- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
ROS Detection in Botryococcus braunii Colonies with CellROX Green Reagent
Published: Vol 7, Iss 16, Aug 20, 2017 DOI: 10.21769/BioProtoc.2508 Views: 10210
Reviewed by: Maria SinetovaIgor Cesarino Anonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Visualization of Nitric Oxide, Measurement of Nitrosothiols Content, Activity of NOS and NR in Wheat Seedlings
Sandeep B. Adavi [...] Shailendra K. Jha
Oct 20, 2019 6354 Views
Spatiotemporal Quantification of Cytosolic pH in Arabidopsis Pollen Tubes
Maria Teresa Portes [...] José A. Feijó
Jul 20, 2021 3565 Views
Colocalization Assay with Fluorescent-tagged ATG8 Using a Nicotiana benthamiana-based Transient System
Jinyan Mai [...] Na Luo
Aug 20, 2022 3446 Views
Abstract
We analyzed the reactive oxygen species (ROS) accumulation in the colony-forming green microalga Botryococcus braunii in response to several stress inducers such as NaCl, NaHCO3, salicylic acid (SA), methyl jasmonate, and acetic acid. A staining assay using the fluorescent dye CellROX Green was used. CellROX Green is a fluorogenic probe used for measuring oxidative stress in live cells. The dye is weakly fluorescent inside cells in a reduced state but exhibits bright green photostable fluorescence upon oxidation by ROS and subsequent binding to DNA. The large amount of liquid hydrocarbons produced and excreted by B. braunii, creates a highly hydrophobic extracellular environment that makes difficult to study short times defense responses on this microalga. The procedure developed here allowed us to detect ROS in this microalga even within a short period of time (in minutes) after treatment of cells with different stress inducers.
Keywords: Botryococcus brauniiBackground
Among the first methods developed to detect and quantify hydrogen peroxide and other organic hydroperoxides was the use of titanium (IV) ion (MacNevin and Urone, 1953). The yellow color resulted from the completion of titanium (IV) and peroxide molecules was detected by colorimetry. This method was used to detect endogenous peroxides, and to assay the catalase activity in two varieties of pear fruits for correlation with fruit ripening (Brennan and Frenkel, 1977). Another method to detect lipid hydroperoxides is based on thiobarbituric acid (TBA) and was used to measure the deterioration of foods such as milk (Sidwell et al., 1955). Although this method did not use organic solvents, steam distillation of an acidified slurry was necessary to detect hydroperoxides, and the resulting red color was quantified spectrophotometrically. The procedures described above have several disadvantages such as low sensitivity, interference with other compounds, and use of solvents or substances which may damage the living cells. A more sensitive method was developed in which the blue fluorescence of scopoletin (6-methyl-7-hydroxy-1:2-benzopyrone) disappeared after its oxidation by peroxidase enzyme (Andreae, 1955; Perschke and Broda, 1961). This method was used to detect the H2O2 production by NADPH in the microsomes from rat liver (Thurman et al., 1972). However, scopoletin is expensive, difficult to extract, and is an extremely toxic natural compound (Ojewole and Adesina, 1983a and 1983b). On the other hand, fluorescein is a dye chemically synthesized (Baeyer, 1871) and the chemical structure was elucidated (Markuszewski and Diehl, 1980). The fluorescence of both compounds, scopoletin and fluorescein, was then explained based on their similar chemical structure. So, further development of novel fluorescent dyes more stable and versatile allowed their use in very specific applications (Cathcart et al., 1983). For instance, 2,7-dichlorohydro-fluorescein diacetate (DCFH-DA) was used to study the intracellular production of active oxygen in the brown alga Fucus evanescens (Collén and Davison, 1997). The same compound DCFH-DA was also used to detect oxidative stress tolerance by abscisic acid (ABA) in the green microalga Chlamydomonas reinhardtii (Yoshida et al., 2003). Due to the wide application of these fluorescent dyes, private companies developed other compounds with different properties and each designed for specific applications. CellROX Green Reagent was designed to detect the production of ROS in living cells. So, we chose this dye to detect ROS in early times in B. braunii living cells (Life Technologies Corp., 2012). These reagents are cell-permeable and show no or very weak fluorescence in a reduced state, but their oxidation results in a strong fluorescence. In presence of ROS, the CellROX Green Reagent undergoes oxidation and produce green fluorescence followed by its binding to the DNA in the nucleus. This fact allows us to distinguish between the fluorescence resulting from ROS and the fluorescence from the chlorophyll molecule. Furthermore, this reagent can be fixed with formaldehyde and is compatible with some detergents. These characteristics of CellROX Green Reagent made it suitable to analyze ROS production in stress conditions in cells of the colonial microalga Botryococcus braunii race B (Nonomura, 1988; Banerjee et al., 2002).
