- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Running Reward Conditioned Place Preference Task
Published: Vol 6, Iss 10, May 20, 2016 DOI: 10.21769/BioProtoc.1816 Views: 13537
Reviewed by: Masahiro MoritaShai BerlinAna Perez-Villalba

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Protocol for Measuring Free (Low-stress) Exploration in Rats
Wojciech Pisula and Klaudia Modlinska
Jan 20, 2020 4459 Views

Operant Vapor Self-administration in Mice
Renata C. N. Marchette [...] Khaled Moussawi
May 20, 2021 4569 Views

Construction of Activity-based Anorexia Mouse Models
Maria Consolata Miletta and Tamas L. Horvath
Aug 5, 2023 1772 Views
Abstract
The conditioned place preference (CPP) test is a standard pre-clinical behavioral tool used to study the motivational effects of drugs and non-drug treatments in experimental animals. The basic characteristic of this task involves the association of a particular environment and contextual cues with a reward stimulus, followed by the association of a different environment with the absence of the reward stimulus (Prus and Rosecrans, 2009). Besides the motor component, voluntary wheel running exercise also has a rewarding component, and has been suggested as a strong natural reinforcer. Consistent with this notion, rodents will readily begin to run when a wheel is introduced (Eikelboom and Mills, 1988; Looy and Eikelboom, 1989), will work by lever pressing to gain access to a running wheel (Pierce et al., 1986), and spend more time in a place previously associated with the aftereffects of running (Lett et al., 2000; Lett et al., 2001). More recently, we underscored an important role for the adipocyte-derived hormone leptin in midbrain dopamine neurons in the modulation of running reward (Fernandes et al., 2015). Here, we describe a CPP protocol to measure the rewarding aftereffects of wheel running exercise in mice.
Keywords: Physical activityMaterials and Reagents
Animals
- Floxed Stat3 mice (C57Bl6 background) [Osaka, Japan (Takeda et al., 1998)]
Note: All experiments were carried out in accordance with the guidelines and approval of the Institutional Animal Care Committee of the CHUM Research Center. Floxed Stat3 mice (C57Bl6 background) in which loxP sites flank exon 22 of the Stat3 gene that encodes a tyrosine residue (tyr705) essential for Stat3 activation, were graciously provided by Dr. Shizuo Akira [Osaka, Japan (Takeda et al., 1998)]. Female mice homozygous for the floxed Stat3 allele were crossed with male mice heterozygous for the floxed Stat3 allele and heterozygous for the DAT-Cre transgene [B6.SJL-Slc6a3tm1.1(cre)Bkmn/J] (Backman et al., 2006) to generate DAT-Cre-Stat3fl/fl mice and littermate controls (Stat3fl/fl or Stat3fl). Male mice were weaned at P28 and housed in a temperature and humidity controlled room that was maintained on a 12:12 hour reverse light/dark cycle. CPP experiment (pre-test, conditioning trials and post-test) was conducted in the dark phase of the cycle.
Equipment
- Low-profile wireless running wheel for mouse (Med Associates Inc., catalog number: ENV-044 )
- Three-compartment automated mouse CPP chambers (Med Associates Inc., catalog number: ENV-3013 )
Software
- Wheel Manager software (Med Associates Inc., catalog number: SOF-860)
Procedure
- Running reward
Singly-housed male mice (sample size of >10 mice, as rodents’ behavior tend to be variable; 11-14 weeks of age) had free access to home cage low-profile wireless running wheels for 3 weeks (Figure 1). Running wheels have a 15.5 cm (6.10 in) diameter and revolve on a bearing-mounted pin, which creates less friction. Wheel Manager software was used to collect and organize data. Following 3 weeks of ad libitum access to the wheels, mice were subjected to a CPP task using a three-compartment, automated mouse CPP apparatus (Figure 2). Each CPP apparatus consisted of three chambers: A black compartment with a stainless steel grid rod floor and a white compartment with a spaced stainless steel mesh floor, separated by a neutral center compartment with a grey finish and PVC floor.

