- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
A Mouse Model of Preterm Brain Injury after Hypoxia-Ischemia
Published: Vol 5, Iss 13, Jul 5, 2015 DOI: 10.21769/BioProtoc.1526 Views: 12025
Reviewed by: Soyun KimEdgar Soria-GomezAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Conditioned Lick Suppression: Assessing Contextual, Cued, and Context-cue Compound Fear Responses Independently of Locomotor Activity in Mice
Youcef Bouchekioua [...] Yu Ohmura
Dec 5, 2022 1645 Views
In situ Microinflammation Detection Using Gold Nanoclusters and a Tissue-clearing Method
Fayrouz Naim [...] Masaaki Murakami
Apr 5, 2023 2677 Views
A One-Step Mouse Model of Parkinson’s Disease Combining rAAV-α-Synuclein and Preformed Fibrils of α-Synuclein
Santhosh Kumar Subramanya [...] Poonam Thakur
Dec 5, 2025 1660 Views
Abstract
The rodent hypoxia-ischemia (HI) model, referred to as the Vannucci model, is the most commonly used model for studying perinatal hypoxic-ischemic brain injury (Zhu et al., 2009; Vannucci and Hagberg, 2004). In the Vannucci model, brain injury is acquired by combining a permanent unilateral common carotid artery ligation with subsequent exposure to hypoxia (Rice et al., 1981). The Vannucci model was originally developed in rat pups at postnatal day (PND)7 (Rice et al., 1981), an age at which the development of the rat brain corresponds to that of the human infant at gestational weeks 32-34 (Zhu et al., 2009; Vannucci et al., 2009). The Vannucci HI model has since been adapted to mouse models of perinatal brain injury, and this has allowed the technique to be used with a broader array of genetically modified animals. Mice at PND9 have been the most commonly used, and these correspond to the human near-term infant (Zhu et al., 2009). In the present protocol, the Vannucci model has been adapted to serve as a model of preterm brain injury in C57bl/6J mice at PND5, an age where the development of the mouse brain corresponds to the brain of a human preterm infant (Zhu et al., 2009; Albertsson et al., 2014). The injury acquired with this protocol is characterized by local white matter injury combined with small areas of focal cortical injury and hippocampal atrophy (Albertsson et al., 2014).
Keywords: MouseMaterials and Reagents
- Animals
C57bl/6J mice (Charles River Laboratories) at PND4-5
The day of birth is defined as PND1
The body weight of PND 5 pups should be 2.0 ± 0.2 g - Anesthetics
Equipment
- Suture
6-0 silk suture attached to a needle for skin closure (Vömel, catalog number: 14719 ) - 7-0 silk suture cut into 1.0 cm–1.5 cm pieces for artery ligation (Ethicon, Johnson & Johnson, catalog number: EH7464G )
- Needle for skin suture
- Micropore surgical tape (Sollentuna, catalog number: 1530-0, 3 M )
- Syringe, 1 ml (B. Braun, Omnifix® 100 Solo)
- Heating plate (Labotect, model: Hot plate 062 )
- Forceps (Figure 1):
Fine-tipped curved forceps for dissecting the artery (Dumont, tweezer style 7, catalog number: 0302-7-CO )
Fine-tipped straight forceps for dissecting the artery (Nopa instruments, catalog number: AB 501/05 )
Hooked forceps for lifting the artery (Dimeda Instrumente GmbH, catalog number: 42.883.07 ) - Fine-tipped scissors (sharp) (Dimeda Instrumente GmbH, catalog number: 08.340.09 )