Materials and Reagents
- Pipette tips 200 μl (Científica Senna, catalog number: 5-20236 )
- 96-well microplate polypropylene (Thermo Fischer Scientific, Thermo ScientificTM, catalog number: 267245 )
- Glass microscope slide (Corning, catalog number: 2947-75X25 )
- Coverslip (Corning, catalog number: 2890-22 )
Note: This product has been discontinued. - Aluminum foil (Reynolds Wrap 15 m x 30 cm)
- CellROX® Green Reagent (Thermo Fischer Scientific, InvitrogenTM, catalog number: C10444 , Excitation/Emission, 485/520 nm)
- Triton X-100 (Karal, catalog number: 9015 )
- Methyl jasmonate (abbreviated MeJA) (Sigma-Aldrich, catalog number: 392707-5ML )
- Potassium nitrate (KNO3) (Karal, catalog number: 5082 )
- Magnesium sulfate heptahydrate (MgSO4·7H2O) (Karal, catalog number: 6056 )
- Potassium phosphate dibasic (K2HPO4) (Karal, catalog number: 5080 )
- Calcium chloride dihydrate (CaCl2·2H2O) (Karal, catalog number: 2016 )
- Ethylenediaminetetraacetic acid ferric-sodium salt (Fe·Na·EDTA) (Sigma-Aldrich, catalog number: E6760-100G )
- Sulfuric acid (H2SO4) (Karal, catalog number: 1032 )
- Boric acid (H3BO4) (Karal, catalog number: 7021 )
- Manganese sulfate monohydrate (MnSO4·H2O) (Karal, catalog number: 1069 )
- Zinc sulfate monohydrate (ZnSO4·7H2O) (Karal, catalog number: 4089 )
- Cupric sulfate pentahydrate (CuSO4·5H2O) (Karal, catalog number: 8024 )
- Sodium molybdate dihydrate (NaMoO4·2H2O) (Karal, catalog number: 4072 )
- Cobalt(II) sulfate heptahydrate (CoSO4·7H2O) (Sigma-Aldrich, catalog number: 12933 )
Note: This product has been discontinued. - Sodium chloride (NaCl) (Karal, catalog number: 6052 )
- Potassium chloride (KCl) (Karal, catalog number: 5087 )
- Sodium bicarbonate (NaHCO3) (Karal, catalog number: 5010 )
- Sodium phosphate dibasic (Na2HPO4) (Karal, catalog number: 6005 )
- Modified CHU-13 media (see Recipes)
- 1x phosphate-buffered saline (PBS) (see Recipes)
Equipment
- 1.5 L flask (Corning, PYREX®, catalog number: 4980-1XL )
- Micropipettes (Mettler-Toledo International, Rainin®, catalog numbers: 17014392 , 17014382 and 17011790 )
- Incubator shaker (Select BioProducts, model: IncuMixTM Incubator Shaker, catalog number: SBS256 )
- Centrifuge (Labnet International, model: SpectrafugeTM 16M, catalog number: C0160 )
- Optical microscope (ZEISS, model: Axio Lab.A1 ) equipped with 470 nm LED module used for fluorochrome excitation and a set of 38 Endow GFP Filters (free exchange (E) EX BP 470/40, BS FT 495, EM BP 525/50) to detect the emission of the fluorochrome
- Digital camera (ZEISS, model: AxioCam ICc3 Rev.3 )
- pH meter (Cole-Parmer, Jenway, model: 3510 )
- Autoclave Sterilmatic (Market Forge Industries, model: STM-EL )
Software
- ZEN lite 2011 (ZEISS)
- GraphPad Prism version 6.00 for Mac OS X, GraphPad Software, La Jolla California USA (http://www.graphpad.com)
Procedure
- Harvest samples of the algal culture grown in modified Chu-13 media (see Recipes), at different time points after specific stress treatments. There should be sufficient colonies in 100 μl to be clearly observed under a microscope. If your alga can be sedimented after centrifugation, 100 μl should make at least a 20 μl of pellet. If the alga doesn’t sediment as in the case of B. braunii, which floats because of the surrounding hydrocarbons, then the OD at 600 nm should give at least 0.5. If needed, concentrate the cells by centrifugation, filtration, or other methods that are suitable for your sample.
