- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
The Application of Quercetin to Study the Effect of Hsp70 Silencing on Plant Virus Infection in Nicotiana benthamiana Plants
Published: Vol 5, Iss 23, Dec 5, 2015 DOI: 10.21769/BioProtoc.1675 Views: 8938
Reviewed by: Zhaohui LiuAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
General Maintenance and Reactivation of iSLK Cell Lines
Ariana C. Calderón-Zavala [...] Ekaterina E. Heldwein
Jun 5, 2025 1893 Views
Inducible HIV-1 Reservoir Reduction Assay (HIVRRA), a Fast and Sensitive Assay to Test Cytotoxicity and Potency of Cure Strategies to Reduce the Replication-Competent HIV-1 Reservoir in Ex Vivo PBMCs
Jade Jansen [...] Neeltje A. Kootstra
Jul 20, 2025 2460 Views
Assembly and Mutagenesis of Human Coronavirus OC43 Genomes in Yeast via Transformation-Associated Recombination
Brett A. Duguay and Craig McCormick
Aug 20, 2025 3038 Views
Abstract
Pepino mosaic virus (PepMV) is a mechanically-transmitted pathogen affecting tomato plants worldwide. Like with other plant viruses (Verchot, 2012), the heat shock cognate protein 70 homolog (Hsc70) was identified as an interactor of the PepMV coat protein (CP) (Mathioudakis et al., 2012). Here, we describe a pharmacological approach to silence Hsp70 in plants using quercetin (Mathioudakis et al., 2014), an Hsp70 protein expression flavonoid inhibitor (Hosokawa et al., 1990; Manwell and Heikkila 2007). In the case of Hsp70, this methodology represents a faster and easier approach than silencing of Hsp70 by reverse genetics assays, such as VIGS methodology. Fully expanded leaves of 2 to 3 weeks old Nicotiana benthamiana plants were infiltrated, using a syringe, with either quercetin (dissolved in DMSO) or DMSO (control plants). The plants were mechanically inoculated with PepMV virus inocula. The accumulation of Hsp70 and PepMV were analyzed on local leaves by immunoblot analysis 4 days post inoculation.
Materials and Reagents
- 1 ml syringe without a needle (BD, Nipro, catalog number: SY3 1 C100U ET )
- Mortar and pestles (Carl Roth GmbH + Co., catalog number: XL96.1 and XP01.1 )
- Pelet pestles (Nippon Genetics, catalog number: NG006 )
- 13 ml round base tubes (SARSTEDT AG & Co, catalog number: 62.515.006 )
- 1.5 ml microtubes (SARSTEDT AG & Co, catalog number: 72.690.001 )
- Nicotiana benthamiana leaves from 2 to 3 weeks old seedlings
- PepMV-Sp13 isolate infected material
- Quercetin hydrate (Sigma-Aldrich, catalog number: 337951 )
- Dimethyl sulfoxide (DMSO) (AppliChem GmbH, catalog number: A3009 )
- 10 mM Sodium carbonate (Na2CO3) (Sigma-Aldrich, Riedel-de Haen, catalog number: 31432 )
- Sodium dihydrogen phosphate monohydrate (0.5 M, pH 7.0) (AppliChem GmbH, catalog number: A3559 )
- Sodium phosphate dibasic (Na2HPO4) (Sigma-Aldrich, catalog number: S0876 )
- Potassium dihydrogen phosphate (KH2PO4) (Merck Millipore Corporation, catalog number: 104873 )
- Potassium chloride (KCl) (Sigma-Aldrich, Riedel-de Haen, catalog number: 31248 )
- Sodium Chloride (NaCl) (AppliChem GmbH, catalog number: A1149 )
- Magnesium Chloride 6-hydrate (MgCl2) (AppliChem GmbH, catalog number: A4425 )
- Silicon carbide (carborundum) 400 mesh (Sigma-Aldrich, catalog number: 357391 )
- Bromophenol blue (Sigma-Aldrich, catalog number: B-8026 )
- Coomassie Brilliant Blue (AppliChem GmbH, catalog number: A1092 )
- Glycerol (Sigma-Aldrich, catalog number: G6279 )
- Tris ultrapure (1.5 M, pH 8.8; 1 M, pH 6.8) (AppliChem GmbH, catalog number: A1086 )
- β-mercaptoethanol (Sigma-Aldrich, catalog number: M3148 )
- 30% 37.5:1 Acrylamide/Bis solution (Bio-Rad Laboratories, AbD Serotec®, catalog number: 161-0158 )
- 10% Ammonium persulfate (APS) (AppliChem GmbH, catalog number: A2941 )
- N, N, N´, N´-tetramethylethylenediamine (TEMED) (AppliChem GmbH, catalog number: A1148 )
