- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Generation of Mammalian Host-adapted Leptospira interrogans by Cultivation in Peritoneal Dialysis Membrane Chamber Implantation in Rats
Published: Vol 5, Iss 14, Jul 20, 2015 DOI: 10.21769/BioProtoc.1536 Views: 9215
Reviewed by: Fanglian HeKanika Gera

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Induction, Isolation and Counting of Akinetes in Aphanizomenon ovalisporum
Assaf Sukenik [...] Dotan Sela
May 20, 2016 8513 Views
Isolation of Commensal Escherichia coli Strains from Feces of Healthy Laboratory Mice or Rats
Tingting Ju and Benjamin P. Willing
Mar 20, 2018 10915 Views

Detachment Procedure of Bacteria from Atmospheric Particles for Flow-cytometry Counting
Carolina M. Araya [...] Isabel Reche
Jun 20, 2019 5593 Views
Abstract
Leptospira interrogans can infect a myriad of mammalian hosts, including humans (Bharti et al., 2003; Ko et al., 2009). Following acquisition by a suitable host, leptospires disseminate via the bloodstream to multiple tissues, including the kidneys, where they adhere to and colonize the proximal convoluted renal tubules (Athanazio et al., 2008). Infected hosts shed large number of spirochetes in their urine and the leptospires can survive in different environmental conditions before transmission to another host. Differential gene expression by Leptospira spp. permits adaption to these new conditions. Here we describe a protocol for the cultivation of Leptospira interrogans within Dialysis Membrane Chambers (DMCs) implanted into the peritoneal cavities of Sprague-Dawley rats (Caimano et al., 2014). This technique was originally developed to study mammalian adaption by the Lyme disease spirochete, Borrelia burgdorferi (Akins et al., 1998; Caimano, 2005). The small pore size (8,000 MWCO) of the dialysis membrane tubing used for this procedure permits access to host nutrients but excludes host antibodies and immune effector cells. Given the physiological and environmental similarities between DMCs and the proximal convoluted renal tubule, we reasoned that the DMC model would be suitable for studying in vivo gene expression by L. interrogans. In a 20 to 30 min procedure, DMCs containing virulent leptospires are surgically-implanted into the rat peritoneal cavity. Nine to 11 days post-implantation, DMCs are explanted and organisms recovered. Typically, a single DMC yields ~109 mammalian host-adapted leptospires (Caimano et al., 2014). In addition to providing a facile system for studying the transcriptional and physiologic changes pathogenic L. interrogans undergo within the mammal, the DMC model also provides a rationale basis for selecting new targets for mutagenesis and the identification of novel virulence determinants.
Caution: Leptospira interrogans is a BSL-2 level pathogen and known to be excreted in the urine of infected animals. Animals should be handled and disposed of using recommended Animal Biosafety Levels (ABSL) for infectious agents using vertebrate animal guidelines.
Note: All protocols using live animals must conform to governmental regulations regarding the care and use of laboratory animals. The success of this protocol is dependent on the proper use of aseptic techniques during all stages of both dialysis membrane chamber preparation and animal surgery.
