- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Extraction and Assays of ADP-glucose Pyrophosphorylase, Soluble Starch Synthase and Granule Bound Starch Synthase from Wheat (Triticum aestivum L.) Grains
Published: Vol 6, Iss 18, Sep 20, 2016 DOI: 10.21769/BioProtoc.1929 Views: 14566
Reviewed by: Samik BhattacharyaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Immunofluorescence for Detection of TOR Kinase Activity In Situ in Photosynthetic Organisms
Ana P. Lando [...] Giselle M. A. Martínez-Noël
Dec 20, 2024 1824 Views

An Activity-Based Proteomics with Two-Dimensional Polyacrylamide Gel Electrophoresis (2D-PAGE) for Identifying Target Proteases in Arabidopsis Apoplastic Fluid
Sayaka Matsui and Yoshikatsu Matsubayashi
Mar 5, 2025 1989 Views
Advancing 2-DE Techniques: High-Efficiency Protein Extraction From Lupine Roots
Sebastian Burchardt [...] Emilia Wilmowicz
Oct 5, 2025 1772 Views
Abstract
Starch biosynthesis in plants involves a network of enzymes of which adenosine-5’-diphosphoglucose (ADP-glucose) pyrophosphorylase (AGPase, E.C. 2.7.7.27), and soluble and granule bound starch synthases (SSS and GBSS, E.C. 2.4.1.21) play central roles. Here, we outline the protocol for extraction and assay of these enzymes in developing grains of wheat (Triticum aestivum L.). The principle of the assays outlined is based on a coupling enzymatic reactions where the product of the initial reaction is used as a substrate for subsequent reactions in order to generate NADPH, which can be measured easily by spectrophotometer. This protocol does not need expensive labelled chemicals and can be carried out using equipment commonly found in a biochemical laboratory. We applied this protocol to study the dynamics of AGPase, SSS and GBSS activity in developing wheat grains at different time points after anthesis.
Keywords: EnzymeBackground
Starch is a carbohydrate polymer made up of amylose, a linear glucan polymer composed of α-1,4-linked glucose molecules, and amylopectin, another glucan polymer composed of α-1,4-linked glucose molecules branched by α-1,6-glycosidic bonds. The enzyme adenosine-5’-diphosphoglucose (ADP-glucose) pyrophosphorylase (AGPase, E.C. 2.7.7.27) catalyzes the first committed step of starch synthesis, converting glucose-1-phosphate and ATP to ADP-glucose and inorganic pyrophosphate (PPi). ADP-glucose is subsequently used by soluble starch synthases (SSS) and granule bound starch synthases (GBSS) (E.C. 2.4.1.21), and starch branching enzymes to elongate and branch the glucan chains of the starch granule.
Initially, AGPase and starch synthase assays were carried out using 14C- and 32P-labelled ADP-glucose (Ghosh and Price, 1966; Vos-Scheperkeuter et al., 1986), which requires the use of expensive chemicals as well as specialized equipment to work with labelled compounds. Here, we outline the method adopted and applied for extracting and assaying AGPase, SSS and GBSS activity in developing wheat (Triticum aestivum L.) grains (Mukherjee et al., 2015). Our protocol is based on the methods reported previously by Nakamura et al. (1989) for measuring AGPase and SSS activities in the endosperm of developing rice grains, and by Schaffer and Petreikov (1997) for measuring the activities of SSS and GBSS in tomato fruits. The method is based on coupling enzymatic reactions where the product of the initial reaction is used as a substrate for subsequent reactions in order to generate NADPH, which can be easily measured by spectrophotometers. The protocol can be carried out using the equipment commonly found in biochemical laboratories and does not require the use of labeled compounds.
Because this protocol is an adoption of the methods developed for different tissues of other plant species (endosperm of rice grain and tomato fruit), the reaction mixture composition for all enzymes was optimized for reaction buffer pH and substrate concentration so that enzyme activity was within the linear phase with respect to incubation time and protein concentration. The amounts of enzyme preparation added (PGM, pyruvate kinase, hexokinase, G6PDH) have been adjusted to achieve completion of coupling reaction in expected time frame. This protocol can be used for studying the activities of AGPase, SSS and GBSS in other tissues of wheat as well as in different tissues of other plant species; however, optimization of reaction buffer pH and substrate concentrations is required.
The principle of AGPase assay is presented in Figure 1. AGPase present in plant tissue extract catalyzes the conversion of ADP-glucose and PPi into glucose-1-phosphate and ATP. After the first reaction is stopped by boiling, phosphoglucomutase (PGM) is added to the reaction mixture; PGM converts quantitatively glucose-1-phosphate into glucose-6-phosphate. Subsequently, glucose-6-phosphate dehydrogenase (G6PDH) is added; G6PDH converts glucose-6-phosphate in the presence of NADP into 6-phosphogluconic acid. At the same time, NADP is converted into NADPH and the quantity of NADPH formed is equivalent to the quantity of glucose-6-phosphate oxidized. By measuring NADPH concentration spectrophotometrically at 340 nm, we can quantify the amount of ADP-glucose degraded by AGPase activity.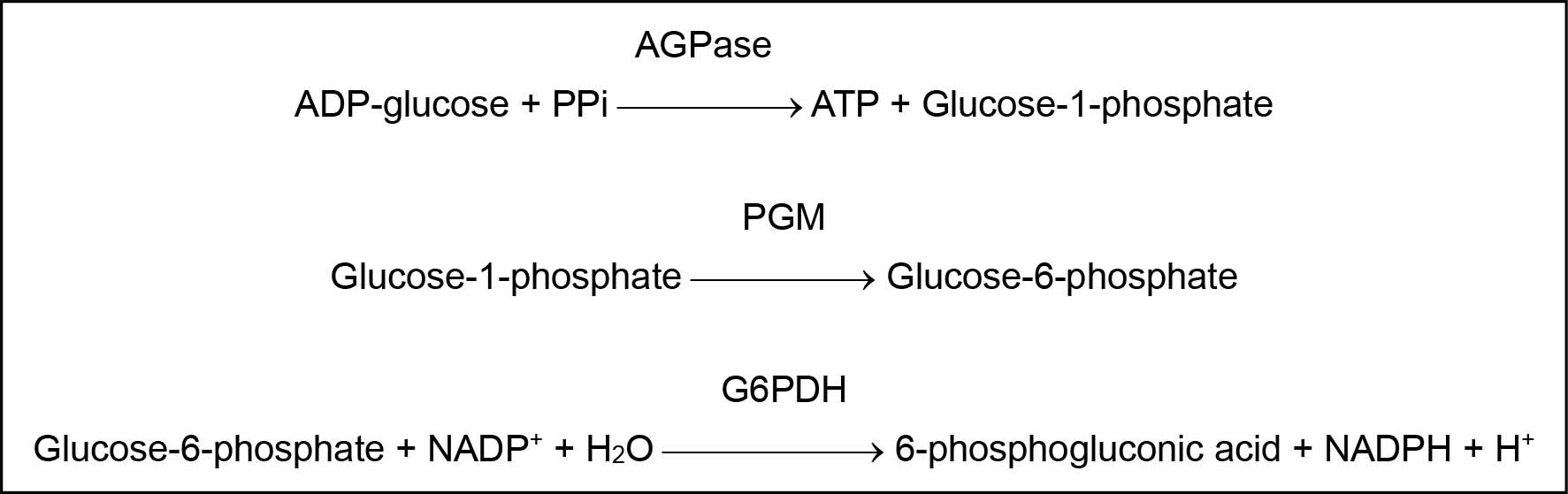
Figure 1. Principle for ADP-glucose pyrophosphorylase (AGPase) assay
The principle of SSS/GBSS assay is presented in Figure 2. Starch synthases present in plant tissue extract (SSS) or in starch granule suspension (GBSS) catalyze the conversion of ADP-glucose into ADP coupled with the elongation of amylopectin primer for one glucose residue. Pyruvate kinase added to the reaction mixture after the first reaction is stopped by boiling converts quantitatively ADP into ATP in the presence of PEP. Subsequently, addition of glucose together with hexokinase leads to the conversion of ATP into glucose-6-phosphate. In the next step of the assay, G6PDH converts glucose-6-phosphate in the presence of NADP into 6-phosphogluconic acid; NADP is converted into NADPH at the same time and the quantity of NADPH formed is equivalent to the quantity of glucose-6-phosphate oxidized. By measuring NADPH concentration spectrophotometrically at 340 nm, we can quantify the amount of ADP-glucose (at the first step of the analysis) degraded by SSS/GBSS activity.