Figure 1. Single-housed mouse running on the wheel
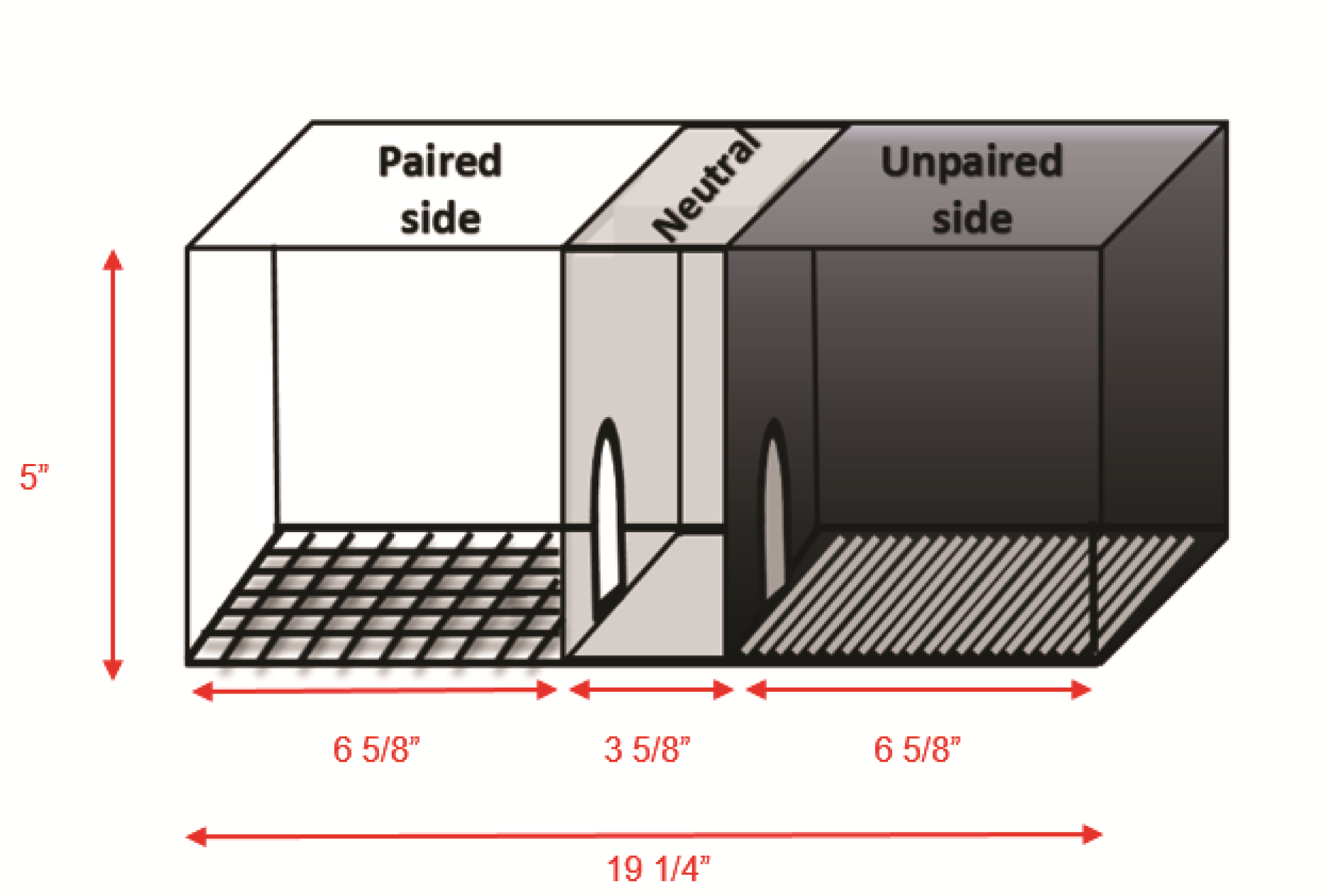
Figure 2. Illustrative schema of a three-compartment place preference chamber for mouse
- Pre-test
Mice were confined to both the black and white compartments of the CPP apparatus for 5-min to permit habituation to each compartment. Mice were then placed along the midline formed by the joining of the two chambers and allowed to move freely in the apparatus for 15-min during which the amount of time spent in each compartment was recorded (Table S1). All mice returned to their home cages afterwards. Of note, a mouse was considered to be in a chamber only when all four paws were in that chamber.