Materials and Reagents
- Adult female Sprague-Dawley rats (175-200 g)
- EMJH medium (prepared as described in Supplementary data)
- Bovine serum albumin (BSA)
EMJH+BSA (10 mg/ml BSA, BSA concentration 20 mg/ml) (Millipore, catalog number: 810037 )
Note: The quality of the albumin used for the cultivation of leptospires is critical. Please verify that the albumin used for DMC implantation supports the growth of virulent leptospires under standard in vitro growth conditions. Other sources of BSA were tested (e.g., Millipore, catalog number: 840644 and Sigma-Aldrich, catalog number: A-9647 ) with similar results. - Leptospira interrogans, logarithmic phase, grown under standard in vitro conditions (28-30 °C) (Zuerner, 2005)
- Ultrapure water (deionized, distilled) (e.g., Milli-Q)
- 1 mM EDTA (pH 8.0)
- Regenerated cellulose dialysis membrane tubing (SpectrumTM Spectra/PorTM 1 RC, 6,000 to 8,000 MWCO, 32 mm width) (Spectrum Labs, catalog number: 132655 )
- Filter units (0.22 µm, 250 ml, sterile) (e.g., Millipore Stericup® filter unit)
- Ketamine/xylazine anesthetic cocktail (40-80 mg per kg/ 5-10 mg per kg, administered intramuscularly)
- Ophthalmic ointment (e.g., Puralube)
- Betadine® surgical scrub solution
- Carprofen (5-10 mg per kg, administered subcutaneously)
- Ethanol (70%)
- Ketamine/xylazine anesthetic cocktail (see Recipes)
- EMJH + BSA medium (see Recipes)
Equipment
- Surgical gloves (individually wrapped, sterile)
- Disposable serological pipets (10 ml sterile, individually-wrapped)
- Surgical drape (cut into 12 in (46 cm) squares, sterile)
- Gauze (4 x 4 in sterile)
- Microscope equipped with a dark field condenser (e.g., Olympus, model: BX40 )
- Three 2-L Pyrex beakers (each containing a magnetic stir bar)
- Hot plate with stirring option
- Extra-long blunt end forceps (sterile)
- Pipet filler (e.g., Drummond Scientific, model: Pipet-Aid )
- Surgical instrument pack, one per animal, sterilized prior to use and kept within sterile package:
Scalpel blades (No. 10)
Scalpel blade holder
Scissors, iris, 4 in (~10.2-cm)
Tissue forceps, 5 ½ in (~14 cm) (1x 2-tooth dissecting or Adson-Brown)
Tissue forceps, 5 ½ in (~14 cm) (blunt-end)
Needle holder forceps with built-in scissors (e.g., Olsen Hegar) (5 ½ in)
(~14 cm)
Suture, Ethicon 40, SH 1, 27 in (~68.6 cm) coated Vicryl, violet-braided suture
Autoclip® 9-mm stainless steel wound closure clips and applicator - Biological safety cabinet (Biosafety Level 2, model: BSL2 )
- Circulating warm-water blanket and pump
- Electric hair/fur clippers
- Glass bead sterilizer (e.g., Braintree Scientific, model: Germinator 500 ) (optional)
Procedure
- Preparation of sterile dialysis membrane tubing
- Fill three 2-Liter Pyrex beakers as follows:
Beaker 1: 1 - 1.5 L Ultrapure water
Beaker 2: 1 - 1.5 L 1 mM EDTA (pH 8.0)
Beaker 3: 1 - 1.5 L Ultrapure water - Add a magnetic stir bar to each beaker, cover with heavy-duty aluminum foil and autoclave.
Note: To ensure sterility, beakers should be autoclaved on the same day at the procedure. - Place each beaker on hot plate and bring to a rolling boil with constant stirring.
- Wearing sterile gloves, cut dialysis membrane tubing into strips 7-9 inches (~18-23 cm) in length using sterile scissors. Cut one strip per animal plus 1-2 extra.
- Gently tie off one end of the tubing using a simple overhand knot by forming a loop at the top of the DMC and passing the free end of the tubing through the loop. Trim away excess tubing from the tied end using sterile scissors.
- Place the tied tubing into Beaker 1 and replace the aluminum foil cover. Be sure to keep the foil cover loose enough to allow steam to escape. Boil tubing for 20 min. with constant stirring.
Note: Tubing should remain submerged at all times. - Using sterile extra-long forceps, transfer tubing to Beaker 2. Replace aluminum foil cover and boil tubing for 20 min. with constant stirring.
- Repeat step A7 using Beaker 3.
- Once cool to the touch, transfer Beaker 3 to a Biological safety cabinet (BSC). To ensure sterility, the beaker containing the tubing should remain in the BSC.
Note: Dialysis membrane tubing may be prepared several days in advance and stored at 4 °C. After boiling, transfer dialysis membrane tubing to the bottom portion of a 0.22 µm Stericup® Filter Unit (Millipore) using sterile blunt tip forceps. Replace the filter unit top and filter in ~100-200 ml of Ultrapure-Q water from Beaker 3. Seal the bottom portion of the filter unit using the sterile cap provided by the manufacturer. The container should be opened only within the BSC.
