- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Trichoderma harzianum Root Colonization in Arabidopsis
Published: Vol 5, Iss 13, Jul 5, 2015 DOI: 10.21769/BioProtoc.1512 Views: 10323
Reviewed by: Arsalan DaudiKanika GeraMalou Fraiture

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

An Assay to Study Botrytis cinerea-infected Grapevine Leaves Primed with Pseudomonas fluorescens
Charlotte Gruau [...] Aziz Aziz
Oct 5, 2016 12843 Views

Assessment of Wheat Resistance to Fusarium graminearum by Automated Image Analysis of Detached Leaves Assay
Alexandre Perochon and Fiona M. Doohan
Dec 20, 2016 12751 Views

Quantification of the Composition Dynamics of a Maize Root-associated Simplified Bacterial Community and Evaluation of Its Biological Control Effect
Ben Niu and Roberto Kolter
Jun 20, 2018 10502 Views
Abstract
Trichoderma is a soil-borne fungal genus that includes species with a significant impact on agriculture and industrial processes. In this article we show a detailed protocol of Trichoderma harzianum (T. harzianum) root invasion procedure described by Alonso-Ramírez et al. (2014). Some Trichoderma strains exert beneficial effects in plants through root colonization. They promote growth and development, modify root architecture, facilitate efficient nutrient use, or stimulate defenses against pathogens, although little is known about how this interaction takes place. For this purpose, Trichoderma-Arabidopsis hydroponic cultures were grown inside Phytatray II boxes, using mycelia obtained from spores of T. harzianum and Arabidopsis thaliana (A. thaliana) seedlings. In this way changes in root architecture, such as callose deposition, promoted by the fungus can be analyzed.
Materials and Reagents
- Arabidopsis thaliana Col-0 ecotype seeds from Arabidopsis Information Service Collection (www.arabidopsis.info)
- Trichoderma harzianum CECT 2413 (Spanish Type Culture Collection, Valencia, Spain) [referred to as T34 along the paper is the strain used in this work. T. harzianum T34 is grown on Potato Dextrose Agar (PDA) and spores are maintained at -80 °C in a 30% glycerol solution]
- Murashige & Skoog medium (MS), including B5 vitamins (Duchefa Biochemia, catalog number: M0255.0050 )
- Sucrose (Applichem Panreac, catalog number: 141621.1211 )
- 85% potassium hydroxide pellets (KOH) (Applichem Panreac, catalog number: 141515.1210 )
- 0.15% agarose (Conda Pronadisa, catalog number: 8016 )
- 0.39% potato dextrose agar (PDA) (Sigma-Aldrich, catalog number: P2182 )
- 0.24% potato dextrose broth (PDB) (Sigma-Aldrich, catalog number: P6685 )
- Potato dextrose agar (PDA) (Conda, catalog number: 1022.00 )
- Potato dextrose broth (PDB) (Difco, catalog number: 254920 )
- Glass wool washed QP (Panreac, catalog number: 211376.1208 )
- Resma filter paper (420 x 500 mm) (Auxilab S.L, catalog number: 80250452 )
- Liquid nitrogen (Air Liquide)
- Ethanol (Panreac, catalog number: 161086 )
- Triton X-100 (Sigma-Aldrich, catalog number: T8787 )
- Sodium hypochlorite
- Sterilization solution (see Recipes)
- MS Medium (see Recipes)
- Washing solution (see Recipes)
Equipment
- Sterile distilled water purification system (EMD Millipore, model: ELIX35 )
- Brand cotton roving (Sigma-Aldrich, catalog number: BR28205 )
- Sterile stainless steel screen (Alunet, catalog number: 174562 )
- Surgical Micropore tape (3M, catalog number: 1530-0 )
- 1.5 ml microtubes (Deltalab, catalog number: 200400P )
- Cold chamber
- Phytatray II boxes [114 mm, 86 mm, 102 mm (W x D x H)] (Sigma-Aldrich, catalog number: P5929 )