- Nasal mask for administering anesthetic gas (Simtec Engineering, catalog number: 10025 )
- Anesthetic vaporizer (Ohmeda Isotec Isoflurane Vaporizer)
- Surgical microscope (Carl Zeiss, model: S100/OPMI pico )
- Water bath with adjustable temperature (Grant, model: W14 )
- Hypoxic chamber with controlled temperature, humidified airflow, and variable oxygen level (Figure 2)
Note: In this protocol, a Dräger transport incubator 5400 and a Gant type W14 water bath (Figure 2A) were used to keep the ambient temperature at 36 °C. A smaller customized incubator, the hypoxia chamber, was kept inside the transport incubator (Figure 2B). Pups were placed inside the hypoxia chamber during the hypoxia procedure. The air and the 10% oxygen were introduced into the bottom of the customized incubator and vented from the top of the incubator. Before being introduced in the hypoxia chamber, the air and the 10% oxygen mixture were humidified and heated to 36 °C with the heated water bath. The customized hypoxia chamber was 19 cm in diameter and 19 cm in length (Figure 2C).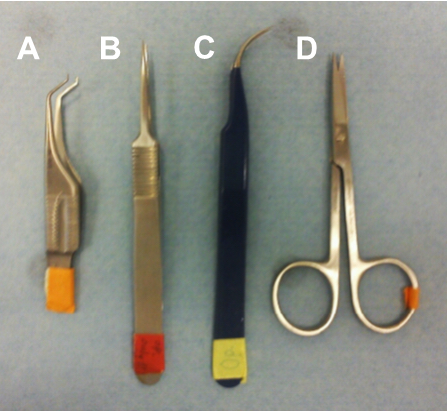
Figure 1. Surgical tools. Hooked fine-tipped forceps for lifting up the artery (A), fine-tipped straight forceps (B) and curved forceps (C) for dissecting the artery, and fine-tipped scissors for the skin incision (D).
Figure 2. Equipment used for hypoxia treatment. A. Water bath and transport incubator containing the hypoxia camber. B. The hypoxia chamber inside the transport incubator, and (C) the dimensions of the customized hypoxia chamber.
Figure 3. Hypoxia-Ischemia procedure time line
Procedure
Note: The duration of hypoxia, the chamber temperature, and the age and strain of the mice are all factors that have significant effects on the brain injury outcome. In this model, we use 70 min of hypoxia (Figure 3). However, this hypoxia time might need to be adjusted empirically for each laboratory’s unique conditions.
- Surgery
Preparation/Notes- Set up the surgery area.
- Work on a ventilated area (Figure 4).
- Weigh each pup before surgery.
- Make sure to keep the pups warm all the time using the heating plate before and after surgery. Minimize the time of surgery to avoid cooling of the pups-this is very important for the outcome of the procedure! Apart from this, no additional attention to animal care is needed. If using normal C57bl/6J pups, the surgery should be done under clean, but not necessarily sterile, conditions. However, this might need to be adjusted depending on the strain of mice used.
- The pups are placed in a small clean paper box that has a thin bottom to allow good contact between the box and the heating plate to be sure that the heat can be conducted to the pups lying at the bottom of the box.

Figure 4. Ventilated area for surgery (A). Heating plate for keeping pups warm (left, B), nasal mask for anesthesia (center, C) and anesthetic vaporizer (right, D).
Procedure- Anesthetize the pup with isoflurane using a nasal mask (5.0% for induction and 1.5% to 3.0% for maintenance).
- Gently fixate the pup on its back using the micropore surgical tape (Figure 5A). Keep the nasal mask with isoflurane in position.
- Open the skin with the scissors by making a 0.2 cm–0.3 cm incision in the neck close to the midline (Figure 5B).
