- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Mouse Models of Uncomplicated and Fatal Malaria
(*contributed equally to this work) Published: Vol 5, Iss 13, Jul 5, 2015 DOI: 10.21769/BioProtoc.1514 Views: 16206
Reviewed by: Migla MiskinyteAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Visualizing Hypoxia in a Murine Model of Candida albicans Infection Using in vivo Biofluorencence
José Pedro Lopes and Constantin F. Urban
Aug 5, 2019 5112 Views
RNA Extraction from Ears and Draining Lymph Nodes of Mice Infected with Leishmania amazonensis
Emilie Giraud and Evie Melanitou
Jun 5, 2020 5516 Views

TetR Regulated in vivo Repression Technology to Identify Conditional Gene Silencing in Genetically Engineerable Bacteria Using Vibrio cholerae Murine Infections as Model System
Franz G. Zingl [...] Stefan Schild
Oct 5, 2020 3749 Views
Abstract
Mouse models have demonstrated utility in delineating the mechanisms underlying many aspects of malaria immunology and physiology. The most common mouse models of malaria employ the rodent-specific parasite species Plasmodium berghei, P. yoelii, and P. chabaudi, which elicit distinct pathologies and immune responses and are used to model different manifestations of human disease. In vitro culture methods are not well developed for rodent Plasmodium parasites, which thus require in vivo maintenance. Moreover, physiologically relevant immunological processes are best studied in vivo. Here, we detail the processes of infecting mice with Plasmodium, maintaining the parasite in vivo, and monitoring parasite levels and health parameters throughout infection.
Keywords: MouseMaterials and Reagents
- Mice. We perform most of our work in C57BL/6 mice due to the wide availability of transgenic and knockout strains on this genetic background. Slight differences in vendor-specific sub-strains exist, so it is preferable to obtain the mice from the same source throughout a given study. The majority of our studies use 9-12 week old females, since males have higher parasite loads and mortality with P. chabaudi infection in many mouse strains (Laroque et al., 2012).
- Plasmodium parasites. Plasmodium parasites can be obtained from the Malaria Research and Reference Reagent Resource (MR4). The most commonly used rodent malaria strains include P. chabaudi AS (MRA-429; used to model uncomplicated malaria), P. berghei ANKA (MRA-311; fatal cerebral malaria), P. yoelii 17XNL (MRA-593; uncomplicated malaria; resolves late relative to P. chabaudi), and P. yoelii YM (MRA-755; lethal malaria without cerebral pathology).
- Immersion oil Type A (Cargille, catalog number: 16482 )
- Methanol (Thermo Fisher Scientific, catalog number: A412P4 )
- Giemsa stain (Acros Organics, catalog number: 295591000 )
- 1x PBS (Mediatech Inc., catalog number: 21-040 CV )
- Alsever’s solution (MP Biomedicals, catalog number: 092801154 ) (see Recipes) (Note 1)
Equipment
- 1 ml syringes (BD, catalog number: 309628 )
- 25 g ⅝” needles (BD, catalog number: 305122 )
- 25 mm x 75 mm x 1 mm unfrosted glass slides (Premiere, catalog number: 9101-E )
- 25 mm x 75 mm x 1 mm frosted glass slides (Globe Scientific Inc., catalog number: 1324W )
- Slide staining rack and chamber (Electron Microscopy Sciences, catalog number: 62543-06 and 62541-01 )
- Glass cutter (optional) (General, catalog number: 06637383 )
- 1.5 ml microfuge tubes (Thermo Fisher Scientific, catalog number: 50809238 )
- Sharp surgical scissors
- Brightfield microscope equipped with 100x objective
- Lens paper (Thermo Fisher Scientific, catalog number: 11-995 )
- Lamellar flow hood (optional; if working in a pathogen-free animal facility)
- Scale with cup for weighing mice (OHAUS, catalog number: SB36853M )
Software
- VersaCount (Kim and DeRisi, 2010)
Procedure
- Starting an infection from cryopreserved parasites
- Order cryopreserved parasite stocks, provided as infected mouse erythrocytes, from the Malaria Research and Reference Reagent Resource (MR4). MR4 is now administered by the NIAID’s BEI Resources program, and you will need to submit paperwork to obtain security clearance before being able to order parasites. Rodent-restricted Plasmodium parasites are classified as BEI level 1; registration instructions are available at http://www.beiresources.org/RegisterLevel1.aspx.