Figure 2. Principle for soluble and granule bound starch synthases (SSS and GBSS) assay
Materials and Reagents
- Chemicals
- Common chemicals, buffers and kits
- Protein assay kit (Bio-Rad Laboratories, catalog number: 5000002 )
- 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) (Thermo Fisher Scientific, Fisher Scientific, catalog number: BP310 )
- Sodium hydroxide (NaOH) (Thermo Fisher Scientific, Fisher Scientific, catalog number: BP359 )
- Magnesium chloride hexahydrate (MgCl2·6H2O) (Sigma-Aldrich, catalog number: M2670 )
- Ethylenediaminetetraacetic acid (EDTA), disodium salt dihydrate (Na2EDTA·2H2O) (Sigma-Aldrich, catalog number: E1644 )
- DL-dithiothreitol (DTT) (Sigma-Aldrich, catalog number: D0632 )
- Polyvinylpyrrolidone (average molecular weight 10,000) (Sigma-Aldrich, catalog number: PVP10 )
- Potassium chloride (KCl) (Thermo Fisher Scientific, Fisher Scientific, catalog number: BP366 )
- Tetrasodium pyrophosphate (PPi) decahydrate (Na4P2O7·10H2O) (Sigma-Aldrich, catalog number: S6422 )
- Protein assay kit (Bio-Rad Laboratories, catalog number: 5000002 )
- Substrates and cofactors
- Adenosine-5’-diphosphoglucose (ADP-glucose), disodium salt (Sigma-Aldrich, catalog number: A0627 )
- Amylopectin from maize (Sigma-Aldrich, catalog number: 10120 )
- β-Nicotinamide adenine dinucleotide phosphate (NADP) sodium salt hydrate (Sigma-Aldrich, catalog number: N0505 )
- Phospho(enol)pyruvic acid (PEP) monosodium salt hydrate (Sigma-Aldrich, catalog number: P0564 )
- Adenosine-5’-diphosphoglucose (ADP-glucose), disodium salt (Sigma-Aldrich, catalog number: A0627 )
- Enzymes
- Hexokinase (E.C. 2.7.1.1) from Saccharomyces cerevisiae (Sigma-Aldrich, catalog number: H5000 )
- Glucose-6-phosphate dehydrogenase (G6PDH, E.C. 1.1.1.49) from Saccharomyces cerevisiae (Sigma-Aldrich, catalog number: G7877 )
- Phosphoglucomutase (PGM, E.C. 5.4.2.2) from rabbit muscle (Sigma-Aldrich, catalog number: P3397 )
- Pyruvate kinase (E. C. 2.7.1.40) from rabbit muscle (Sigma-Aldrich, catalog number: P1506 )
- Hexokinase (E.C. 2.7.1.1) from Saccharomyces cerevisiae (Sigma-Aldrich, catalog number: H5000 )
- Stock solutions (see Recipes)
- 20 mM ADP-glucose
- 10 mM NADP
- 20 mM PEP
- 20 mM ADP-glucose
- Common chemicals, buffers and kits
- Enzymatic activities reaction mixtures
- Extraction buffer (see Recipes, section 1)
- AGPase activity reaction mixture (see Recipes, section 3a)
- Starch synthase activity reaction mixture (see Recipes, section 3b)
- Solution 1 for starch synthase activity assay (Pyruvate kinase reaction mixture) (see Recipes, section 3c)
- Solution 2 for starch synthase activity assay (Glucose-6-phosphate dehydrogenase reaction mixture) (see Recipes, section 3d)
- Extraction buffer (see Recipes, section 1)
- Plant material
- Grains of common wheat (Triticum aestivum L.)
- Grains of common wheat (Triticum aestivum L.)
- Other materials
- Eppendorf tubes (1.5 and 2.0 ml)
- Falcon tubes (15 ml)
- Liquid nitrogen
- Microcentrifuge tube locker (for example, Sigma-Aldrich, catalog number: Z708372 , or Sorenson Bioscience MCT LidLockTM, catalog number: 11870 )
Note: Product Z708372 has been discontinued. - Miracloth (Merck Millipore, catalog number: 475855 )
- Eppendorf tubes (1.5 and 2.0 ml)
Equipment
- Porcelain mortar and pestle
- Analytical balance (capacity - 100 g or higher, resolution - 0.001 g or better)
- Digital heat block or water bath suitable for 30 °C and digital heat block or water bath suitable for 100 °C
Note: For some steps of the analysis, immediate transfer from 30 °C to 100 °C is necessary. This is why two heating devices are necessary simultaneously for the protocol. - Refrigerating ultracentrifuge with appropriate rotor suitable for spinning of 15 ml Falcon tube and 1.5-2 ml Eppendorf tubes at up to 12,000 x g
- Ultraviolet (UV)/Visible spectrophotometer equipped with heated cell holder suitable for maintaining cuvette temperature at 30 °C. We used GE Healthcare/Amersham Bioscience Ultrospec 3100 Pro UV/Visible spectrophotometer (Biochrom Ltd, Cambridge, UK).
Procedure
Summary of the enzyme extraction procedure is shown in a flowchart (Figure 3).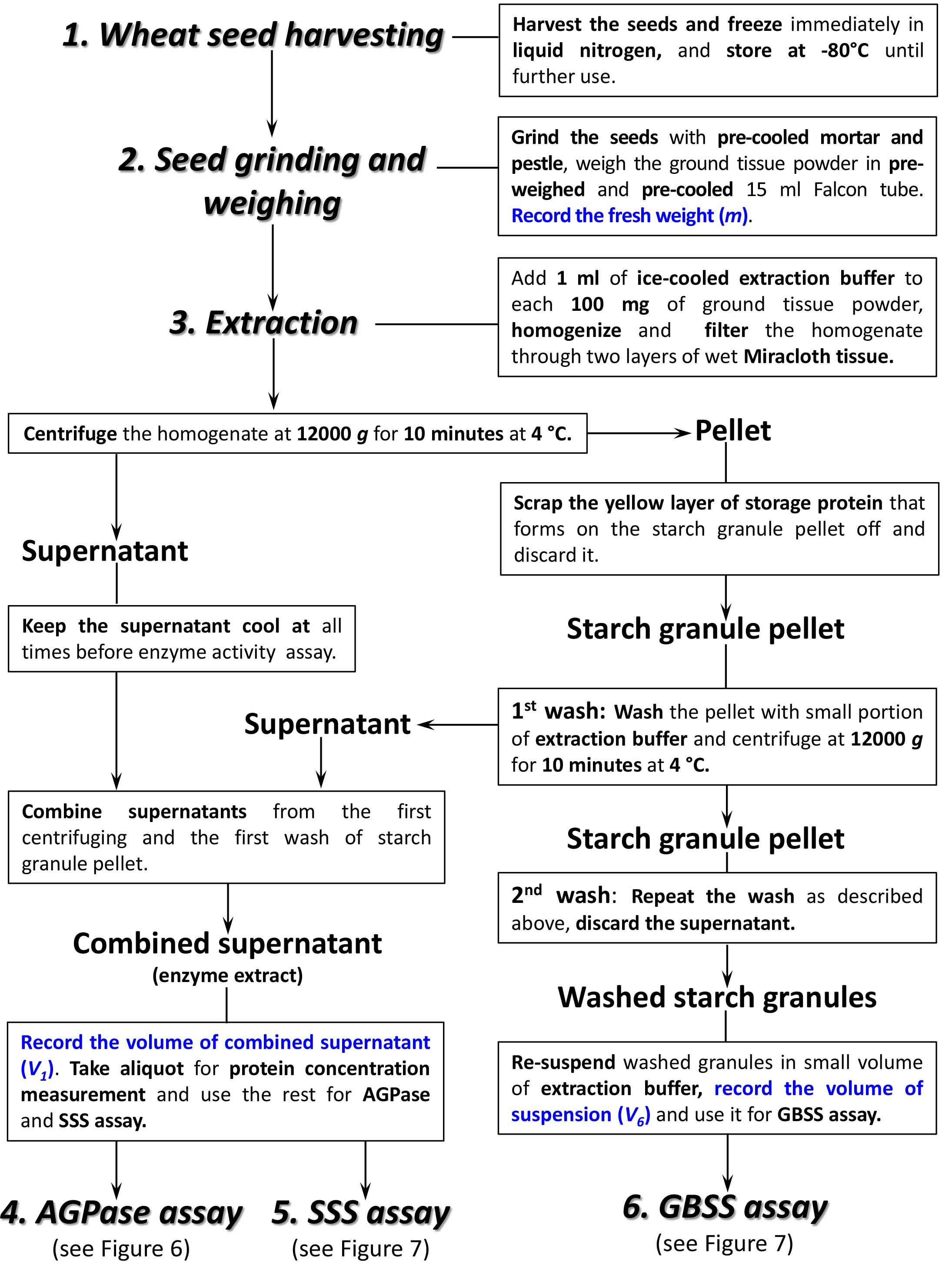
Figure 3. Flowchart for extraction of AGPase, SSS and GBSS from developing wheat seeds. Please, refer to the main text of the protocol for specific conditions and buffers/media composition required for each step.