- Conditioning trials
Mice had 2-h access to a running wheel (paired trial) or a locked running wheel (unpaired trial) in their respective home cages, and were then confined to either the black or white compartment of the CPP apparatus for 30 min. Paired and unpaired conditioning trials took place in opposite compartments (counterbalanced across mice) on alternating days, thus each mouse ran every-other day. Conditioning trials were conducted for 14 days (Figure 3).
- Post-test
The CPP post-test occurred on the day after the last conditioning trial. For the post-test, mice were placed back in the open CPP apparatus for 15 min. Please note that animals only have access to the wheels in their home cages-running wheels where not inserted into the CPP apparatus. As previously mentioned, at the start of the CPP test, the mouse was placed along the midline formed by the joining of the two chambers. The amount of time spent in each compartment was once again recorded and the proportion of time spent in the paired side was compared to that obtained during the pre-test (Table S2). Importantly, each mouse was tested in the same CPP apparatus where it was trained, and CPP chambers were wiped with 70% EtOH in between animals.
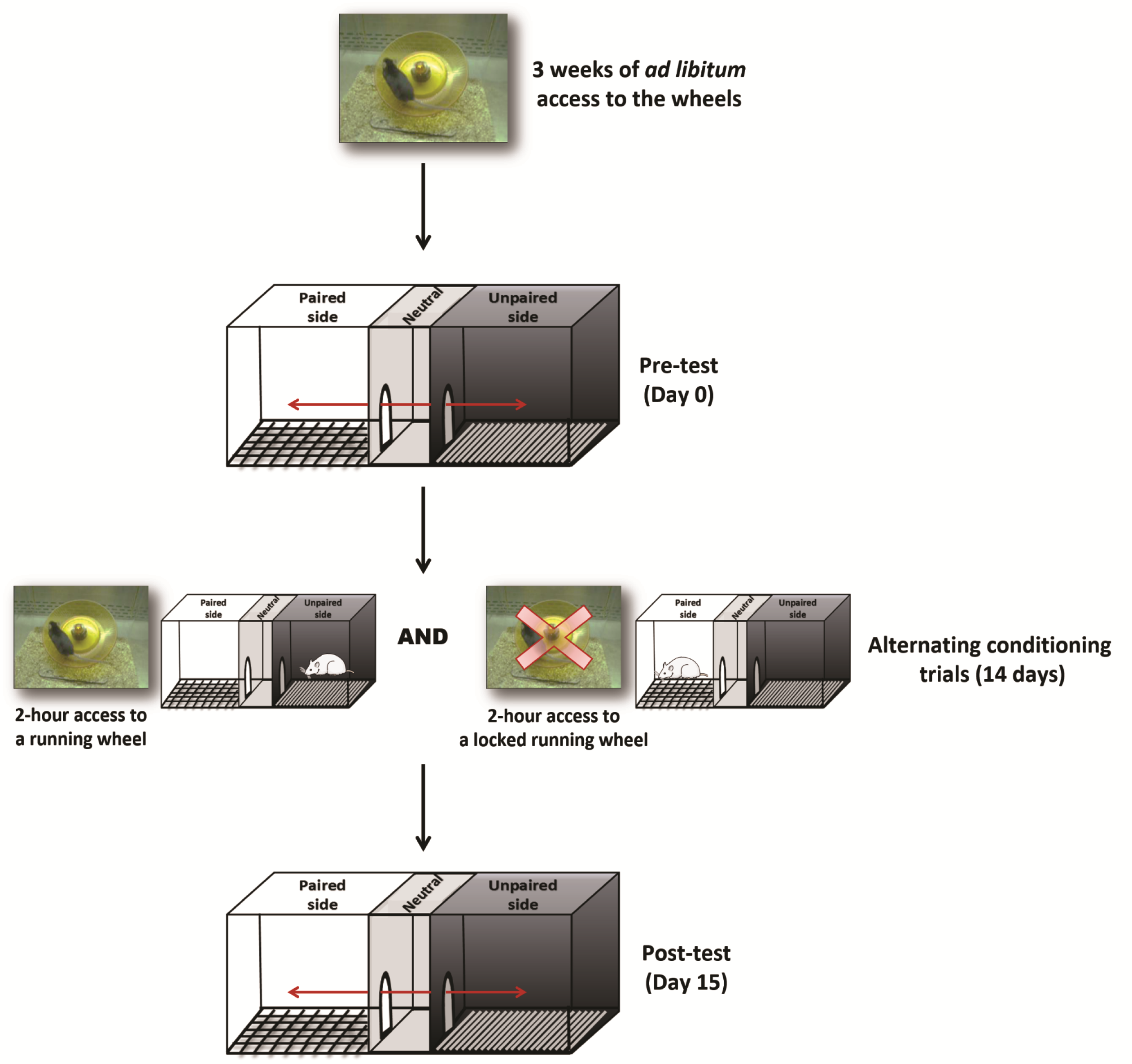
Figure 3. Running reward CPP task
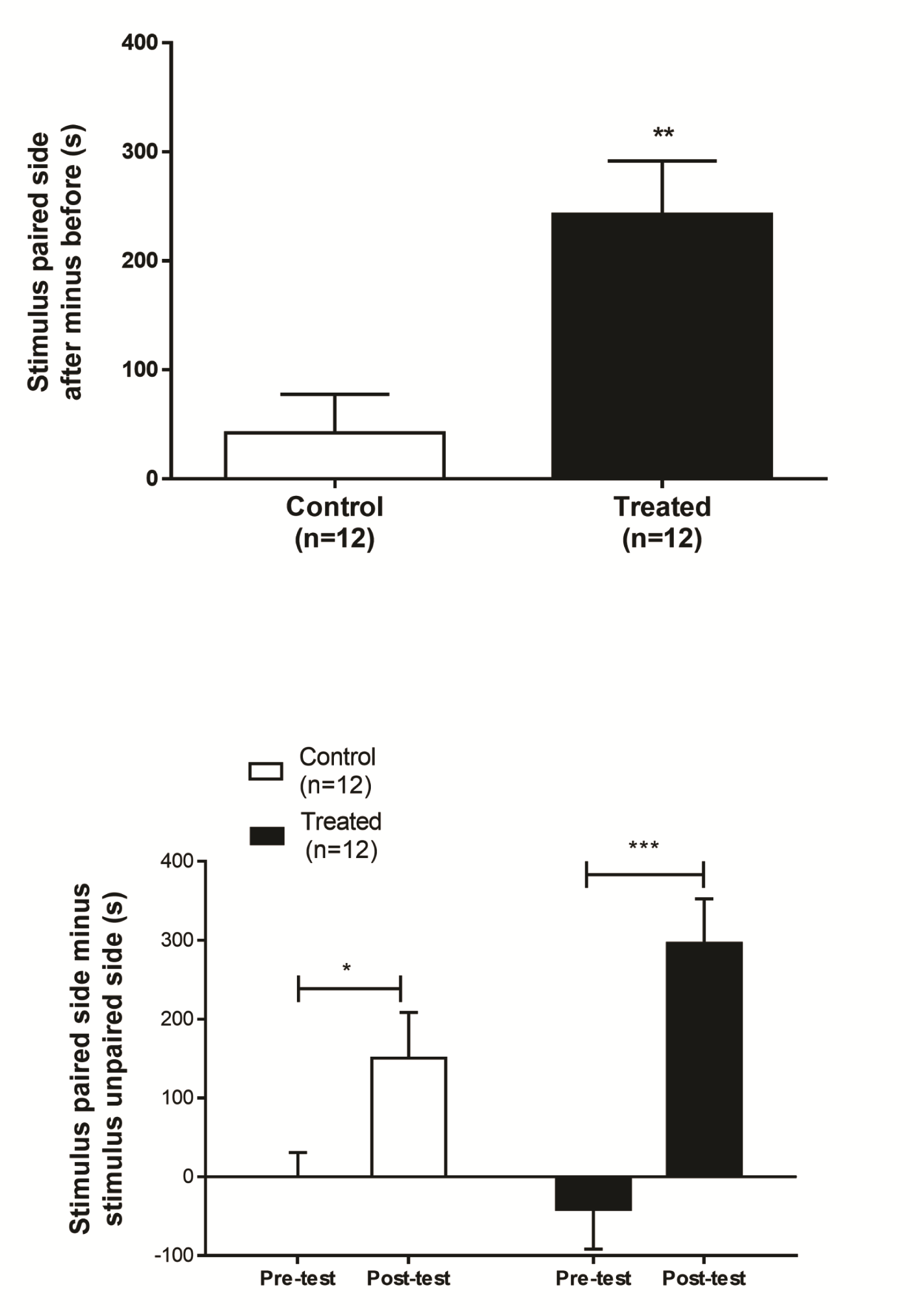