- Cryopreserved parasite stocks will arrive on dry ice. They can be stored at -80 °C in the short term, but will quickly lose viability under these conditions. If they are not used within two days of receipt, store in liquid nitrogen to maximize parasite viability. Cryopreserved parasite stocks can be stored in liquid nitrogen for several years with minimal loss of viability.
- On infection day, retrieve the cryovial from storage and place it in a small cooler of dry ice until just before injection.
Important safety note: Use personal protective equipment to prevent injury from frostbite or cryovial explosion. - Identify one or two mice to be infected. Typically, we use wild type 9-12 week old C57BL/6 female mice.
- Prepare a 1 ml syringe with a 25 g ⅝” needle.
- Thaw the vial containing cryopreserved infected erythrocytes rapidly in your gloved hand. Reposition the vial frequently to minimize the risk of frostbite.
- Load the syringe with the thawed parasites. The parasites are sensitive to lysis and should be loaded slowly, at a rate of approximately 25 µl per second, to minimize shear stress. Do not pass the infected erythrocytes through the needle more than necessary.
- Inject 200-250 µl of the cryopreserved parasites per mouse intraperitoneally. We recommend measuring the parasitemia every day starting on day 3 post-infection (with the day following injection being day 1). If increased susceptibility to infection is expected as a possible outcome, begin monitoring earlier. Procedures for measuring parasitemia are described below (Note 2).
- Order cryopreserved parasite stocks, provided as infected mouse erythrocytes, from the Malaria Research and Reference Reagent Resource (MR4). MR4 is now administered by the NIAID’s BEI Resources program, and you will need to submit paperwork to obtain security clearance before being able to order parasites. Rodent-restricted Plasmodium parasites are classified as BEI level 1; registration instructions are available at http://www.beiresources.org/RegisterLevel1.aspx.
- Preparing thin-film blood smears
One of the most classic assays of malaria research is the thin-film blood smear. In this assay, parasitized red blood cells can be visibly identified by the presence of stained nucleic acid, which is present at comparatively low levels in the anucleate mouse red blood cells. This allows simple assessment of parasitemia-the proportion of red blood cells that are infected-using basic light microscopy. Although parasitemia reflects the combined influence of both parasite load and red blood cell density, its simplicity, sensitivity, and precision make it the most useful measure of parasite load for routine work. We recommend collecting blood smears before 12:00 PM due to the circadian rhythm of some species of Plasmodium parasites and their tendency to cytoadhere to the vasculature in later life cycle stages (Note 3).
As per our institutional requirements, we record the weight of the mice throughout the study using a scale with 0.1 g resolution. We also score and document the body condition of the mice, as well as other health parameters (e.g., anemia, activity level, hematuria, neurological complications). These measurements may be useful for assessing the progress of the infection and may be required by your institutional guidelines.- Using a pair of sharp surgical scissors, cut off a 0.5 to 1 mm section from the tip of the mouse’s tail. The snip should only remove enough of the tail to obtain a drop of blood.
- In one fluid motion towards the distal end of the tail, gently milk the tail to collect a drop of blood at the tip. In some cases, the drop of liquid may lack obvious red blood cells. This can occur as a result of severe anemia, but can also occur if the tail snip is too small. If other infection parameters (e.g. day post-infection, animal activity) are inconsistent with severe anemia, re-snip the tail.
- Transfer a 1 to 2 µl drop of blood, directly from the tip of the tail, close to the edge of a microscope slide (Figure 1A).
- As the blood will begin to coagulate within several seconds, quickly touch the edge of a second slide to the blood sample at a 45° angle, allowing the blood to spread across the edge (Figures 1B and 1C). Materials can be saved by scoring and snapping unfrosted slides into quarters using a glass scoring tool, and using these to spread the blood (Figure 1D).
Important safety note: Use caution when snapping glass, as it can result in sharp edges if performed improperly. Dispose of all glass waste according to your institutional guidelines. - Apply gentle pressure and evenly spread the blood across the width of the sample slide to obtain a thin-film blood smear (Note 4) (Figure 1E). Allow the blood to dry for a few seconds.
- Place the slides into a rack and submerge the slides in methanol for approximately 5 seconds to fix the smears.
- Remove the slides from methanol and allow them to air dry, preferably in front of a fan. While the slides are drying, pipette 1 ml of Giemsa into 250 ml of 1x PBS in a slide staining chamber (Note 5).