- Wheat grain harvesting and storage before analysis
Developing grains of wheat were harvested from primary and secondary tillers at different time points after anthesis (Figure 4), in liquid nitrogen and stored at -80 °C until further use. Care has to be taken not to thaw the frozen grains till enzyme assay as thawing the grains would seriously affect the result of the analysis.
Figure 4. Developing seeds of wheat cv. Yecora Rojo at 4, 8, 16 and 25 days after anthesis - Extraction
- Wheat grains were ground to a fine powder using liquid nitrogen in a pre-cooled mortar and pestle and ground tissue was transferred into empty pre-weighed Falcon tube pre-cooled with liquid nitrogen. The weight of the ground tissue was determined. This was performed by first weighing an empty Falcon tube, and then weighing it again filled with the ground tissue. The difference of the two readings equals to the net weight of the ground tissue. This value was recorded and used for further calculations (m, see Calculations, Equations 1, 2, 4, 5, 7).
- The extraction buffer was prepared and kept on ice (see Recipes, section 1).
- 1 ml of ice-cold extraction buffer was added to each 100 mg of the ground tissue and then homogenized.
- The homogenate was filtered through two layers of Miracloth moistened with extraction buffer and centrifuged at 12,000 x g for 10 min at 4 °C, and the supernatant containing the crude enzyme was transferred to a fresh tube and stored on ice.
- The yellow layer of storage protein that forms over the pellet of starch granules was scraped off and discarded (Figure 5).
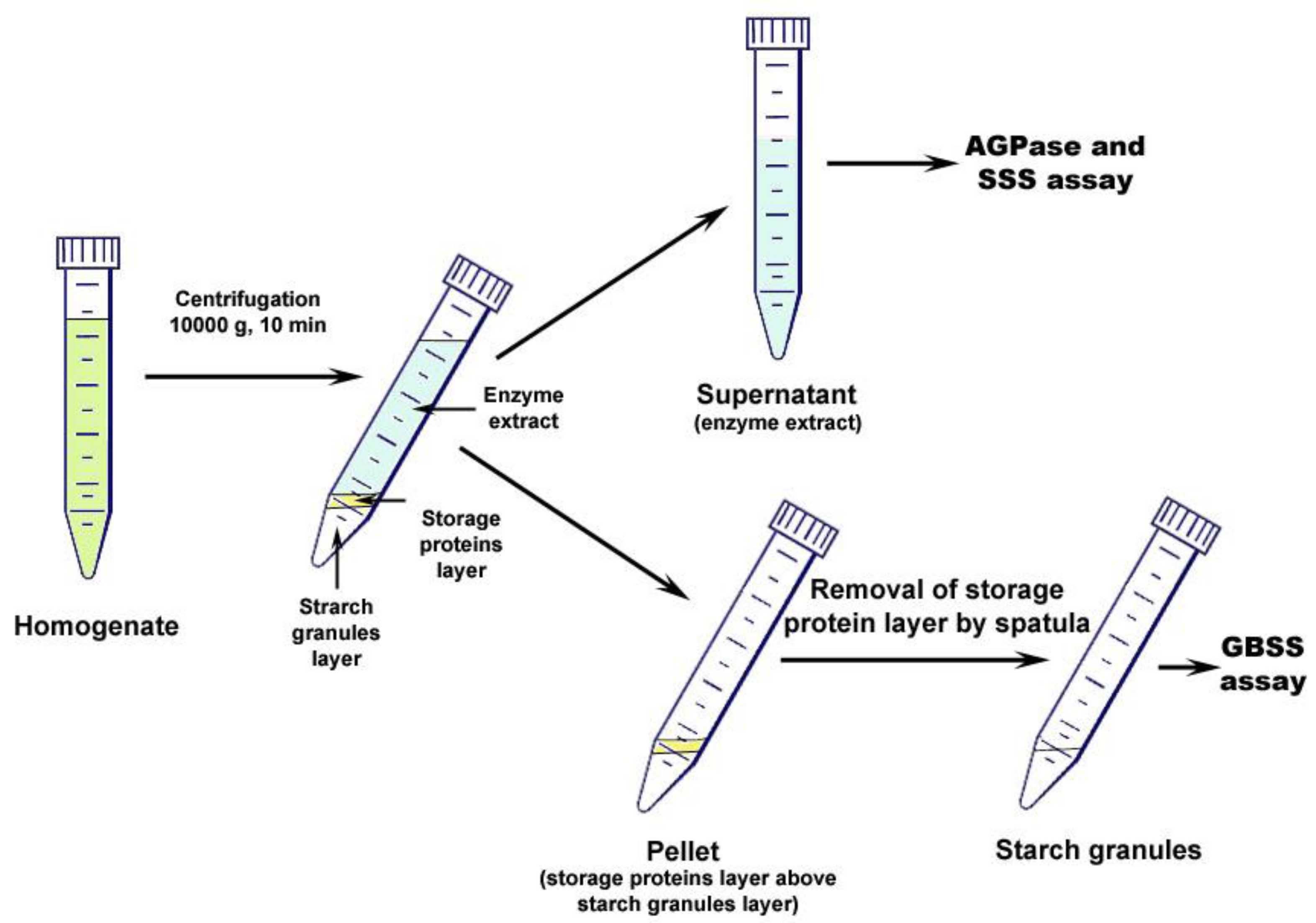
Figure 5. Fractioning of plant tissue homogenate. After centrifugation, the pellet consists of two layers; the upper layer formed by storage proteins and the lower layer by starch granules. The supernatant is collected into a separate tube and is used for AGPase and SSS assay. The upper layer of the pellet (storage proteins) is removed by spatula and discarded. The lower layer (starch granules) is used for GBSS assay after washing. - The pellet of starch granules was washed with 2-4 ml* of extraction buffer twice by centrifugation to remove all traces of SSS. Supernatant obtained after first washing was combined with initial one (step B4) and the pooled supernatant was used for the AGPase and SSS activity assays. The volume of the pooled supernatant was recorded for further calculations (V1, see Calculations, Equations 1, 2, 4, 5). Supernatant after second washing was discarded**.
*Note: The volume of extraction buffer used for washing should be sufficient to wash the starch granule pellet, but not in excess to avoid dilution of the extract. Use of extraction buffer volume that is equal to 20-25% of the volume used in step B3 is a good practice.
**Note: The aim of starch granule pellet washing is to eliminate SSS contamination of the GBSS fraction. The amount of SSS in the first wash may significantly affect the final result of the SSS activity assay. This is why the supernatant from the first wash is combined with the main extract. The second wash may contain some SSS activity, but this amount does not significantly affect the final result of the assay and it can be discarded to avoid extra dilution of the main extract. - The pellet of starch granules was re-suspended in 0.5-2.0 ml* of extraction buffer and stored on ice until used for the GBSS activity assay. The volume of suspension was recorded and used for further calculations of GBSS activity (V6, see Calculations, Equation 7).
*Note: The volume of buffer used to re-suspend starch granules depends on the amount of granules collected. For larger quantities of granules, use a larger volume of re-suspending buffer, but do not exceed the volume used in step B6.
