Stock Incubating solution (store in aliquots at -20°C)
Nitro – BT (4 mg/ml) – 2.5 ml
Tris buffer pH 7.4 – 2.5 ml
MgCl2 (5mM) – 1.0 ml
Distilled water – 4.0ml
5. Tris buffer (Working solution)
50x Tris EDTA Buffer - 5 ml
Distilled water - 245ml
6. Gelatin mountant (for mounting)
10gms gelatin dissolved in 60ml of warm distilled water. Add a pinch of thymol as preservative.
Data analysis
NADH-TR/SDH-Myofiber – blue mosaic pattern in a normal muscle
Type 1 fibers – dark;
Type 2A fibers – Intermediate;
Type 2B fibers – Weakest
Fibre type distribution is similar to that seen with both the techniques.
Type 1 fibres have high oxidative and low glycolytic activity, so they are of dark blue intensity with these two oxidative stains.
Type 2A fibres have has a moderate oxidative capacity,hence intermediate colour
Type 2B fibres low oxidative and high glycolytic activity,leading to pale blue colour.
Abnormalities-
Cores- Central area devoid of enzyme activity
Targets-Dark rim around the central pale area
Mitochondrial aggregation(Ragged blue fibres) – Intense blue reaction peripherally
Tubular aggregates – Intense blue reaction in the form of rods
Lobulated fibres-Peripheral aggregate in the form of triangular deposits
COX-Myofibers – Brown mosaic pattern
Type 1 fibers – dark (high oxidative and low glycolytic activityfibre)
Type 2A fibers – intermediate
Type 2B fibers – weakest
Cores- Central area devoid of enzyme activity5
COX Negative fibres-Myofibers devoid of COX( white -no stain)seen in mitochondrial disorders
COX-SDH-Myofibers – Brown mosaic pattern
Type 1 fibers – dark
Type 2A fibers – intermediate
Type 2B fibers – weakest
COX Negative,SDH positive (blue stain)-Myofibers devoid of COX,but retain SDH activity , seen in mitochondrial disorders.6
Table1: Systematic interpretation of changes seen in the muscle fibres and their implications
Features | Implication |
Cores(central/peripheral) | When numerous, suggests central core disease |
Target fibres | Reinnervation followed by denervation |
Large ‘wiped out’ areas devoid of stain | Myofibrillar myopathies |
Ragged blue fibres (Uneven distribution of stain) | Uneven distribution of mitochondria (mitochondrial myopathies) and/or myofibrillar disruption |
Lobulated fibres | More common in limb-girdle 2A(calpainopathy) |
Presence of tubular aggregates | Nemaline rod disease |
Moth-eaten and whorled fibres | Myopathy, Chronic dystrophy |
Targetoid fibres | Myopathies and denervation |
Notes
Always wear gloves and mask as reagents like NADH is toxic by inhalation, Catalase - Irritant(Avoid contact and inhalation) and 3’diaminoberzidine tetrahydrochloride is carcinogenic
Acknowledgments
We would like to thank
Competing interests
The authors declare no financial and non-financial competing interests.
Ethics
Not required
References
- Meier-Ruge WA, Bruder E. Current concepts of enzyme histochemistry in modern pathology. Pathobiology. 2008;75(4):233-43.
- Murray G I, Ewen S W B.A new approach to enzyme histochemical analysis of biopsy specimens.J Clin Pathol 1989;42:767-771
- Dubowitz V, Sewry Caroline A,Oldfros Anders, Lane R.Muscle Biopsy: A Practical Approach . 4th Edition ; Saunders, Elsevier, London 2013
- Lojda Z ,Gossrau R, Schiebler TH. Enzyme Histochemistry: A Laboratory Manual.1st Edition; Springer, Berlin, Heidelberg 1979
- Kaur A, Mathur K, Harsh A. Core Disease in an Adult-Letter to the Editor. AIAN 2022;25(4):729-31
- Kaur A, Mathur K, Harsh A, Thakar K. Progressive external ophthalmoplegia‑ A case report. Indian J Patho Microbiol. DOI:10.4103/ijpm.ijpm_893_21accepted for publication
Legends
Fig 1A)Transverse section stained for SDH showing different fibre types.(80X) 1B)Longitudinal section showing core along the entire length of fibre in SDH stain.(40X) 1C)Transverse section showing central core.(40X) 1D)Transverse section of a target fibre showing dark rim around central pale area.(80X)
Fig 2A)-Transverse section stained for NADH-TR showing different fibre types.(80X)2B) NADHTR stain showing cores devoid of enzyme activity in a core disease patient (40X) 2C)Scattered lobulated fibres stained for NADHTR(80X) 2D)Intensely stained ragged blue fibre in NADHTR showing abnormal mitochondria (80X)
Fig 3A)-Transverse section stained for COX showing different fibre types.(80X)3B) Transverse section showing COX negative fibre.Adjacent type2b fibre shows pale staining. (80X) 3C)Transverse section showing multiple cores.(40X) 3D)Muscle fibre devoid of COX but retained SDH activity (blue)in a progressive external ophthalmoplegia patient.(80X)
Fig 4A)Artifacts caused by section lifting off the slide during staining(40X) 4B) Holes due to ice cystal damage.(40X)
Related files
 1 SDH.tiff
1 SDH.tiff  2 NADH TR.tif
2 NADH TR.tif  artifacts.tif
artifacts.tif  COX FINAL.tif
COX FINAL.tif  MANUSCRIPT FOR BIOPROTOCOL.docx
MANUSCRIPT FOR BIOPROTOCOL.docx - Kaur, D, Harsh, D, Mathur, D, Vijay, D and Bakolia, P K(2022). Muscle Fiber Types in Human Vastus Lateralis muscle by Oxidative Stains. Bio-protocol Preprint. bio-protocol.org/prep1982.
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link