Figure 1. Preparation of thin-film blood smears. A. Transfer a drop of blood onto a microscope slide. B. Side-view: Place the edge of a second slide at a 45° angle, allowing the blood to spread across the edge of the second slide. C. Top-view: Place the edge of a second slide at a 45° angle, allowing the blood to spread across the edge. D. A glass scoring tool and snapped slide fragments. Properly snapped slides should not have sharp edges. E. Place three smears on a single microscope slide for convenience when staining and counting large numbers of smears. - Incubate the slides in the chamber for 10 min to 1 h, depending on the strength of your staining solution. The slide is sufficiently stained when leukocytes present in the smear have dark blue-purple nuclei.
- Thoroughly rinse the slides with water and place them in front of a fan to dry. If counting will be performed immediately, the rinsed slides can also be gently blotted dry and analyzed using the procedures described below. The stained slides are stable; if counting will not be performed immediately, the dried slides can be stored in a slide box at room temperature indefinitely.
- Using a pair of sharp surgical scissors, cut off a 0.5 to 1 mm section from the tip of the mouse’s tail. The snip should only remove enough of the tail to obtain a drop of blood.
- Counting thin-film blood smears
For routine monitoring, parasite burden is most conveniently calculated as parasitemia. In order to obtain this number, infected erythrocytes must be distinguished from uninfected erythrocytes, leukocytes, and platelets, and care must be taken to differentiate between reticulocytes (immature red blood cells that still harbor some nucleic acid) and parasitized normocytes (fully mature erythrocytes). Debris and platelets can also pose a challenge for inexperienced researchers, as these particles, when associated with erythrocytes, can have the appearance of parasites. For these reasons, it is important to become very familiar with parasite morphology.
Notably, different species of malaria parasites have different predilections for different types of red blood cells. P. chabaudi AS parasites display a preference for normocytes, whereas P. berghei ANKA parasites display a preference towards reticulocytes (Sinden et al., 2002). This is most apparent during early infection when parasite numbers are low. At higher loads, parasites tend to be found in all erythrocyte stages. Parasites also differ in other properties with which the researcher should become familiar. For example, P. chabaudi AS parasites cytoadhere, and as a result, ring-stage trophozoites are the main lifecycle stage observed on smears. P. berghei ANKA parasites do not cytoadhere; therefore, all life cycle stages are observed on smears. Our standard practice is to count all stages toward an aggregate parasitemia value; in some cases, it may be preferable to count lifecycle stages separately. See Figure 2 for images of the primary blood stages of Plasmodium parasites.- Place a small drop of immersion oil directly onto the smear and find the focal plane on a brightfield microscope with a 100x objective. The smear can also be coverslipped in a mounting medium prior to microscopic analysis, if long-term preservation of the smear is desired. For routine smear analysis, a coverslip is not required.
- Count approximately 500 red blood cells per smear with a tally counter, separately keeping track of infected and uninfected cells. If the parasite life cycle stage is important for your experiment, count the various stages separately (Note 6).
- Calculate parasitemia, the percentage of erythrocytes infected with parasites.
Note: For parasitemia below 0.1% or analyses where high precision is required, more cells should be counted for more precise results. In such cases, estimate the parasitemia by examining up to 10,000 total cells. To do so efficiently, the total number of cells in a given field can be estimated with experience, and successive fields can be summed to reach approximately 10,000 cells, while simultaneously tallying infected cells. At high parasitemia, adequate precision can be obtained from fewer than 500 cells.
Figure 2. The major Plasmodium life cycle stages observed in blood. Example images of ring trophozoites (commonly referred to as “rings”), late stage trophozoites, and very late stage schizonts are shown. Note that the schizonts in these images have segmented into individual merozoites, which immediately precedes parasite egress. All images are P. berghei ANKA; P. chabaudi AS rings have a similar appearance to P. berghei ANKA rings.
- Place a small drop of immersion oil directly onto the smear and find the focal plane on a brightfield microscope with a 100x objective. The smear can also be coverslipped in a mounting medium prior to microscopic analysis, if long-term preservation of the smear is desired. For routine smear analysis, a coverslip is not required.