- Wheat grains were ground to a fine powder using liquid nitrogen in a pre-cooled mortar and pestle and ground tissue was transferred into empty pre-weighed Falcon tube pre-cooled with liquid nitrogen. The weight of the ground tissue was determined. This was performed by first weighing an empty Falcon tube, and then weighing it again filled with the ground tissue. The difference of the two readings equals to the net weight of the ground tissue. This value was recorded and used for further calculations (m, see Calculations, Equations 1, 2, 4, 5, 7).
- Enzyme activity assay
- AGPase activity assay
Summary of the procedures for AGPase activity assay is shown in a flowchart (Figure 6).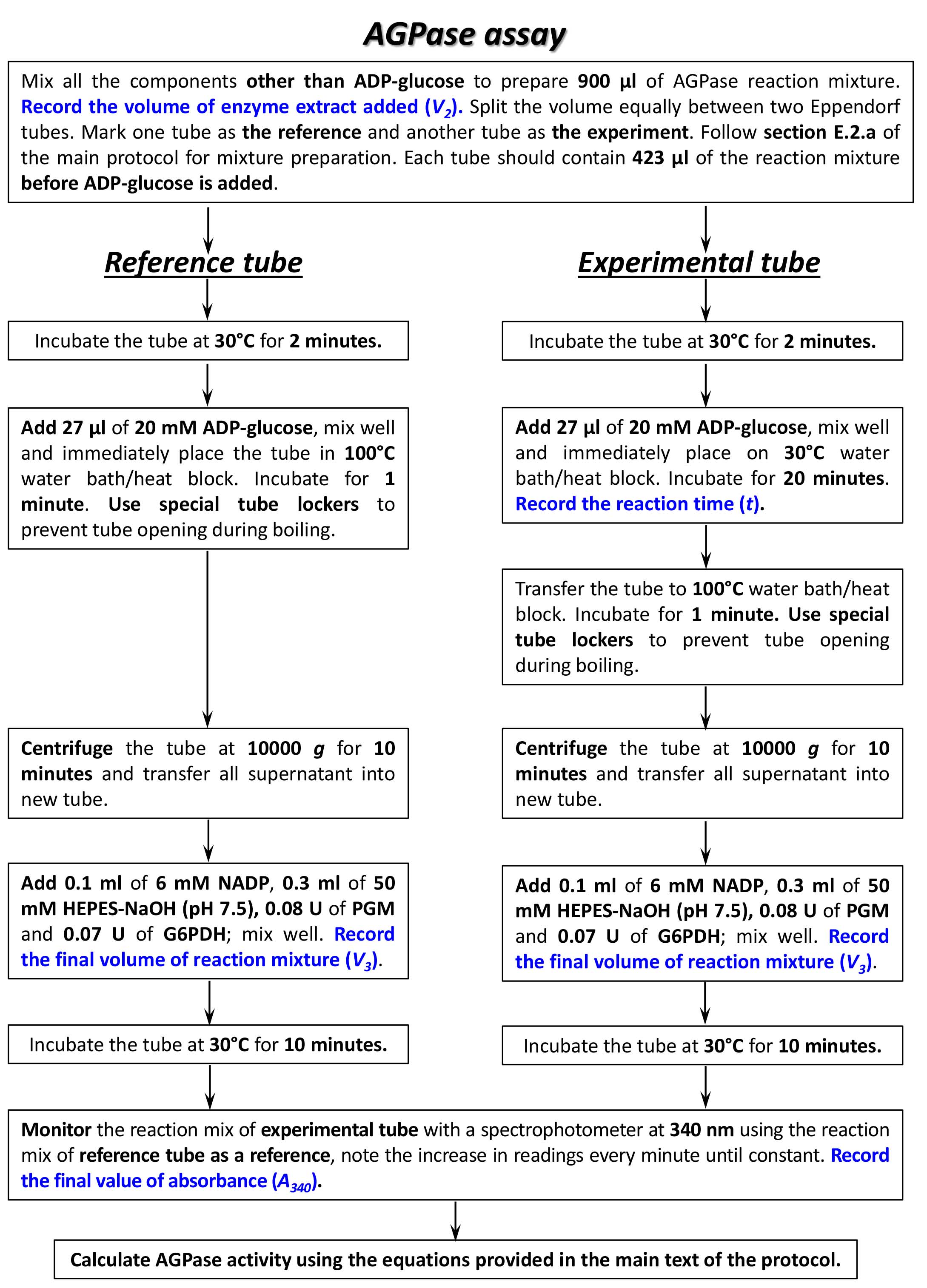
Figure 6. Flowchart of AGPase assay. Please, refer to the main text of the protocol for specific conditions and buffers/media composition needed for each step of the assay.- Two tubes with AGPase reaction mixture, each containing 50 mM HEPES-NaOH (pH 7.5), 1.2 mM ADP-glucose*, 5 mM PPi, 6 mM MgCl2, 3 mM DTT and 50 µl of enzyme extract (V2, see Calculations, Equations 1, 3) in a total volume of 0.45 ml were prepared.
*Note: The reaction mixture is prepared first by mixing all components other than ADP-glucose in a volume of 423 µl as shown in Table 1 (see Recipes, section 3a). - The tubes were pre-incubated at 30 °C for 2 min prior to starting the reaction by adding 27 µl of 20 mM ADP-glucose stock solution.
- The reaction in the first tube (reference tube) was stopped immediately after the addition of ADP-glucose by incubating the tube in water bath at 100 °C for 1 min, whereas the second tube (experimental tube) was incubated at 30 °C for 15 min (t, reaction time, see Calculations, Equations 1, 3) followed by incubation of the tube at 100 °C for 1 min*. Subsequently (steps C1d-f), the reactions in the experimental and reference tubes were processed simultaneously in a similar manner.
*Note: Use special microcentrifuge tube lockers (see Materials and Reagents, section D4) to prevent tube opening during boiling. Tube opening at this stage may lead to partial evaporation of water and uncontrolled decrease of reaction mixture volume that may seriously affect the accuracy of the measurement. - The reaction mixtures were allowed to cool down to room temperature followed by centrifuging at 10,000 x g at room temperature for 10 min.
- The supernatant (0.45 ml) was transferred to a new tube, and 0.1 ml of 6 mM NADP and 0.3 ml of 50 mM HEPES-NaOH (pH 7.5) were added to it. The total volume of the final reaction mix (0.85 ml, V3, see Calculations, Equations 1, 3) was recorded and used for further calculations.
- 0.08 U of PGM and 0.07 U of G6PDH were added in a volume of 1 μl each and the tubes were incubated at 30 °C for 10 min.
- The reaction mix of experimental tube was monitored in a spectrophotometer at 340 nm using the reaction mix of reference tube as a reference and the increase in readings noted every minute until constant. The final value of absorbance (A340, see Calculations, Equations 1, 3) was recorded and used for further calculations.
- Two tubes with AGPase reaction mixture, each containing 50 mM HEPES-NaOH (pH 7.5), 1.2 mM ADP-glucose*, 5 mM PPi, 6 mM MgCl2, 3 mM DTT and 50 µl of enzyme extract (V2, see Calculations, Equations 1, 3) in a total volume of 0.45 ml were prepared.