Figure 4. Illustrative graphs showing how to represent CPP data
Acknowledgments
This protocol was designed by S. F. and M. F. A. F., and was implemented in (Fernandes et al., 2015). This work was supported by an operating grant (MOP-123280) and New Investigator award to S. F. from the Canadian Institutes of Health Research, and doctoral fellowships to M. F. A. F. from the Canadian Diabetes Association and from the Department of Physiology, Universite de Montreal. We thank Shizuo Akira for the Stat3-floxed mice.
References
- Backman, C. M., Malik, N., Zhang, Y., Shan, L., Grinberg, A., Hoffer, B. J., Westphal, H. and Tomac, A. C. (2006). Characterization of a mouse strain expressing Cre recombinase from the 3' untranslated region of the dopamine transporter locus. Genesis 44(8): 383-390.
- Buccafusco, J. J. (2009). Conditioned Place Preference. In: Prus, A. J., James, J. R., Rosecrans, J. A. (eds). Methods of behavior analysis in neuroscience 2nd frontiers in neuroscience. Augusta CRC Press.
- Eikelboom, R. and Mills, R. (1988). A microanalysis of wheel running in male and female rats. Physiol Behav 43(5): 625-630.
- Fernandes, M. F., Matthys, D., Hryhorczuk, C., Sharma, S., Mogra, S., Alquier, T. and Fulton, S. (2015). Leptin suppresses the rewarding effects of running via STAT3 signaling in dopamine neurons. Cell Metab 22(4): 741-749.