- Passaging and initiating experimental infections
Because it is not practical to maintain mouse malaria parasites in vitro and cryopreserved parasites require one to two passages to return to optimal growth, maintaining an infected passage mouse at all times provides a convenient source of parasites for initiating experiments. In our experience, infections are the most reproducible when parasites are passaged during the ascending phase of parasitemia. The parasitemia levels should be high enough to count accurately and to provide enough inoculum to initiate experiments, but not so high that the parasites have neared or reached the peak of their growth. For non-lethal parasites such as P. chabaudi AS, a range of 5-20% parasitemia gives satisfactory results. For P. chabaudi infection of C57BL/6 mice, this level of parasitemia is typically reached between days 5 and 7 (Note 7). For P. yoelii 17XNL, this level of parasitemia is reached later, typically around day 10 to day 13. For lethal strains such as P. berghei ANKA, passage should be performed at lower parasitemias to avoid loss of the passage parasites due to death of the passage mouse (see below); we typically passage P. berghei ANKA no later than 5 days post infection, and around 5% parasitemia. See Figure 3 for representative parasitemia courses.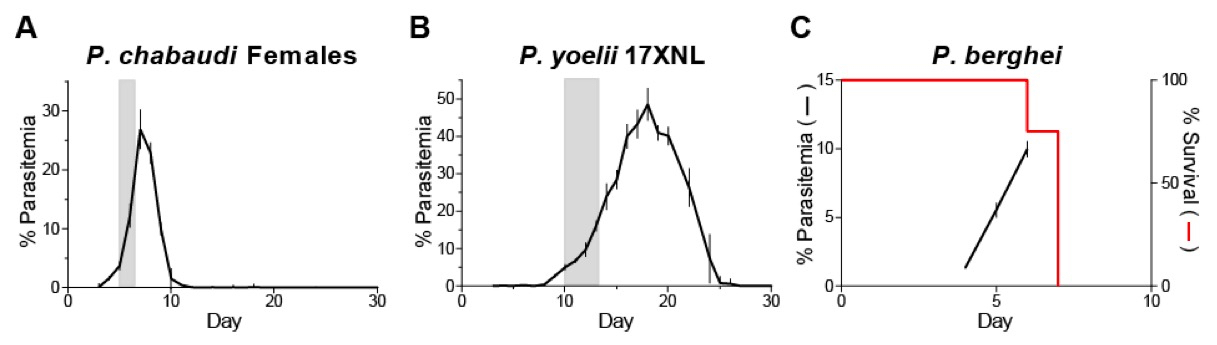
Figure 3. Parasitemia and survival courses for commonly used Plasmodium strains in mice. A. The course of P. chabaudi AS parasitemia in female C57BL/6 mice. Data were pooled from two independent experiments (n=11). B. The course of P. yoelii 17XNL parasitemia in C57BL/6 mice. Data were from one experiment (n=5). C. The course of P. berghei ANKA parasitemia (n=14; pooled from two independent experiments) and survival (n=28; pooled from three independent experiments) in C57BL/6 mice. Shaded boxes represent our preferred window for passaging the infections. Parasitemias are presented as geometric means with SEM.
The infection should be passaged as soon after collecting and counting the thin-film blood smear of the passage mouse as possible to minimize the impact of replication and cytoadherence during the interim (Notes 8-9). In addition, an important consideration is that mosquito infectivity can be lost upon serial parasite passage; although we have not tested this ourselves, previous reports have suggested limiting the number of passages to eight (Sinden et al., 2002). Serial passage in mice also results in enhanced virulence compared to mosquito-transmitted parasites (Spence et al., 2013). Thus, new parasite stocks should initially be expanded by infecting several mice, and aliquots of parasites should be cryopreserved. We prefer cryopreservation in Glycerolyte solutions using published protocols (Moll et al., 2013).- Monitor the passage infection daily until the desired parasitemia is reached.
- Many parasites are sensitive to thermal fluctuations. Prewarm the sterile Alsever’s solution to 37 °C to minimize shock to the parasites.