- Soluble starch synthase (SSS) activity assay
Summary of the procedures for SSS and GBSS activity assay is shown in a flowchart (Figure 7).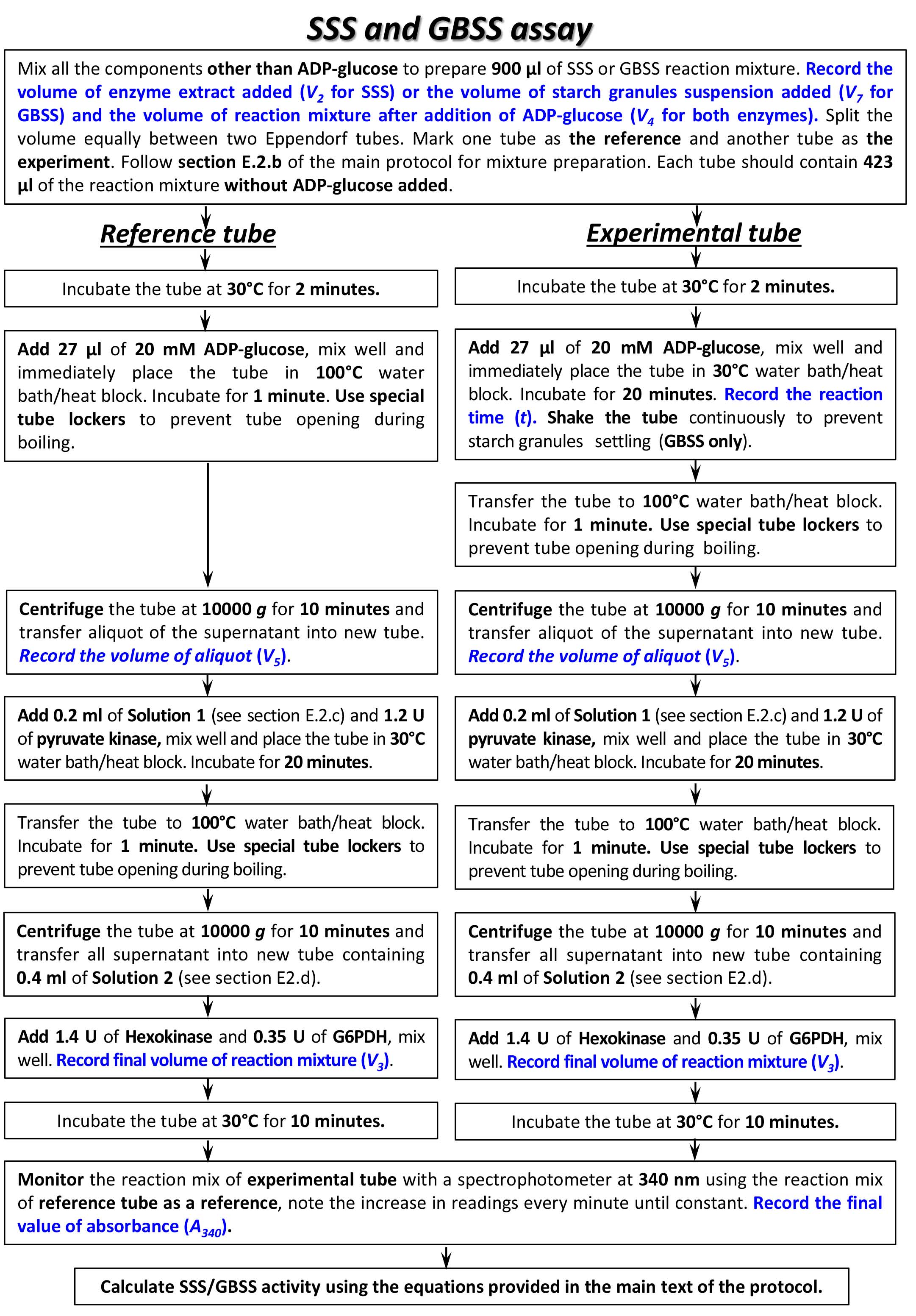
Figure 7. Flowchart of SSS and GBSS assay. Please, refer to the main text of the protocol for specific conditions and buffers/media composition needed for each step of the assay.- For SSS activity assay, two tubes with reaction mixture, each containing 50 mM HEPES-NaOH (pH 7.5), 1.6 mM ADP-glucose*, 1.4 mg amylopectin, 15 mM DTT, and 0.2 ml of enzyme extract in a total volume of 0.45 ml were prepared. The volume of enzyme extract added to starch synthase reaction mixture, as well as total volume of reaction mixture (V2 and V4, correspondingly; see Calculations, Equations 4, 6, 7) were recorded and used for further calculations.
Note*: The reaction mixture is prepared first by mixing all the components other than ADP-glucose in a volume of 423 µl as shown in Table 2 (see Recipes, section 3b). - The tubes were pre-incubated in a water bath at 30 °C for 2 min prior to starting the reaction by adding 27 µl of 20 mM ADP-glucose stock solution.
- The reaction in the first tube (reference tube) was stopped immediately after the addition of ADP-glucose by incubating the tube in a water bath at 100 °C for 1 min*, whereas the second tube (experimental tube) was incubated at 30 °C for 20 min (t, reaction time, see Calculations, Equations 4, 6, 7) followed by incubating the tube at 100 °C for 1 min*. Subsequently (steps C2d-g), the reactions in the experimental and reference tubes were processed simultaneously in a similar manner.
*Note: Use special microcentrifuge tube lockers (see Materials and Reagents, section D4) to prevent tube opening during boiling. Tube opening at this stage leads to partial evaporation of water and uncontrolled decrease of reaction mixture volume that may seriously affect the accuracy of the measurement. - The reaction mixtures were allowed to cool down to room temperature followed by the centrifuging at 10,000 x g at room temperature for 10 min, 0.3-0.4 ml aliquot of supernatant was collected for further analysis. The volume of the aliquot was recorded and used for further calculations (V5, see Calculations, Equations 4, 6, 7).
- 0.2 ml of Solution 1 (see Recipes, section 3c) was added to the aliquot of supernatant from step C2d. along with 1.2 U of pyruvate kinase in a volume of 1-2 μl and incubated at 30 °C for 20 min followed by heating the tube at 100 °C for 1 min.
- The reaction mixture was allowed to cool down to room temperature followed by the centrifuging at 10,000 x g at room temperature for 10 min, supernatant was transferred to a fresh tube containing 0.4 ml of Solution 2 (see Recipes, section 3d). The final volume of reaction mixture was recorded and used for further calculations (V3, see Calculations, Equations 4, 6, 7).
- 1.4 U hexokinase and 0.35 U G6PDH were added to the final mixture in a volume of 1-2 μl each and the tubes were incubated at 30 °C for 10 min.
- The reaction mix of experimental tube was monitored in a spectrophotometer at 340 nm using the reaction mix of reference tube as a reference and the increase in readings noted every minute until constant. The final value of absorbance (A340, see Calculations, Equations 4, 6, 7) was recorded and used for further calculations.
- For SSS activity assay, two tubes with reaction mixture, each containing 50 mM HEPES-NaOH (pH 7.5), 1.6 mM ADP-glucose*, 1.4 mg amylopectin, 15 mM DTT, and 0.2 ml of enzyme extract in a total volume of 0.45 ml were prepared. The volume of enzyme extract added to starch synthase reaction mixture, as well as total volume of reaction mixture (V2 and V4, correspondingly; see Calculations, Equations 4, 6, 7) were recorded and used for further calculations.
- Granule bound starch synthase (GBSS) activity assay
- Insoluble starch synthase was assayed as described above for SSS (step C2), but the reaction mixture described in section C2a was modified (see Recipes, section 3b). Amylopectin and enzyme extract were omitted, while 0.1 ml aliquot of the starch granules pellet suspension was added as a source of both enzyme and reaction substrate. The suspension was vigorously mixed before being added to the reaction mixture. Settling of granules during the assay was prevented by slow and continuous shaking of the reaction tubes.
- The volume of starch granules pellet suspension added was recorded and used for further calculations (V7, see Calculations, Equation 7).
- All further steps of GBSS assay were similar to that described for SSS (see steps C2b-h).
- Insoluble starch synthase was assayed as described above for SSS (step C2), but the reaction mixture described in section C2a was modified (see Recipes, section 3b). Amylopectin and enzyme extract were omitted, while 0.1 ml aliquot of the starch granules pellet suspension was added as a source of both enzyme and reaction substrate. The suspension was vigorously mixed before being added to the reaction mixture. Settling of granules during the assay was prevented by slow and continuous shaking of the reaction tubes.
- Determination of protein content
- Protein content was determined in grain extracts by Bradford assay (Bradford, 1976) using Bio-Rad Protein Assay Kit and BSA as a standard.
- To perform the analysis, follow the instructions in the manual supplied with the kit available in your laboratory.
- Protein content in the enzyme extract (CPR, expressed in mg ml-1) was used for calculation of enzymatic activity on the basis of mg of protein according to Equations 2, 3, 5, 6 (see Calculations, sections D1c and D2c).
- Protein content was determined in grain extracts by Bradford assay (Bradford, 1976) using Bio-Rad Protein Assay Kit and BSA as a standard.
- AGPase activity assay
- Calculations
- AGPase
- Enzyme activity of AGPase was expressed in enzyme units. In terms of this protocol, one enzyme unit (U) of AGPase is determined as the amount of enzymatic activity that converts 1 micromole of ADP-glucose into glucose-1-phosphate per 1 min at pH 7.5 and at 30 °C.