- 10% Sodium dodecyl sulfate (SDS) (AppliChem GmbH, catalog number: A2263 )
- Glycine (AppliChem GmbH, catalog number: A1067 )
- Phenylmethanesulfonyl fluoride (PMSF) (Sigma-Aldrich, catalog number: P7626 )
- Methanol (Thermo Fisher Scientific, catalog number: M/4000/17 )
- Acetic acid (Sigma-Aldrich, catalog number: 33209 )
- PepMV CP polyclonal antibody (Neogen/Adgen Phytodiagnostics, catalog number: 1127-01 )
- HSP70 monoclonal antibody (Enzo Life Sciences, Stressgen, catalog number: N27F3-4 )
- Anti-rabbit IgG, Alkaline phosphatase-conjugated antibody (Promega Corporation, catalog number: S3731 )
- Anti-mouse IgG, Alkaline phosphatase-conjugated antibody (Promega Corporation, catalog number: S3721 )
- BCIP/NBP Color Development Substrate (Promega Corporation, catalog number: S3771 )
- PVDF membrane, Westran Clear Signal (Thermo Fisher Scientific, Whatmann, catalog number: 10485289 )
- Tween-20 (Sigma-Aldrich, catalog number: P2287 )
- Phosphate buffer saline (PBS) (see Recipes)
- Protein extraction buffer (see Recipes)
- 4x Laemmli buffer (see Recipes)
- SDS-PAGE buffers: Separation and Stacking gel buffers (see Recipes)
- Western Blot buffers: Running, Transfer, Wash and Detection buffers (see Recipes)
Equipment
- Plant growth chamber (25 °C, light:dark =16:8 h)
- Biofuge stratos highspeed table centrifuge (Thermo Fisher Scientific, Heraeus, catalog number: 75005282 )
- Fixed angle rotor #3334 (Thermo Fisher Scientific, Heraeus, catalog number: 75003334 )
- Fixed angle microliter rotor #3332 (Thermo Fisher Scientific, Heraeus, catalog number: 75003332 )
- Mini-PROTEAN 3 Cell Electrophoresis System (Bio-Rad Laboratories, AbD Serotec®, catalog number: 165-3301 )
- Mini Trans-Blot Electrophoretic Transfer Cell apparatus (Bio-Rad Laboratories, AbD Serotec®, catalog number: 170-3930 )
- Power Pack Supply model 200/2.0 (Bio-Rad Laboratories, AbD Serotec®, catalog number: 165-4761 )
- Western blot membranes were visualized using the Gel DocTM XR Molecular Imager & System (Bio-Rad Laboratories, AbD Serotec®, catalog number: 170-8195 EDU )
- Julabo Water-bath TW12 (Sigma-Aldrich, catalog number: Z615498 )
Note: Pricing & availability is not currently available.
Software
- Quantification of the band intensity as absorbance units was conducted by Quantity One analysis software (Bio-Rad Laboratories, AbD Serotec®, catalog number: 170-9600)
Procedure
- Preparation of PepMV inocula
Infected freeze dried material with the PepMV-Sp13 isolate (Aguilar et al., 2002, kindly provided from Dr. M. Aranda, CSIC-CEBAS Murcia, Spain) was homogenized with a ratio 1:10 (w/v) in NaH2PO4 0.5 M phosphate buffer using a mortar and pestle, and used as a fresh virus inocula. - Silencing of Hsp70 by quercetin application
- Quercetin was easily dissolved in DMSO in order to prepare an initial stock of 200 mM. The quercetin stock was subsequently diluted in a final concentration of 1 mM using a 10 mM Na2CO3 solution (e.g., 5 μl of 200 mM quercetin per 1 ml). DMSO alone was diluted accordingly to quercetin using the 10 mM Na2CO3 solution. Stock and working solutions of quercetin were always freshly prepared.
- One fully expanded leaf of 2 to 3 weeks old N. benthamiana plants (Figure 1) was totally (entirely) infiltrated, from the bottom side, using a syringe (Figure 2) either with 1 mM quercetin solution or diluted DMSO (control plants). Approximately to infiltrate an entire fully expanded leaf 1 ml of the corresponding solution was used.
- Quercetin was easily dissolved in DMSO in order to prepare an initial stock of 200 mM. The quercetin stock was subsequently diluted in a final concentration of 1 mM using a 10 mM Na2CO3 solution (e.g., 5 μl of 200 mM quercetin per 1 ml). DMSO alone was diluted accordingly to quercetin using the 10 mM Na2CO3 solution. Stock and working solutions of quercetin were always freshly prepared.