- Transfer 100 μl of each sample in a 96-well microplate and mix with 2 μl of 5 mM CellROX Green, incubate the microplate for 30 min at 36 °C in the dark by shaking at 120 rpm in an incubator shaker.
- Then incline the plates, carefully discard the liquid with a micropipette and wash the cells twice with 100 μl of 1x PBS (see Recipes) containing 0.1% Triton X-100, by shaking at 120 rpm for 5 min at room temperature. Triton X-100 is a commonly used detergent in laboratories widely used to permeabilize the membranes of living cells.
- Transfer an aliquot of 10-20 μl of each sample onto a glass microscope slide, cover with a coverslip, and observe under the microscope.
- Observe samples first under the white light to locate the algal colonies and then switch to fluorescence conditions. Count at least 100 colonies for each sample.
- Colonies with more than 90% of cells having the fluorescent nuclei are considered ROS positive.
- Take pictures with a digital camera and the representative images are shown below (Figure 1).
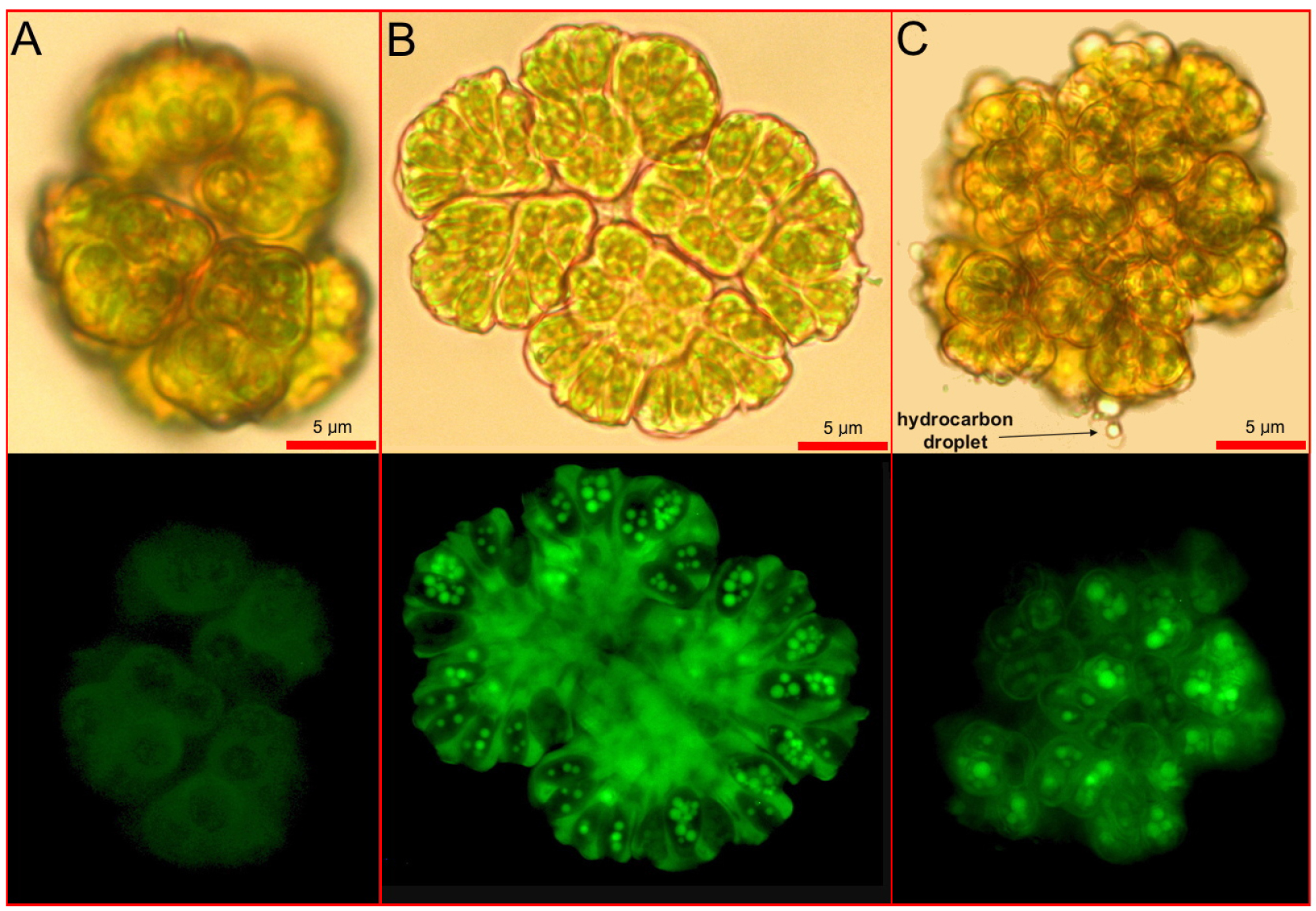
Figure 1. Staining of Botryococcus braunii with CellROX dye. Detect ROS in vivo by analyzing fluorescent nuclei of B. braunii cells in each colony. Upper panels are under white light and lower panels are under fluorescent conditions. A. Control without stress treatment; B. Treatment with strong stress inducer (10 μM MeJA for 60 min); C. Treatment with weak stress inducer (120 mM NaHCO3, for 60 min). - Determine the percentage of ROS positive colonies according to the equation:
(FC/TC) x 100 = %PC
where, FC = number of colonies with fluorescence, TC = total number of observed colonies, and PC = percent of ROS positive colonies.
Data analysis