- Lett, B. T., Grant, V. L. and Koh, M. T. (2001). Naloxone attenuates the conditioned place preference induced by wheel running in rats. Physiol Behav 72(3): 355-358.
- Lett, B. T., Grant, V. L., Byrne, M. J. and Koh, M. T. (2000). Pairings of a distinctive chamber with the aftereffect of wheel running produce conditioned place preference. Appetite 34(1): 87-94.
- Looy, H. and Eikelboom, R. (1989). Wheel running, food intake, and body weight in male rats. Physiol Behav 45(2): 403-405.
- Pierce, W. D., Epling, W. F. and Boer, D. P. (1986). Deprivation and satiation: The interrelations between food and wheel running. J Exp Anal Behav 46(2): 199-210.
- Takeda, K., Kaisho, T., Yoshida, N., Takeda, J., Kishimoto, T. and Akira, S. (1998). Stat3 activation is responsible for IL-6-dependent T cell proliferation through preventing apoptosis: generation and characterization of T cell-specific Stat3-deficient mice. J Immunol 161(9): 4652-4660.
Article Information
Copyright
© 2016 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Fernandes, M. F. A. and Fulton, S. (2016). Running Reward Conditioned Place Preference Task. Bio-protocol 6(10): e1816. DOI: 10.21769/BioProtoc.1816.
Category
Neuroscience > Behavioral neuroscience > Animal model > Mouse
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