- Fill three 2-Liter Pyrex beakers as follows:
- Preparation of leptospires
- Use a standard low passage in vitro culture of virulent L. interrogans, kept at 28-30 °C in EMJH medium, before it reaches stationary phase in the growth curve.
- Just prior to starting the surgical procedure, count bacteria by dark field microscopy.
- Dilute culture to 106 organisms/ml using fresh EMJH medium.
- Working in the BSC, transfer 10 ml of EMJH+BSA to sterile 50-ml conical tubes. Prepare one conical tube per DMC.
Note: It is important that the EMJH medium used for DMCs is supplemented with additional BSA to maintain the appropriate osmotic pressure. - Add 0.1 ml of dilute culture to each 50-ml conical tube containing EMJH+BSA medium (Final density, 104 organisms/ml).
- Use a standard low passage in vitro culture of virulent L. interrogans, kept at 28-30 °C in EMJH medium, before it reaches stationary phase in the growth curve.
- Preparation of DMCs
- Working in the BSC, place several disposable, individually-wrapped serological pipets within the surgical field. Lay down a sterile drape to use as a workspace. Clean the surface of an automatic pipeter using 70% ethanol and place on the sterile drape. After donning a new pair of sterile surgical gloves, loosen the cap of a 50-ml conical tube.
- While still wearing gloves, carefully remove a strip of tubing from its storage container using blunt end forceps. Use your free hand to hold the tubing between index finger and thumb. With other hand, remove the cap from the 50-ml conical tube containing diluted bacteria and transfer up to 9 mls to the tubing using a 10-ml disposable serological pipette. While continuing to hold the open end, flatten the unfilled tubing between the fingers of your free hand to eliminate any air bubbles.
- Gently twist the top end of the tubing (1-2 turns) to remove any remaining void volume and close off the top of the DMC Use your thumb and index finger to hold on to the twisted area to prevent liquid from leaking out of the DMC while you tie off the top. Tie off the open end using a simple overhand knot (e.g., form a loop at the top of the DMC, passing the free end of the tubing through the loop, then, without releasing your thumb and index finger, gently tighten the knot). Trim away excess tubing from the tied end using sterile scissors.
Note: Try to make the DMC as taut as possible; this will help to maneuver the chamber into the peritoneal cavity during surgery. The resulting filled DMC should be ~1.5 - 2 in. Larger DMCs may interfere with normal intestinal or bladder functions. - Place the filled DMC into a sterile 50-ml conical tube containing ~5-7 ml of fresh EMJH+ BSA medium.
- Repeat steps C3-4 until all bags are filled then proceed directly to the surgical procedure.
Note: It is not recommended that filled dialysis membrane chambers be stored for extended periods of time as this may increase the risk of contamination.
- Working in the BSC, place several disposable, individually-wrapped serological pipets within the surgical field. Lay down a sterile drape to use as a workspace. Clean the surface of an automatic pipeter using 70% ethanol and place on the sterile drape. After donning a new pair of sterile surgical gloves, loosen the cap of a 50-ml conical tube.
- Peritoneal implantation procedure.
- Anesthetize animal by intramuscular injection with the ketamine/xylazine cocktail.
- Apply a small amount of ophthalmic ointment to each eye.
- Administer preoperative analgesia (e.g., Carprofen).
- Shave the abdomen and prepare surgical site (e.g., successive washes with Betadine® Surgical solution followed by alcohol).
- Cover the surgical site with sterile gauze and transfer the animal to the BSC in a supine position.
- Prior to beginning the procedure, remove the sterile gauze covering the surgical site and perform a ‘toe pinch’ to ensure that the animal is properly anesthetized.
- Using a sterile scalpel blade, make a 5 cm incision through the skin only, starting ~2.5 cm below the ribcage, using the xiphoid process as a guide. Using tissue forceps, pull up the skin on either side of the incision and gently trim the fascia connecting the skin to the abdominal wall using a scalpel. Repeat on the other side.