- Enzyme activity of AGPase on g of tissue fresh weight may be calculated according to equation (1).
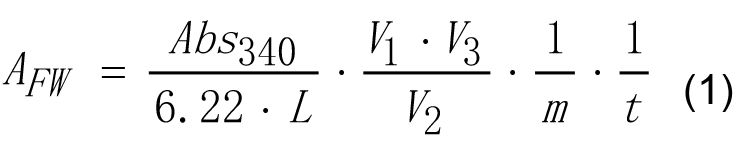
Where,
6.22 - Micromolar extinction coefficient for NAD(P)H at 340 nm, cm2 μmol-1,
Abs340 - Absorbance of reaction mixture of experimental tube measured at 340 nm against the reaction mixture of reference tube,
AFW - Enzyme activity of AGPase calculated on fresh weight basis, U g-1,
L - Length of optical path, cm. This parameter is 1.0 cm in most cases,
m - Fresh weight of the sample, g,
t - Reaction time, min,
V1 - The volume of the extract, ml,
V2 - The volume of the extract added to the reaction mixture, ml,
V3 - The volume of final reaction mixture, ml. - Enzyme activity of AGPase on mg of protein basis may be calculated according to equations (2) and (3).
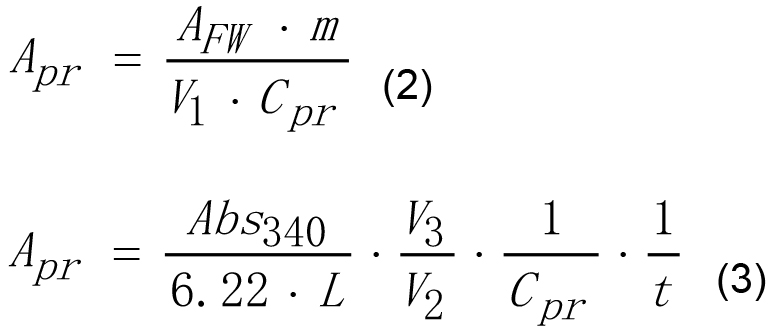
Where,
Apr - Enzyme activity of AGPase calculated on protein basis, U·mg-1,
Cpr - Protein concentration in the extract, mg·ml-1,
and other parameters are the same as for equation (1).
- Enzyme activity of AGPase was expressed in enzyme units. In terms of this protocol, one enzyme unit (U) of AGPase is determined as the amount of enzymatic activity that converts 1 micromole of ADP-glucose into glucose-1-phosphate per 1 min at pH 7.5 and at 30 °C.
- Starch synthase
- Enzyme activity of starch synthase (both SSS and GBSS) was expressed in enzyme units. In terms of this protocol, one enzyme unit (U) of starch synthase (both SSS and GBSS) is determined as the amount of enzymatic activity that converts 1 micromole of ADP-glucose into starch and 1 micromole of ADP per 1 min at pH 7.5 and at 30 °C.
- Enzyme activity of SSS on g of tissue fresh weight may be calculated according to equation (4).
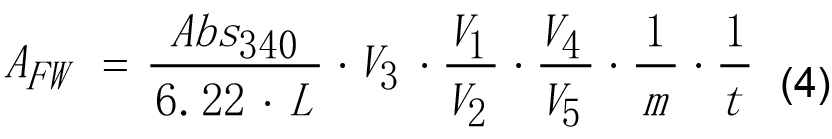
Where,
AFW - Enzyme activity of SSS calculated on fresh weight basis, U g-1,
6.22 - Micromolar extinction coefficient for NAD(P)H at 340 nm, cm2 μmol-1,
Abs340 - Absorbance of reaction mixture of experimental tube measured at 340 nm against the reaction mixture of reference tube,
L - Length of optical path, cm. This parameter is 1.0 cm in most cases,
m - Fresh weight of the sample, g,
t - Reaction time, min,
V1 - The volume of the extract, ml,
V2 - The volume of the extract added to the reaction mixture, ml,
V3 - The volume of final reaction mixture, ml,
V4 - The volume of SSS reaction mixture, ml,
V5 - The volume of an aliquot of SSS reaction mixture taken after stopping the reaction by boiling, ml. - Enzyme activity of SSS on mg of protein basis may be calculated according to equations (5) and (6).
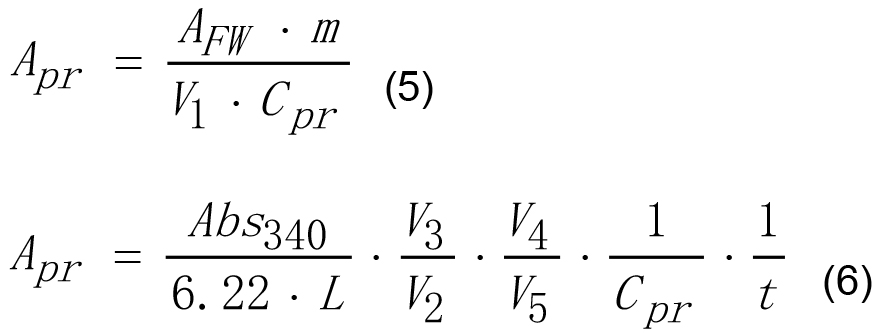
Where,
Apr - Enzyme activity of SSS calculated on protein basis, U mg-1,
Cpr - Protein concentration in the extract, mg ml-1,
and other parameters are the same as for equation (4). - Enzyme activity of GBSS on g of tissue fresh weight may be calculated according to equation (7).
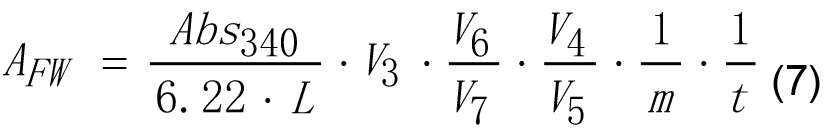
Where,
AFW - Enzyme activity calculated on fresh weight basis,
6.22 - Micromolar extinction coefficient for NAD(P)H at 340 nm, cm2 μmol-1,
Abs340 - Absorbance of reaction mixture of experimental tube measured at 340 nm against the reaction mixture of reference tube,
L - Length of optical path, cm. This parameter is 1.0 cm in most cases,
m - Weight of sample, g,
t - Reaction time, min,
V3 - The final volume of the reaction mixture, ml,
V4 - The volume of GBSS reaction mixture, ml,
V5 - The volume of an aliquot of GBSS reaction mixture taken after stopping the reaction by boiling, ml,
V6 - The volume of starch granules suspension, ml,
V7 - The volume of the starch granules suspension aliquot added to the reaction mixture, ml. - In this protocol, we did not measure protein content in starch granule suspension and this is why GBSS activity in the frame of this protocol can be calculated on g of fresh weight basis only.
- Enzyme activity of starch synthase (both SSS and GBSS) was expressed in enzyme units. In terms of this protocol, one enzyme unit (U) of starch synthase (both SSS and GBSS) is determined as the amount of enzymatic activity that converts 1 micromole of ADP-glucose into starch and 1 micromole of ADP per 1 min at pH 7.5 and at 30 °C.
- AGPase
Recipes
- Extraction buffer
100 mM HEPES-NaOH (pH 7.5)
8 mM MgCl2
2 mM EDTA
1 mM DTT
12.5% (v/v) glycerol
5% (w/v) polyvinylpyrrolidone (PVP-10) - Stock solutions
- ADP-glucose, disodium salt (MW = 633.31), 20 mM
ADP-glucose stock solution was prepared in the stock container. 0.79 ml of sterile water was added directly to a bottle containing 10 mg of the powder. Solution was aliquoted in small amounts and stored at -20 °C to avoid repetitive thawing. - NADP, sodium salt hydrate (MW = 765.39, anhydrous basis), 10 mM
7.8 mg of NADP was dissolved in 1 ml sterile water. The solution was aliquoted in small amounts and stored at -20 °C to avoid repetitive thawing. Being frozen, NADP solution is stable for about a year. - PEP monosodium salt hydrate (MW = 190.02, anhydrous basis), 20 mM
3.9 mg of PEP were dissolved in 1 ml sterile water. The solution was aliquoted in small amounts and stored at -80 °C to avoid repetitive thawing.