- Viral infection by rub inoculation
- One hour after the plant infiltration (kept at room temperature), the treated leaves with quercetin or DMSO were slightly dusted with carborundum.
- Five minutes later the leaves were mechanically inoculated with equal amount (~30 μl) of freshly prepared virus inocula. After the inoculation, plants were kept at growth chambers for four days at 25 °C and 16-h light and 8-h dark cycle.

Figure 1. N. benthamiana leaf before infiltration with DMSO or quercetin (left panel), and DMSO- or quercetin-treated N. benthamiana plants 4 days after PepMV rub inoculation (arrows indicate the DMSO- or quercetin-infiltrated leaves)
Figure 2. The left panel shows the infiltration method by syringe in N. benthamiana leaves (bottom side) and the right panel shows the leaves after infiltration
- One hour after the plant infiltration (kept at room temperature), the treated leaves with quercetin or DMSO were slightly dusted with carborundum.
- Analysis of the Hsp70 expression levels in Hsp70-silenced plants and the effect in viral Infection
- Four days post PepMV inoculation the leaves of N. benthamiana plants treated with quercetin or DMSO were collected (Figure 1). Control plants did not show any phenotype.
- Total proteins were extracted from 0.1 g of plant tissue using 300 μl of protein extraction buffer. Briefly: after the grinding of the infiltrated leaves to a fine powder using liquid nitrogen with a mortar and pestle, 0.1 g of tissue powder was homogenized using 300 μl of extraction buffer in 1.5 ml microtube (using pellet pestle sticks) and good vortex (2 min). 150 μl were mixed with 50 μl of 4x Laemmli buffer and boiled at 95 °C for 5 min. After a centrifugation at 12,000 rpm for 1 min the supernatant of the samples was used in the following steps.
- Small Rubisco unit protein was used for quantification of equal loading of total protein extracts in Coomassie Brilliant Blue staining (Figure 3C). After the SDS-polyacrylamide gel electrophoresis (PAGE) on 12% gels, the proteins were electrophoretically transferred onto PVDF membrane. Western Blot analysis was performed under standard conditions. Briefly, the blotted membranes were firstly incubated with the blocking buffer (1x TBS-T, 4% milk) for 1 hour at room temperature and subsequently with the primary antibodies (all conjugated to alkaline phosphatase [AP]), overnight at 4 °C. The following morning the membranes were washed 3 times and then incubated with the secondary AP-conjugated antibodies for 1 h at room temperature. Finally, after washing the membranes 3 times, the blotted proteins were detected using the NBT/BCIP substrate.
- In this given example, the results showed that the application of quercetin for silencing Hsp70 reduced its protein levels up to 73% (Figure 3A). This reduction of Hsp70 in the case of our example on PepMV infection had a corresponding negative effect on viral accumulation up to 92% (Figure 3B).
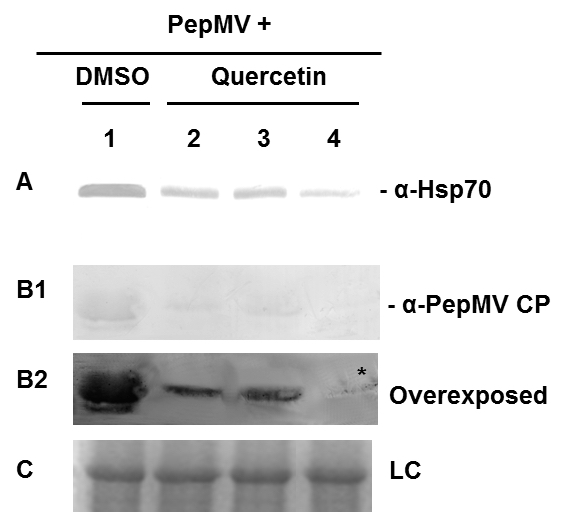
Figure 3. Immunoblot analysis of Hsp70 expression levels and PepMV accumulation in PepMV-inoculated leaves treated either with DMSO (lane 1) or quercetin (lanes 2 to 4, corresponding to different replicate plants), using α-Hsp70 (A panel) and α-PepMV CP (B1 panel) antibodies. B2 panel is an overexposure of B1 and the asterisk indicates the faint CP band. In panel C the small unit of Rubisco protein stained with Coomassie brilliant blue served as protein loading control (LC).