With this procedure both the concentration of the inducers and the time of induction are optimized. The number of ROS positive colonies after CellROX staining is determined by counting the fluorescent cells induced with the different treatments for 10, 60 and 120 min. Data represent the mean ± SE of at least three replicates. The statistical significance is evaluated with the Tukey’s test (P value = ** 0.0022, *** 0.0003, **** < 0.0001). Statistical tests are performed using GraphPad Prism version 6.00 for Mac OS X, GraphPad Software, La Jolla California USA (http://www.graphpad.com). Data are visualized using Microsoft Excel for Mac 2011 version 14.1.0. The number of positive colonies for ROS are expressed in % positive colonies (Table 1).
Table 1. Percentage of ROS positive B. braunii colonies. Take samples at different times and with different concentrations of the inducer.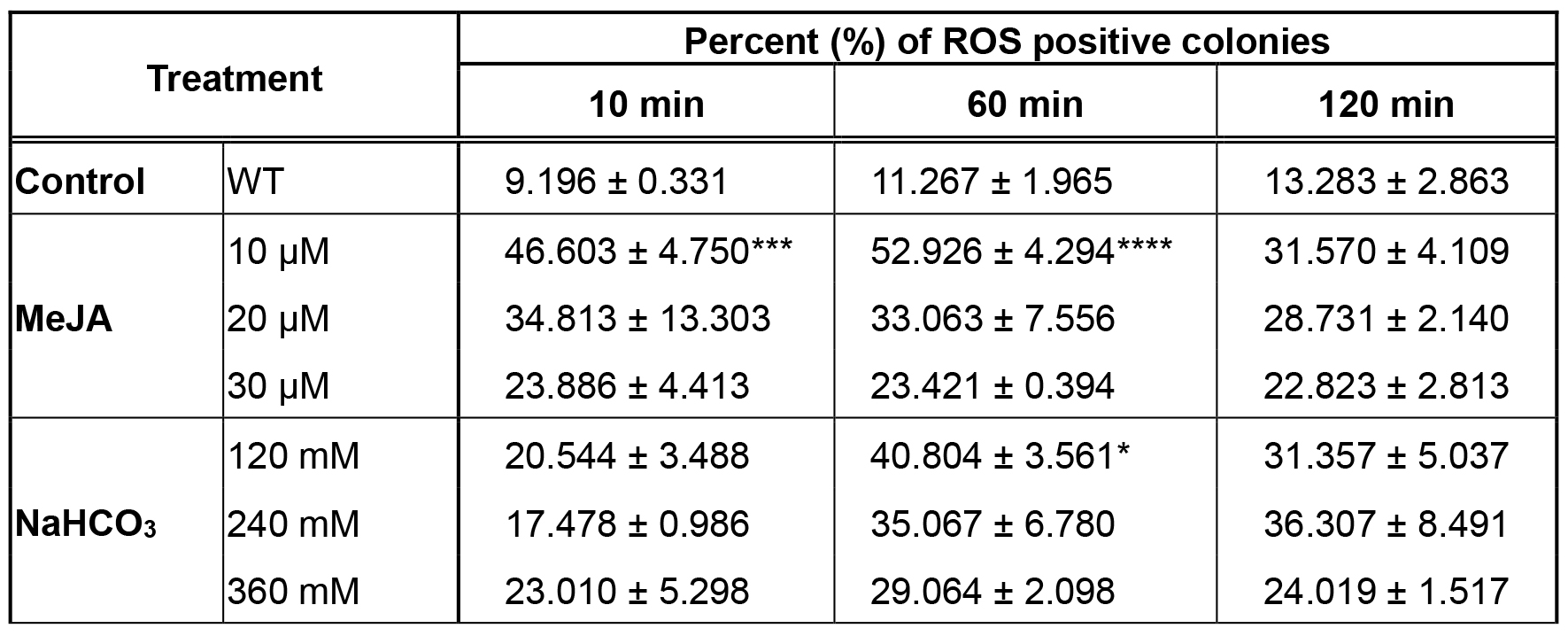
These data show that the best conditions to detect ROS-positive colonies of B. braunii, were 60 min for 10 μM MeJA as well as for 120 mM NaHCO3. However, the best conditions regarding time and concentration of inducers must be determined for other microalgae.
Notes
- A small number of colonies are identified as ROS positive even without treatment of any stress inducer, this may be due to damage during handling of samples. However, they never exceed 13.5% of the total colonies analyzed and are lower than the number of colonies observed in any of the treatments tested.
- Due to the colony organization of B. braunii it was sometimes difficult to count individual cells in a selected sample without moving the field under the microscope. So, once the field was fixed under fluorescent light, colonies were considered ROS positive if more than 90% of the cells in the field had fluorescent nuclei. It is recommended to try different focus on the same colony to distinguish the stained nuclei of the cells at different planes.