- Use the same scalpel, make a small incision (~4 cm) in the abdominal wall, using the linea alba as guide. The incision should be clean and straight.
- Using tissue forceps, raise one side of the abdominal incision and place a DMC inside rat peritoneal cavity (Figure 1). Gently push the DMC towards either side of the peritoneal cavity to prevent it from being nicked/ruptured during suturing. Be sure to position so that it does not become entangled in the intestines or interfere with bladder expansion.
Figure 1. Implantation of DMC into rat peritoneal cavity- Close the abdominal incision site by suturing. Begin by suturing each end of the incision with double knots. Working upwards, close the remaining incision by placing sutures ~2-3 mm apart.
- Close the skin incision site using a contiguous line of AutoClip® wound clips (Figure 2). Note: Alternatively, the skin excision site may be closed using subcuticular stitches followed by liquid skin adhesive (e.g., Nexaband).
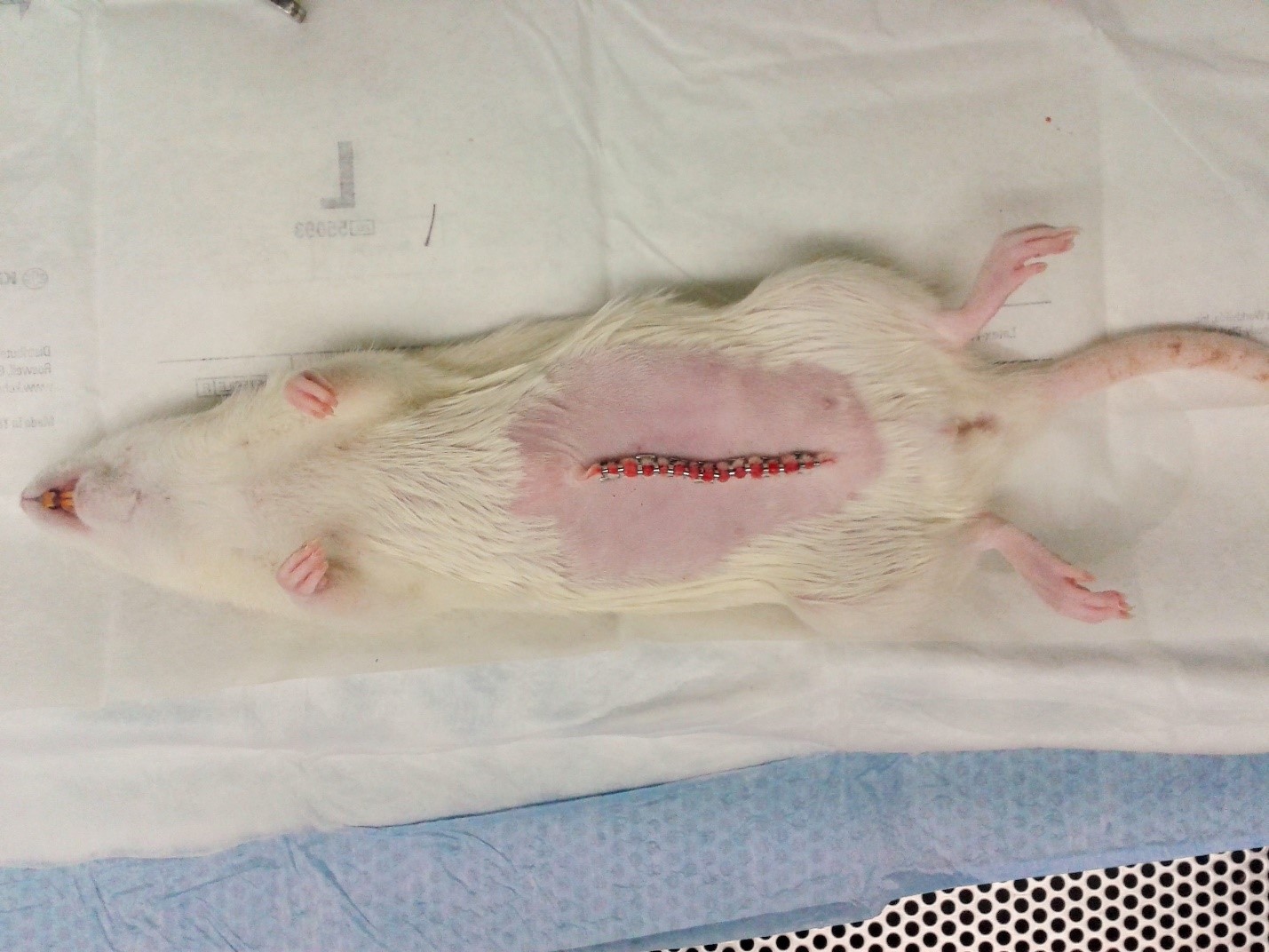
Figure 2. Closing the skin incision- Place the rat on top of a clean surgical drape in a clean cage containing fresh bedding. Place the cage on top of a circulating water heating pad to maintain the appropriate core body temperature. Monitor the animal continuously until alert and responsive.