- Calculate the volume of blood required per 1 ml of Alsever’s solution. Infections are typically initiated by injecting an inoculum of 104 to 106 infected red blood cells in 100 µl of Alsever’s solution per mouse (Sanni et al., 2002). Calculate the volume of blood needed for 107 infected red blood cells per 1 ml of Alsever’s solution using the following equation, where parasitemia is expressed as a fraction:
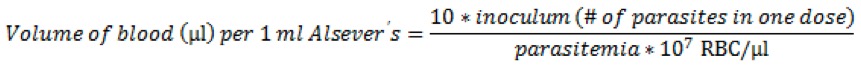
Or, more simply, if targeting 106 cells per 100 µl dose: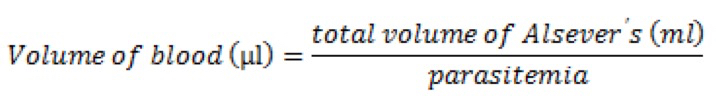
For example, a dose of 106 parasites delivered in 100 µl from a passage infection at 10% parasitemia requires 10 µl of whole blood in 1 ml Alsever’s solution. - Euthanize the passage mouse according to your institutional guidelines and harvest blood via cardiac puncture using a 25 g ⅝” needle and a 1 ml syringe (Note 10). If permitted, avoid cervical dislocation before performing cardiac puncture, as this will cause internal bleeding that reduces blood recovery.
- Insert the needle approximately 0.5 cm deep into the thoracic cavity (Figure 4A). Pull the plunger to 50 µl to create slight negative pressure.
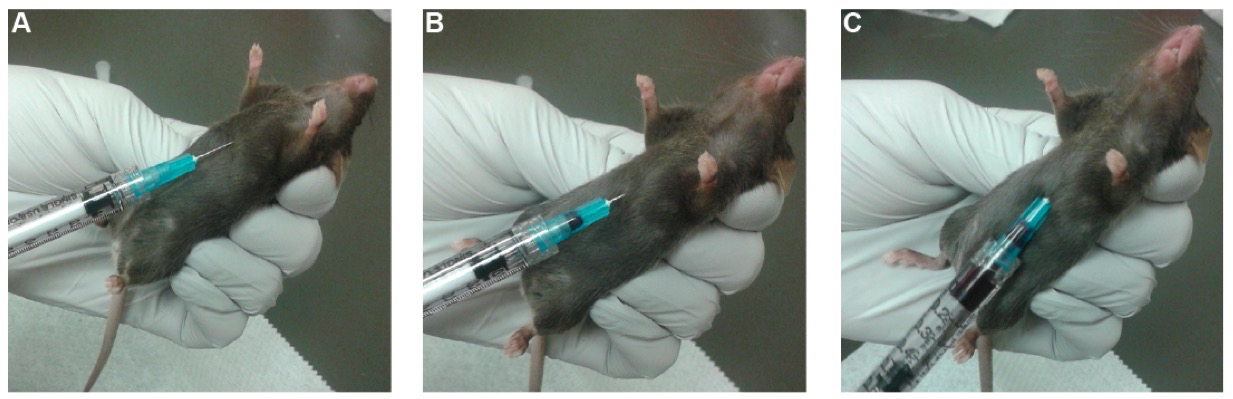
Figure 4. Cardiac Puncture. A. Insert the needle approximately 0.5 cm into the thoracic cavity. At this point, pull approximately 50 µl of volume to create a very light vacuum. B. Slowly advance the needle until puncturing the heart. Note the visible flash of blood entering the needle base. C. Slowly pull on the plunger, filling the syringe with blood. - Slowly advance the needle, up to an additional 0.5 to 1 cm, until the needle punctures the heart. This can be observed by a visible flash of blood entering the needle (Figure 4B).
- Once the heart has been punctured, slowly pull on the plunger until blood ceases to fill the syringe barrel (Figure 4C). A good rate of draw is approximately 50 µl per second, which avoids stoppage of the blood flow due to a collapsed heart. Once no more blood is readily entering the syringe, more blood can often be obtained by gently twisting and/or moving the needle while pulling gentle vacuum. This should be done quickly, as the blood clots quickly – typically within a minute or two. An effective cardiac puncture on a 9-12 week old female mouse typically yields 300 to 700 µl of whole blood.
- Expel the blood from the syringe into a clean 1.5 ml microfuge tube. We typically perform this quickly in the absence of anticoagulant, but tubes containing anticoagulant can be used to reduce the urgency of preparing the inoculum.
- Quickly dilute the calculated amount of blood into pre-aliquotted and pre-warmed sterile Alsever’s solution using a pipetteman.
- Insert the needle approximately 0.5 cm deep into the thoracic cavity (Figure 4A). Pull the plunger to 50 µl to create slight negative pressure.
- Administer 100 µl of the inoculum to each mouse by intraperitoneal injection using a 25 g ⅝” needle and 1 ml syringe (Note 11). Perform this step rapidly, as parasite viability decreases quickly. We inoculate all mice within 15 min of the original blood draw to maximize reproducibility. Large experiments require multiple experienced handlers performing inoculations. Gently agitate the inoculum regularly, as the erythrocytes settle rapidly.