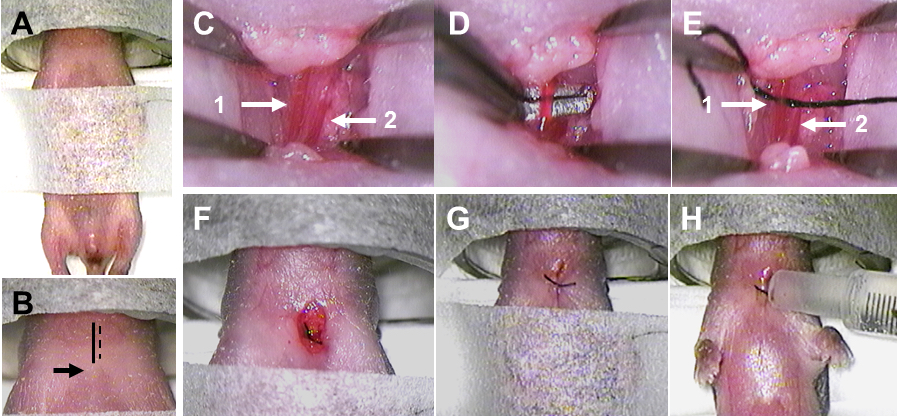
Figure 5. Surgery and artery ligation. A. The fixated pup with nasal mask for anesthesia. B. The midline is indicated by the solid black line, the incision is indicated by the dashed black line, and the top of the sternum is indicated by the black arrow. C. The artery is indicated by arrow 1 and the vein is indicated by arrow 2. D. The dissected artery is lifted up by hooked forceps. E. The suture under the artery is indicated by arrow 1, and the vein without the suture is indicated by arrow 2. F. The ligated artery. G. The sutured wound. H. The application of Xylocain on the wound. - Use fine-tip forceps to carefully push away fat and muscles. Close to the midline there will be a pocket underneath the fat and muscles, and this is where the left common carotid artery will be located.
- Very close to the common carotid artery will be a vein. Make sure not to confuse the artery and the vein (Figure 5C).
- The vein is more superficial than the artery, and the artery is more medial (to the left) than the vein.
- Compared to the vein, the artery is more reddish and will have an obvious pulse.
- Use the fine-tip forceps to carefully dissect the artery free from the vein, the vagal nerve, and other surrounding soft tissues.
- When the artery has been dissected, use a curved fine-tip forceps or hooked forceps to isolate and lift up the artery (Figure 5D).
- With the other hand, take up the prepared 1 cm-1.5 cm long 7-0 suture using the straight or curved forceps. Grab one end of the suture with the curved or hooked forceps holding up the artery and gently pull the suture under the artery (Figure 5E).
- Ligate the artery by making two knots with the suture (Figure 5F).
- Close the wound with the 6-0 suture. Make two knots (Figure 5G).
- Infiltrate the wound with local anesthetic by adding one drop of Xylocain onto the wound using the 1 ml syringe (Figure 5H).
- Put the pup back on the heating plate and allow the pup to recover from anesthesia. Pups have recovered when they have woken up and started to move again.
- When all pups have gone through surgery, take all pups back to their dam to rest for 1 h.
- Set up the surgery area.
- Hypoxia
Preparation/Notes- The temperature in the chamber should be kept strictly at 36 °C. It is important that this temperature does not change because the pups are not able to maintain their body temperature. A 1-2 °C drop in temperature (hypothermia) can result in reduced brain injury, and increased temperature can result in increased brain injury.
- To keep the chamber temperature strictly at 36 °C, the air and 10% oxygen must be warmed and humidified before flowing through the chamber.
- The airflow and the 10% oxygen flow through the chamber are both 4 L/min.
- Under normal situation in our lab, the hypoxia-ischemia treatment causes less than 10% mortality. The experimenter needs to pay attention to increases in mortality that might be indicating problems with the equipment or the procedure.
Procedure- Start the flow of warmed and humidified air through the chamber 5 min prior to putting the pups in the chamber.
- After the pups have recovered from surgery for 1 h with their dam, put the pups in the chamber and maintain the airflow through the chamber for 10 min.
- After 10 min of airflow, change the flow from air to 10% oxygen (by mixing air and nitrogen) for 70 min.
- Check the temperature regularly during the 10% oxygen incubation to make sure that the temperature is stable at 36 °C.
- After 70 min in 10% oxygen, change the flow from 10% oxygen to air for 10 min.
- After 10 min in air, take the pups from the chamber and return them to their dam.
- The temperature in the chamber should be kept strictly at 36 °C. It is important that this temperature does not change because the pups are not able to maintain their body temperature. A 1-2 °C drop in temperature (hypothermia) can result in reduced brain injury, and increased temperature can result in increased brain injury.