- Analgesia should be administered for at least two days post-operatively.
- Animals should be monitored at least once daily for the first week post-operatively and then every other day thereafter. If animals show any sign of distress or discomfort, consult institutional veterinary staff immediately.
- Anesthetize animal by intramuscular injection with the ketamine/xylazine cocktail.
- Recovery of mammalian host adapted organisms from DMCs
- At 9 to 12 days after implantation, euthanize animals by CO2 asphyxiation.
- Place animal in a BSC in a supine position.
- Using sterile surgical scissors, expose the abdominal wall.
- Using sterile tissue forceps, lift one side of the sutured abdominal incision site and re-open using sterile scissors.
- Locate and remove the DMC using blunt-end forceps. Transfer to a sterile 50-ml conical tube.
- Using sterile forceps to hold the DMC by one knot, make a small cut in the tubing just below the knot. Slowly remove the contents of the DMC using a sterile disposable serological pipet. Transfer the DMC fluid to a sterile 15-ml conical tube. Alternatively, DMC contents can be removed using a syringe 18-G, 1 in. needle attached to a sterile 10-ml syringe. However, aspiration of leptospires by syringe may disrupt cells and/or cause cell lysis.
- Examine a small aliquot of the contents DMC fluid from each chamber under dark field microscopy.
- Proceed to downstream applications (e.g., RNA isolation, analysis by 1D and 2D SDS-PAGE).
- At 9 to 12 days after implantation, euthanize animals by CO2 asphyxiation.
Representative data
Figure 3. Virulent leptospires become mammalian host-adapted during growth within dialysis membrane chambers. Representative whole cell lysates of leptospires cultivated to late-logarithmic phase in EMJH medium at 30 °C in vitro (IV) and within dialysis membrane chambers (DMC) implanted into the peritoneal cavities of female Sprague-Dawley rats. A. Lysates were loaded according to the numbers of leptospires (5 x 106 per lane) or total protein (5 µg per lane) and stained with SYPRO Ruby gel stain. Arrows and asterisks are used to highlight examples of polypeptides whose expression appears to be increased or decreased, respectively, within DMCs compared to in vitro. Molecular mass markers are indicated on the left. B. Immunoblot analyses using rabbit polyclonal antisera directed against Sph2 (Matsunaga et al., 2007), LipL32 (Haake et al., 2000) and LipL41 (Shang et al., 1996). An arrow is used to indicate a band of the predicted molecular mass for SphH, a second, closely related sphingomyelinase in L. interrogans recognized by antiserum directed against Sph2 (Matsunaga et al., 2007; Carvalho et al., 2010). Image reproduced from Caimano et al. (2014).
Recipes
- Ketamine/xylazine anesthetic cocktail
Stock solutions
100 mg/ml ketamine-HCl
20 mg/ml xylazine-HCl - EMJH + BSA medium
Prepare 1-L of EMJH medium as described in Supplementary data and previously published (Zuerner, 2005)
Weight out 10 mg of BSA using an analytical balance
Slowly stir in 10 mg of BSA with constant stirring, avoiding bubbles, until the BSA is completely dissolved
Sterilize by filtration using 0.22 µm Stericup® Filter Unit
Stored at 4 °C
Acknowledgments
The authors would like to thank Ms. Anna Allard for her superb technical assistance and Dr. Justin Radolf his continued support of our work. This work is supported by NIH Grants AI029735 (MJC), Brazilian CNPq grants 481133/2011-9 and 483052/2012-4 (AJAM) and CNPq/SWE/CSF grant 246433/2012-4 (AAG).