- Monitor the parasitemia as described above.
- Monitor the passage infection daily until the desired parasitemia is reached.
- P. berghei ANKA experimental cerebral malaria
Infection of C57BL/6 mice with P. berghei ANKA is the most widely used mouse model of cerebral malaria, one of the most severe complications of Plasmodium infection in humans. In this cerebral malaria model, infected mice exhibit neurological symptoms such as ataxia, convulsions, and/or paralysis, and typically die between days 6 to 8 post-infection (Baccarella et al., 2014; Coban et al., 2007). P. berghei has also been used as a model for malaria-associated organ pathology in the liver, lung, and spleen, in addition to the extensively studied cerebral pathology (reviewed by Oca et al., 2013).
We also note that lethal P. yoelii YM infection follows a similar course as that described here and can largely be performed using the same procedures, although the pathology of this infection is different from that observed in P. berghei ANKA infection of C57BL/6 mice.- P. berghei ANKA replicates quickly, and must be passaged earlier than P. chabaudi AS due to the risk of passage mouse mortality. Begin monitoring the parasitemia of the passage mouse on day 3.
- Perform passage infections and/or initiate new experiments during ascending parasitemia levels of approximately 5%, which are typically reached between days 4 and 6. Follow the protocol under section D.
- Monitor and record the number of surviving mice daily. Record health parameters as outlined by your institutional guidelines, and record the identifier number of dead mice if needed for association studies (Note 12).
- Take thin-film blood smears daily to track parasitemia, if desired. It is recommended to begin monitoring parasitemia no later than day 3.
- P. berghei ANKA replicates quickly, and must be passaged earlier than P. chabaudi AS due to the risk of passage mouse mortality. Begin monitoring the parasitemia of the passage mouse on day 3.
Notes
- KSGH is another anticoagulant buffer that can be used for dilution of parasites (Spence et al., 2011). We have not ascertained any significant impact on our infections between using Alsever’s solution and KSGH, but we prefer Alsever’s solution as it is simpler to prepare and is readily commercially available.
- Cryopreserved parasites have poor viability, and even injection of a seemingly large cryopreserved inoculum can exhibit slow growth. It is not unusual for infections started from cryopreserved stocks to peak as late as day 12, and at relatively low parasitemias; for this reason, if parasites fail to appear in the blood during the expected window, smears should still be monitored for parasites even on days that are typically considered post-acute for P. chabaudi AS or post-lethal for P. berghei ANKA.
- Parasitemia will progressively decline as the day progresses due to more parasites in later life cycle stages cytoadhering to the vasculature. For the most accurate results when comparing parasitemia for multiple days, it is best to collect smears at roughly the same time every morning.
- Although traditional protocols push the spreader away from the drop of blood, we pull the spreader across the drop of blood to create a thin-film blood smear. We find this approach to be easier for most individuals to master for preparation of uniform smears.
- Other buffers, such as Tris-EDTA, can be used for Giemsa staining. However, the pH impacts the quality of the staining. Higher pH, in particular, will prevent the red color from appearing, although this is not vital for parasite identification. Giemsa preparations also vary across vendors, so it is recommended that the Giemsa concentration and duration of staining be titrated upon receipt of a new bottle.
- A convenient replacement for manual tally counters is VersaCount, a free customizable program (Kim and DeRisi, 2010).
- Because the course of P. chabaudi approaches the peak approximately one day earlier in males as compared with females, it is possible to “shift” the course of passage forward by a day for convenience in scheduling. We have not observed any significant impact of switching the parasites between male and female mice.
- Because of cytoadherence of late stage parasites, if a significant amount of time elapses between gathering the thin blood smear and passaging the infection, the parasitemia used to calculate the inoculum concentration will not accurately reflect the parasitemia of the mouse at the time of passage. This will lead to an overestimation of the number of parasites administered to each mouse, which will increase inter-experiment variability.
- Passage of parasites during the descending, post-peak phase of acute parasitemia is possible, but many of these parasites are dying due to the activation of anti-malarial mechanisms, making it difficult to know the precise number of live parasites being delivered. For this reason, we recommend initiating experiments only from parasites harvested during the ascending parasitemia.