- Four days post PepMV inoculation the leaves of N. benthamiana plants treated with quercetin or DMSO were collected (Figure 1). Control plants did not show any phenotype.
Recipes
- 10x PBS
40 g NaCl
1 g KCl
7.2 g Na2HPO4
1.2 g KH2PO4
dd H2O to 1 L
Fix pH to 6.8 - Protein extraction buffer
For 10 ml: Mix 1 ml 10x PBS
(40 g NaCl, 1 g KCl, 7.2 g Na2HPO4, 1.2 g KH2PO4, dd H2O to 1 L, fix pH to 6.8)
2% β-mercaptoethanol
1 mM PMSF - 4x Laemmli buffer
250 mM Tris-HCl (pH 6.8)
8% SDS
40% glycerol
20% β-mercaptoethanol
0.02% Bromophenol Blue - SDS-PAGE buffers
- Separation gel:
0.375 M Tris-HCl (pH 6.8)
12% Acrylamide/Bis solution
0.1% SDS
0.1% APS
0.01% TEMED - Stacking gel:
0.125 M Tris-HCl (pH 6.8)
4% acrylamide/Bis solution
0.1% SDS
0.1 M APS
0.01% TEMED
- Separation gel:
- Western Blot buffers
- Running buffer
3 g Tris base
14.4 g glycine
1 g SDS
dd H2O to 1 L, fix pH to 8.3 - Transfer buffer (1x TBS)
3.03 Tris base
14.4 g glycine
200 ml methanol
dd H2O to 1 L - 1x TBS buffer
6.57 g Tris base
8.76 g NaCl
dd H2O to 1 L, fix pH to 7.4 - Wash buffer (1x TBS-T)
1x TBS
10% Tween-20
Antibodies were diluted in 1x TBS-T and 4% milk powder - Detection buffer
0.1 M Tris base
0.15 M NaCl
1 mM MgCl2
33 μl NBT and 16.5 μl BCIP substrates were mixed in 5 ml of detection buffer
- Running buffer
Acknowledgments
This protocol was adopted from previous work (Hafrén et al., 2010; Wang et al., 2009). Matthaios Mathioudakis was a recipient of an Onassis Foundation doctoral fellowship.
References
- Aguilar, J. M., Hernandez-Gallardo, M. D., Cenis, J. L., Lacasa, A. and Aranda, M. A. (2002). Complete sequence of the Pepino mosaic virus RNA genome. Arch Virol 147(10): 2009-2015.
- Hafren, A., Hofius, D., Ronnholm, G., Sonnewald, U. and Makinen, K. (2010). HSP70 and its cochaperone CPIP promote potyvirus infection in Nicotiana benthamiana by regulating viral coat protein functions. Plant Cell 22(2): 523-535.
- Hosokawa, N., Hirayoshi, K., Nakai, A., Hosokawa, Y., Marui, N., Yoshida, M., Sakai, T., Nishino, H., Aoike, A., Kawai, K. and et al. (1990). Flavonoids inhibit the expression of heat shock proteins. Cell Struct Funct 15(6): 393-401.
- Mathioudakis, M. M., Rodriguez-Moreno, L., Sempere, R. N., Aranda, M. A. and Livieratos, I. (2014). Multifaceted capsid proteins: multiple interactions suggest multiple roles for Pepino mosaic virus capsid protein. Mol Plant Microbe Interact 27(12): 1356-1369.
- Mathioudakis, M. M., Veiga, R., Ghita, M., Tsikou, D., Medina, V., Canto, T., Makris, A. M. and Livieratos, I. C. (2012). Pepino mosaic virus capsid protein interacts with a tomato heat shock protein cognate 70. Virus Res 163(1): 28-39.
- Verchot, J. (2012). Cellular chaperones and folding enzymes are vital contributors to membrane bound replication and movement complexes during plant RNA virus infection. Front Plant Sci 3: 275.
- Wang, R. Y., Stork, J. and Nagy, P. D. (2009). A key role for heat shock protein 70 in the localization and insertion of tombusvirus replication proteins to intracellular membranes. J Virol 83(7): 3276-3287.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Mathioudakis, M. M. and Livieratos, I. (2015). The Application of Quercetin to Study the Effect of Hsp70 Silencing on Plant Virus Infection in Nicotiana benthamiana Plants. Bio-protocol 5(23): e1675. DOI: 10.21769/BioProtoc.1675.
Category
Microbiology > Microbe-host interactions > Virus
Plant Science > Plant immunity > Host-microbe interactions
Plant Science > Plant molecular biology > Protein
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