References
- Akins, D. R., Bourell, K. W., Caimano, M. J., Norgard, M. V. and Radolf, J. D. (1998). A new animal model for studying Lyme disease spirochetes in a mammalian host-adapted state. J Clin Invest 101(10): 2240-2250.
- Athanazio, D. A., Silva, E. F., Santos, C. S., Rocha, G. M., Vannier-Santos, M. A., McBride, A. J., Ko, A. I. and Reis, M. G. (2008). Rattus norvegicus as a model for persistent renal colonization by pathogenic Leptospira interrogans. Acta Trop 105(2): 176-180.
- Bharti, A. R., Nally, J. E., Ricaldi, J. N., Matthias, M. A., Diaz, M. M., Lovett, M. A., Levett, P. N., Gilman, R. H., Willig, M. R., Gotuzzo, E., Vinetz, J. M. and Peru-United States Leptospirosis, C. (2003). Leptospirosis: a zoonotic disease of global importance. Lancet Infect Dis 3(12): 757-771.
- Caimano, M. J. (2005). Cultivation of Borrelia burgdorferi in dialysis membrane chambers in rat peritonea. Curr Protoc Microbiol Chapter 12: Unit 12C 13.
- Caimano, M. J., Sivasankaran, S. K., Allard, A., Hurley, D., Hokamp, K., Grassmann, A. A., Hinton, J. C. and Nally, J. E. (2014). A model system for studying the transcriptomic and physiological changes associated with mammalian host-adaptation by Leptospira interrogans serovar Copenhageni. PLoS Pathog 10(3): e1004004.
- Carvalho, E., Barbosa, A. S., Gomez, R. M., Oliveira, M. L., Romero, E. C., Goncales, A. P., Morais, Z. M., Vasconcellos, S. A. and Ho, P. L. (2010). Evaluation of the expression and protective potential of Leptospiral sphingomyelinases. Curr Microbiol 60(2): 134-142.
- Haake, D. A., Chao, G., Zuerner, R. L., Barnett, J. K., Barnett, D., Mazel, M., Matsunaga, J., Levett, P. N. and Bolin, C. A. (2000). The leptospiral major outer membrane protein LipL32 is a lipoprotein expressed during mammalian infection. Infect Immun 68(4): 2276-2285.
- Ko, A. I., Goarant, C. and Picardeau, M. (2009). Leptospira: the dawn of the molecular genetics era for an emerging zoonotic pathogen. Nat Rev Microbiol 7(10): 736-747.
- Matsunaga, J., Medeiros, M. A., Sanchez, Y., Werneid, K. F. and Ko, A. I. (2007). Osmotic regulation of expression of two extracellular matrix-binding proteins and a haemolysin of Leptospira interrogans: differential effects on LigA and Sph2 extracellular release. Microbiology 153(Pt 10): 3390-3398.
- Shang, E. S., Summers, T. A. and Haake, D. A. (1996). Molecular cloning and sequence analysis of the gene encoding LipL41, a surface-exposed lipoprotein of pathogenic Leptospira species. Infect Immun 64(6): 2322-2330.
- Zuerner, R. L. (2005). Laboratory maintenance of pathogenic Leptospira. Curr Protoc Microbiol Chapter 12: Unit 12E 11.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Grassmann, A. A., McBride, A. J. A., Nally, J. E. and Caimano, M. J. (2015). Generation of Mammalian Host-adapted Leptospira interrogans by Cultivation in Peritoneal Dialysis Membrane Chamber Implantation in Rats. Bio-protocol 5(14): e1536. DOI: 10.21769/BioProtoc.1536.
Category
Microbiology > Microbial cell biology > Cell isolation and culture
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link