Representative data
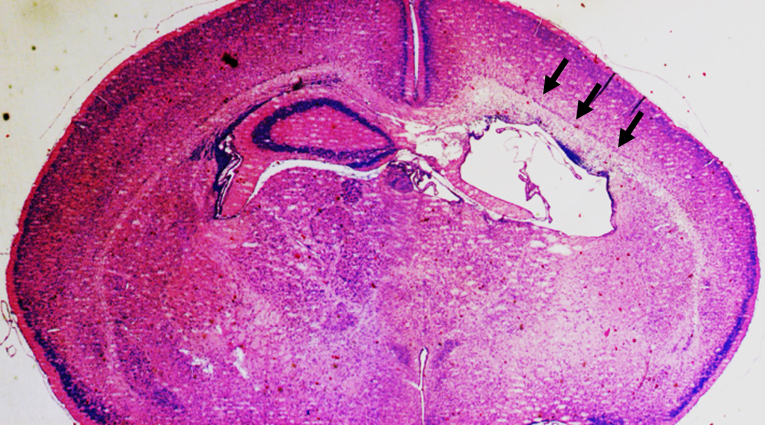
Figure 6. Representative data. Representative photomicrograph of a thionin/acid fuchsin-stained brain coronal section shows the degree of injury in the ipsilateral hemisphere (the right side) at 3 days after HI at PND5 with 70 min of hypoxia. Arrows indicate focal subcortical white-matter injury and the loss of hippocampus in the ipsilateral hemisphere of the mouse brain.
Notes
HI should be performed during the daytime when the dam is less active. This makes it more likely that the dam stays in the nest to keep the pups warm when they return after the surgery/HI. Otherwise, the pups might suffer from hypothermia that would influence the degree of brain injury degree and contribute to variation in the results.
Acknowledgments
This protocol was adapted from the previously published study of Albertsson et al. (2014). XW and AM research is supported by the Swedish Medical Research Council (VR 2008-2286 and VR 2013-2475 to XW), the Bill & Melinda Gates Foundation Grand Challenge Explorations and Global Health (OPP1036135 to XW), Swedish governmental grants to researchers in the public health service (ALFGBG-367051 and ALFGBG-367051 to XW), VINNMER-Marie Curie international qualification (VINNOVA, 2011-03458 to XW), and Wilhelm and Martina Lundgren (37/2013 to XW and vet2-26/2013 to AM).
References
- Albertsson, A. M., Bi, D., Duan, L., Zhang, X., Leavenworth, J. W., Qiao, L., Zhu, C., Cardell, S., Cantor, H., Hagberg, H., Mallard, C. and Wang, X. (2014). The immune response after hypoxia-ischemia in a mouse model of preterm brain injury. J Neuroinflammation 11(1): 153.
- Hedtjarn, M., Leverin, A. L., Eriksson, K., Blomgren, K., Mallard, C. and Hagberg, H. (2002). Interleukin-18 involvement in hypoxic-ischemic brain injury. J Neurosci 22(14): 5910-5919.
- Rice, J. E., 3rd, Vannucci, R. C. and Brierley, J. B. (1981). The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol 9(2): 131-141.
- Sheldon, R. A., Sedik, C. and Ferriero, D. M. (1998). Strain-related brain injury in neonatal mice subjected to hypoxia-ischemia. Brain Res 810(1-2): 114-122.
- Vannucci, R. C., Connor, J. R., Mauger, D. T., Palmer, C., Smith, M. B., Towfighi, J. and Vannucci, S. J. (1999). Rat model of perinatal hypoxic-ischemic brain damage. J Neurosci Res 55(2): 158-163.
- Vannucci, S. J. and Hagberg, H. (2004). Hypoxia-ischemia in the immature brain. J Exp Biol 207(Pt 18): 3149-3154.
- Zhu, C., Wang, X. and Blomgren, K. (2009). Cerebral Hypoxia-Ischemia in neonatal rats or mice: A model of perinatal brain injury, in animal models of acute neurological injuries. In: Chen, J. et al. (eds). Humana Press, 221-230.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Albertsson, A. and Wang, X. (2015). A Mouse Model of Preterm Brain Injury after Hypoxia-Ischemia. Bio-protocol 5(13): e1526. DOI: 10.21769/BioProtoc.1526.
Category
Neuroscience > Nervous system disorders > Animal model
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link