- Triton X-100 was added at the washing steps to improve the introduction of the CellROX Green into the B. braunii cells. In step 2 of the Procedure, when the dye was added, it covered and bonded to the surface of the colony but hardly got into it to act in the inner cells. In step 3, the Triton X-100 allowed the introduction of the dye by its detergent property. This was a key step because of this alga secretes large amounts of liquid hydrocarbons which surround the cells, creating a highly hydrophobic extracellular environment. During the washing steps, the dye reached the inner cells to detect ROS and the excess of CellROX was discarded. In our hands, after the second wash, the reaction stopped because all produced ROS were detected with the dye. Perhaps, other microalgae do not require this step although it may help to get better results, mainly if the cultures were forced to produce a high amount of lipids.
Recipes
- Modified CHU-13 media
0.4 g/L KNO3
0.1 g/L MgSO4·7H2O
0.052 g/L K2HPO4
0.054 g/L CaCl2·2H2O
0.01 g/L Fe·Na·EDTA
5 ml/L trace elements
Dissolve each salt in 800 ml deionized water. Set the pH to 7.2-7.5 using diluted H2SO4. The pH of the media should be over 7.0, do not adjust with NaOH. In case the pH is below 7.0, discard the solution and prepare it again
Trace elements:
572 mg/L H3BO4
308 mg/L MnSO4·H2O
44 mg/L ZnSO4·7H2O
16 mg/L CuSO4·5H2O
12 mg/L NaMoO4·2H2O
18 mg/L CoSO4·7H2O
Mix all in 900 ml deionized water in a 1.5 L flask, cover with aluminum foil and sterilize by autoclaving at 121 °C for 20 min - 1x phosphate-buffered saline (PBS)
8 g/L NaCl
0.2 g/L KCl
1.44 g/L Na2HPO4
0.24 g/L KH2PO4
Dissolve the reagents in 800 ml of deionized water. Adjust the pH to 7.4 (or 7.2, if required) with HCl, and then add H2O to 1 L. Sterilize by autoclaving for 20 min at 121 °C or by filter sterilization. Store PBS at room temperature
Acknowledgments
This technique was adapted from the original procedure described in the Manual of CellROX® Oxidative Stress Reagents Man0003555 from Life Technologies Corp. This work was supported by a PhD scholarship to IC-C from Consejo Nacional de Ciencia y Tecnología (CONACYT) Mexico, and grant from the 2012 Texas A&M University CONACYT Collaborative Research Grant Program to EL-G and TPD.