- ADP-glucose, disodium salt (MW = 633.31), 20 mM
- Enzymatic activities reaction mixtures
- AGPase activity reaction mixture (Table 1)
Note: If AGPase activity is low (Abs340 is below 0.05), the volume of enzyme extract can be increased at the expense of corresponding decrease of dH2O volume added.
Table 1. Composition of AGPase activity reaction mixture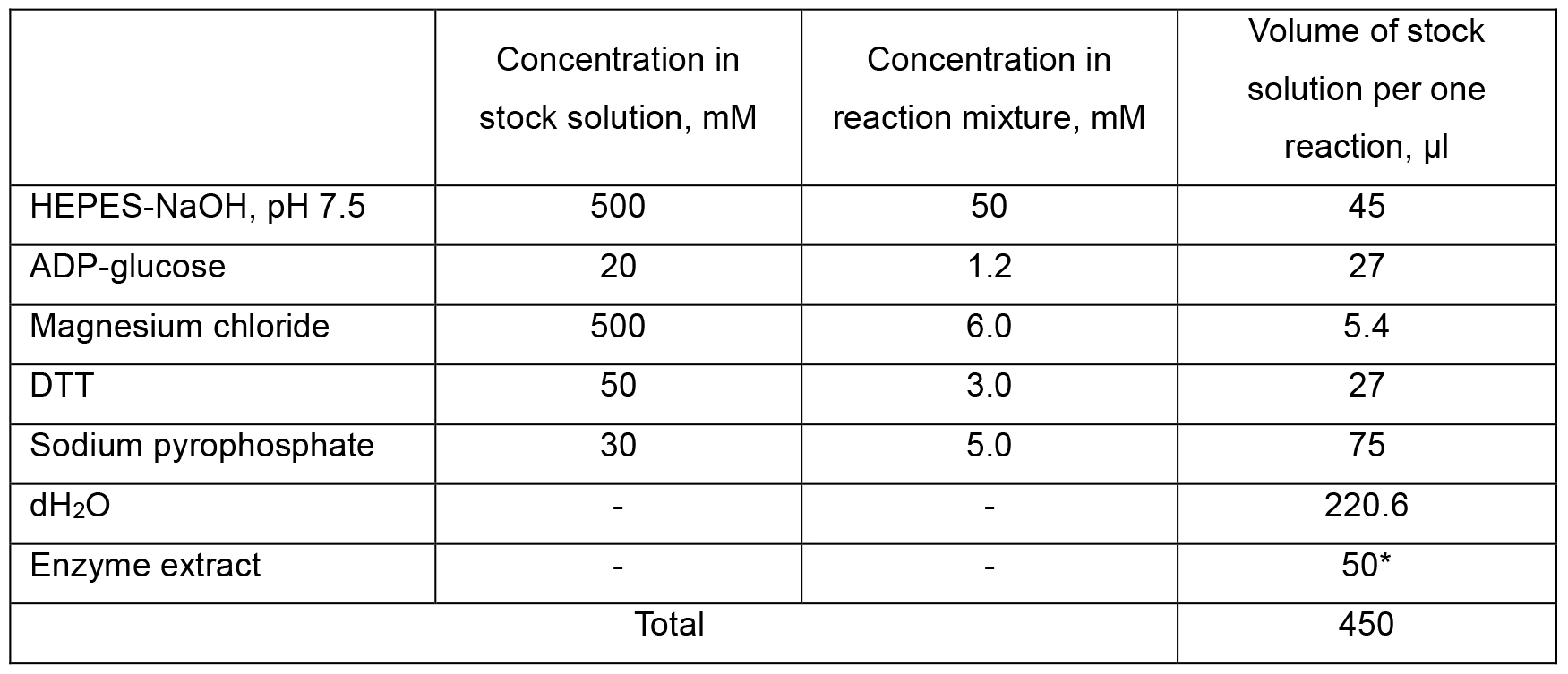
- Starch synthase activity reaction mixture (Table 2)
Note: If SSS/GBSS activity is low (Abs340 is below 0.05), the volume of enzyme extract/starch granule suspension can be increased at the expense of corresponding decrease of dH2O volume added. In case of SSS, more concentrated DDT can be used. For example, in case of 250 mM stock, 27 μl instead of 135 μl of DTT stock has to be added and 308 μl instead of 200 μl of enzyme extract can be added to the reaction mixture.Table 2. Composition of starch synthase activity reaction mixture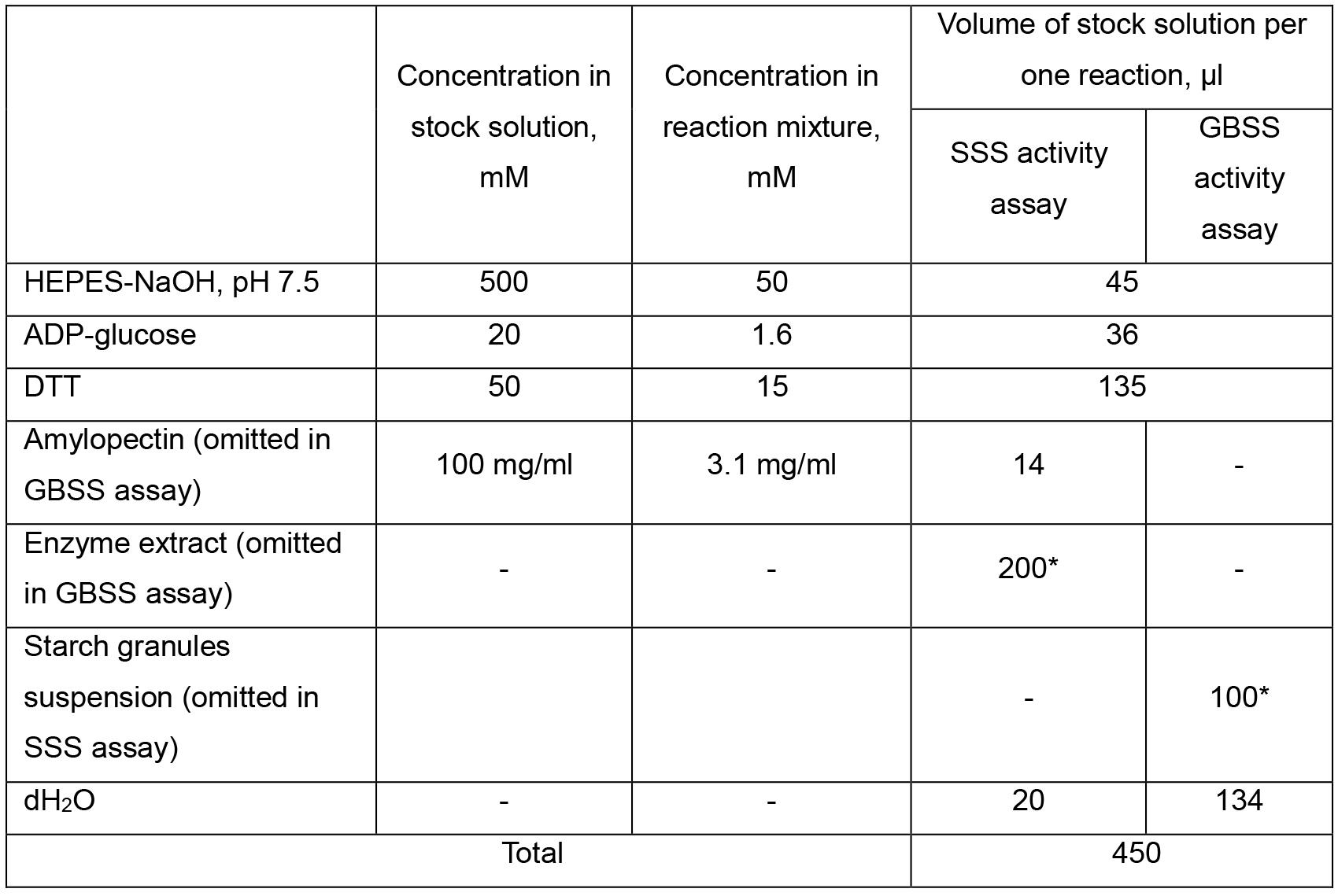
- Solution 1 for starch synthase activity assay (Pyruvate kinase reaction mixture) (Table 3)
Note: Solution 1 should be prepared fresh every time while performing the assay.