- Although submandibular blood collection can be used to harvest blood from a passage mouse, cardiac puncture is the preferred technique for obtaining a large, sterile blood sample. Cardiac punctures in 8 week old mice typically yield 300-700 µl of blood, whereas our institutional guidelines only permit submandibular collection up to 50 µl of blood. Submandibular blood collection must also be performed under isoflurane anesthesia, which requires specialized equipment for controlled delivery. The disadvantage to cardiac puncture is that it requires more skill; if it is not performed quickly, the blood can coagulate before sufficient quantities are obtained to prepare the inoculum.
- The referenced article published in Journal of Visualized Experiments provides an instructive video demonstrating how to perform an intraperitoneal injection (Machholz et al., 2012).
- We find that marking mouse tails with colored permanent markers is an effective, convenient, and minimally traumatic method for tracking mouse identities. This approach is well suited for short-term experiments, since tails will require routine re-marking every 4-5 days. For long-term experiments, tattooing or other forms of identification are preferred.
Recipes
- Alsever’s solution
Sodium chloride 4.2 g/L Sodium citrate dihydrate 8 g/L Citric acid monohydrate 0.55 g/L D-glucose 20.5 g/L
Acknowledgments
This work was supported by NIH K99 AI085035, NIH R00 AI085035, NCATS UCSF-CTSI UL1 TR000004, and the UCSF Sandler Neglected Tropical Diseases Program. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. We thank Alyssa Baccarella, Joshua Craft, Mary Fontana, Aqieda Bayat, Kim D’Costa, and Nicole Lee for technical assistance.
References
- Baccarella, A., Huang, B. W., Fontana, M. F. and Kim, C. C. (2014). Loss of Toll-like receptor 7 alters cytokine production and protects against experimental cerebral malaria. Malar J 13: 354.
- Coban, C., Ishii, K. J., Uematsu, S., Arisue, N., Sato, S., Yamamoto, M., Kawai, T., Takeuchi, O., Hisaeda, H., Horii, T. and Akira, S. (2007). Pathological role of Toll-like receptor signaling in cerebral malaria. Int Immunol 19(1): 67-79.
- Kim, C. C. and Derisi, J. L. (2010). VersaCount: customizable manual tally software for cell counting. Source Code Biol Med 5(1): 1.
- Laroque, A., Min-Oo, G., Tam, M., Radovanovic, I., Stevenson, M. M. and Gros, P. (2012). Genetic control of susceptibility to infection with Plasmodium chabaudi chabaudi AS in inbred mouse strains. Genes Immun 13(2): 155-163.
- Machholz, E., Mulder, G., Ruiz, C., Corning, B. F. and Pritchett-Corning, K. R. (2012). Manual restraint and common compound administration routes in mice and rats. J Vis Exp(67): e2771.
- Moll, K., Kaneko, A., Scherf, A. and Wahlgren, M. (2013). Methods in malaria research (6th edition). In: Glasgow, E. (ed). UK MR4/ATCC. Manassas.
- Oca, M. M. de, Engwerda, C. and Haque, A. (2013). Plasmodium berghei ANKA (PbA) infection of C57BL/6J mice: A model of severe malaria. In: Allen, I. C. (ed). Mouse Models of Innate Immunity. Humana Press, pp 203-213.
- Sanni, L. A., Fonseca, L. F. and Langhorne, J. (2002). Mouse models for erythrocytic-stage malaria. Methods Mol Med 72: 57-76.
- Sinden, R. E., Butcher, G. A. and Beetsma, A. L. (2002). Maintenance of the Plasmodium berghei life cycle. Methods Mol Med 72: 25-40.
- Spence, P. J., Cunningham, D., Jarra, W., Lawton, J., Langhorne, J. and Thompson, J. (2011). Transformation of the rodent malaria parasite Plasmodium chabaudi. Nat Protoc 6(4): 553-561.
- Spence, P. J., Jarra, W., Levy, P., Reid, A. J., Chappell, L., Brugat, T., Sanders, M., Berriman, M. and Langhorne, J. (2013). Vector transmission regulates immune control of Plasmodium virulence. Nature 498(7453): 228-231.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Huang, B. W., Pearman, E. and Kim, C. C. (2015). Mouse Models of Uncomplicated and Fatal Malaria. Bio-protocol 5(13): e1514. DOI: 10.21769/BioProtoc.1514.
Category
Microbiology > Microbe-host interactions > In vivo model > Mammal
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