References
- Andreae, W. A. (1955). A sensitive method for the estimation of hydrogen peroxide in biological materials. Nature 175(4463): 859-860.
- Baeyer, A. (1871). Ueber eine neue Klasse von Farbstoffen. Eur J Inorg Chemy 4(2): 555-558.
- Banerjee, A., Sharma, R., Chisti, Y. and Banerjee, U. C. (2002). Botryococcus braunii: a renewable source of hydrocarbons and other chemicals. Crit Rev Biotechnol 22(3): 245–279.
- Brennan, T. and Frenkel, C. (1977). Involvement of hydrogen peroxide in the regulation of senescence in pear. Plant Physiol 59(3): 411-416.
- Cathcart, R., Schwiers, E. and Ames, B. N. (1983). Detection of picomole levels of hydroperoxides using a fluorescent dichlorofluorescein assay. Anal Biochem 134(1): 111-116.
- Collén, J. and Davison, I. R. (1997). In vivo measurement of active oxygen production in the brown alga Fucus evanescens using 2’,7’-dichlorohydrofluorescein diacetate. J Phycol 33(4): 643-648.
- Life Technologies Corp. (2012). Manual of CellROX® Oxidative Stress Reagents. Man0003555: 1-6.
- MacNevin, W. M. and Urone, P. F. (1953). Separation of hydrogen peroxide from organic hydroperoxides. Anal Chem 25(11): 1760-1761.
- Markuszewski, R. and Diehl, H. (1980). The infrared spectra and structures of the three solid forms of fluorescein and related compounds. Talanta 27(11): 937-946.
- Nonomura, A. M. (1988). Botryococcus braunii var. showa (Chlorophyceae) from Berkeley, California, United States of America. Japanese J Phycol 36: 285-291.
- Ojewole, J. A. and Adesina, S. K. (1983a). Cardiovascular and neuromuscular actions of scopoletin from fruit of Tetrapleura tetraptera. Planta Med 49(2): 99-102.
- Ojewole, J. A. O. and Adesina, S. K. (1983b). Mechanism of the hypotensive effect of scopoletin isolated from the fruit of Tetrapleura tetraptera. Planta Med 49(9): 46-50.
- Perschke, H. and Broda, E. (1961). Determination of very small amounts of hydrogen peroxide. Nature 190: 257-258.
- Sidwell, C. G., Salwin, H. and Mitchell, J. H. (1955). Measurement of oxidation in dried milk products with thiobarbituric acid. J Am Oil Chem Soc 32(1): 13-16.
- Thurman, R. G., Ley, H. G. and Scholz, R. (1972). Hepatic microsomal ethanol oxidation. Hydrogen peroxide formation and the role of catalase. Eur J Biochem 25(3): 420-430.
- Yoshida, K., Igarashi, E., Mukai, M., Hirata, K. and Miyamoto, K. (2003). Induction of tolerance to oxidative stress in the green alga, Chlamydomonas reinhardtii, by abscisic acid. Plant Cell Environ 26(3): 451-457.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Lozoya-Gloria, E., Cornejo-Corona, I., Thapa, H. R., Browne, D. R. and Devarenne, T. P. (2017). ROS Detection in Botryococcus braunii Colonies with CellROX Green Reagent. Bio-protocol 7(16): e2508. DOI: 10.21769/BioProtoc.2508.
Category
Plant Science > Phycology > Cell analysis
Microbiology > Microbial physiology > Stress response
Cell Biology > Cell imaging > Fluorescence
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