Table 3. Composition of Solution 1 (Pyruvate kinase reaction mixture)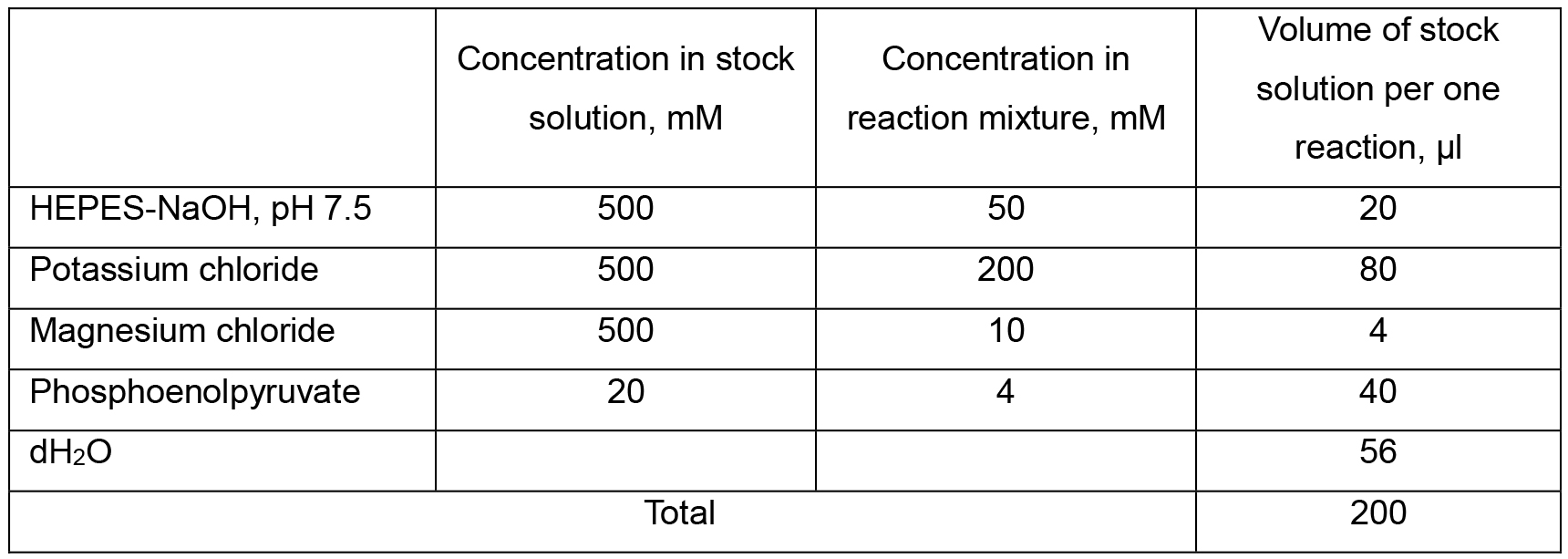
- Solution 2 for starch synthase activity assay (Glucose-6-phosphate dehydrogenase reaction mixture) (Table 4)
Note: Solution 2 should be prepared fresh every time while performing the assay.
Table 4. Composition of Solution 2 (G6PDH reaction mixture)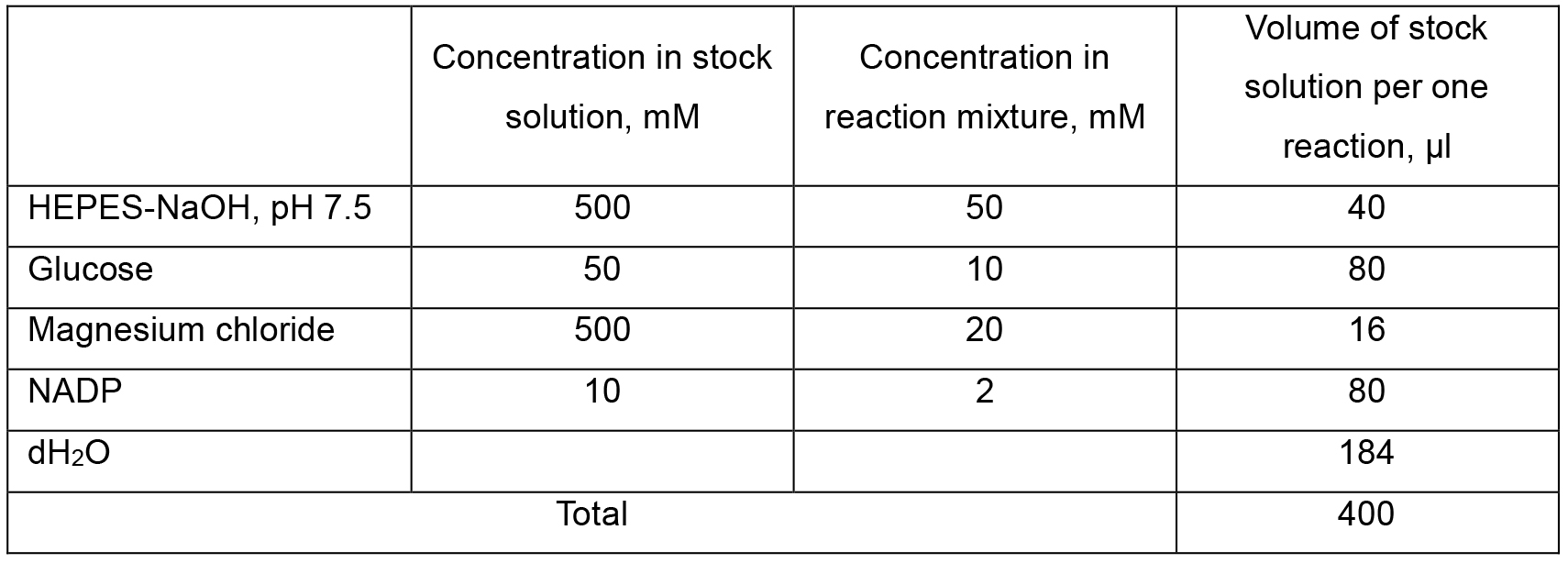
- AGPase activity reaction mixture (Table 1)
Acknowledgments
This work was supported by a grant from the Natural Sciences and Engineering Research Council of Canada to BTA. The authors would like to acknowledge Ms. Nina Kulichikhina for her technical assistance. This protocol is adapted from Nakamura et al. (1989) and Schaffer and Petreikov (1997).
References
- Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Ghosh, H. P. and Preiss, J. (1966). Adenosine diphosphate glucose pyrophosphorylase. A regulatory enzyme in the biosynthesis of starch in spinach leaf chloroplasts. J Biol Chem 241(19): 4491-4504.
- Mukherjee, S., Liu, A., Deol, K. K., Kulichikhin, K., Stasolla, C., Brule-Babel, A. and Ayele, B. T. (2015). Transcriptional coordination and abscisic acid mediated regulation of sucrose transport and sucrose-to-starch metabolism related genes during grain filling in wheat (Triticum aestivum L.). Plant Sci 240: 143-160.
- Nakamura, Y., Yuki, K., Park, S. Y. and Ohya, T. (1989). Carbohydrate metabolism in the developing endosperm of rice grains. Plant and Cell Physiology 30(6): 833-839.
- Schaffer, A. A. and Petreikov, M. (1997). Sucrose-to-starch metabolism in tomato fruit undergoing transient starch accumulation. Plant Physiology 113(3): 739-746.
- Vos-Scheperkeuter, G. H., de Boer, W., Visser, R. G., Feenstra, W. J. and Witholt, B. (1986). Identification of granule-bound starch synthase in potato-tubers. Plant Physiology 82(2): 411-416.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kulichikhin, K., Mukherjee, S. and Ayele, B. T. (2016). Extraction and Assays of ADP-glucose Pyrophosphorylase, Soluble Starch Synthase and Granule Bound Starch Synthase from Wheat (Triticum aestivum L.) Grains . Bio-protocol 6(18): e1929. DOI: 10.21769/BioProtoc.1929.
Category
Plant Science > Plant biochemistry > Protein > Isolation and purification
Biochemistry > Protein > Isolation and purification
Plant Science > Plant biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