- Laminar flow cabinet (Telstar, model: AV-100 )
- Plant Growth Chamber AGP-1400-HR (Radiber SA)
- Shaker Certomat® R (B. Braun, model: 986302/4 )
- Petri dishes (90 x 14 mm) (Deltalab, catalog number: 200209 )
- Surgeon carbon steel (surgical blade sterile) (Jai Surgicals, catalog number: 0835147 )
- 1.5 ml microtubes with glass wool (homemade)
- 15 ml Tubes (Deltalab, catalog number: 401402 )
- Thoma cell counting chamber (BRAND, catalog number: 7180 05 )
- Coverslip EUROTUBO (22 x 22 mm) (Deltalab, catalog number: D102222 )
- Optical microscope (Leica Microsystems AG, model: DC300F; catalog number: 10447115 )
- Erlenmeyer flask (Thermo Fischer Scientific, catalog number: 11972233 )
- Kühner shaker (Thermo Fisher Scientific, model: ISF-1-W )
- Vacuum/pressure pump PALL (Life Sciences, model: DOA-P730-BN )
- Safety glass (vacuum flask, 10 ml pipette and gums) (homemade)
- X5 Graduated pipette (type1, class B, ISO 835) (Thermo Fisher Scientific, catalog number: 11912178 )
- Medical grade silicone tubing (1x 1.5, internal diameter x external diameter) (Deltalab, catalog number: 3500115 )
- Rubber plugs (VWR International, catalog number: 217-9463 )
- Forceps (stainless, L= 105 mm) (Thermo Fisher Scientific, catalog number: 10458242 )
- Vacuum flask pirex (1,000 ml) (Thermo Fisher Scientific, catalog number: 12693182 )
- Magnetic filter funnel (VWR International, catalog number: 516-7590 )
- Scissors stainless 170 mm (Thermo Fisher Scientific, catalog number: 12693182)
- Lyophilizer Virtis Advantage (SP Scientific)
Procedure
- Arabidopsis thaliana seeds have to be washed and sterilized superficially before sowing. For this purpose, approximately 200 seeds are placed in a 1.5 ml microtube, and 1 ml of washing solution is added. Shake seeds for 30 min at room temperature in a shaker (Kühner shaker). Remove washing solution. Add 1 ml of sterilization solution. Shake again for 10 min at room temperature. Remove sterilization solution and wash seeds by tube inverting four times with sterile distilled water. Finally, seeds are placed in 1.5 ml microtubes and kept in stratification at 4 °C in the cold chamber for 3 days in order to break seed dormancy and synchronize the germination.
- Place Arabidopsis seeds inside Phytatray II boxes with 50 ml of liquid MS media on a sterile gauze sheet over a sterile stainless steel screen (Figures 1 and 2) in a laminar flow cabinet, using an aqueous agarose solution (0.15%) to sow seeds individually.

Figure 1. Set up of a sterile gauze sheet over a sterile stainless steel screen in Phytatray II boxes
Figure 2. Pictures showing the hydroponic cultures in the Phytatray II boxes - For the propagation of the T. harzianum strain, incubate a fungal plug of 5 mm of diameter, grown and sporulated on a petri dish with PDA, at least 7 days at room temperature, to achieve full coverage of the plate surface with spores.
- To harvest the spores, add 5 ml of sterile distilled water to the plate. Filter this suspension in 1.5 ml microtubes with glass wool, placed on 15 ml tubes, in order to remove traces of mycelium (Figure 3 and 4). Spores are maintained at 4 °C no more than 1 week. Finally, determine spore concentration in a Thoma cell counting chamber by pipetting 100 μl of a 1:100 dilution, using the following formula:
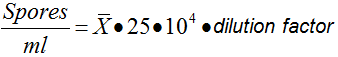
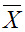 : It is the mean of the spores counted in four different quadrants.
: It is the mean of the spores counted in four different quadrants.
Finally, keep spores at 4 °C until use.
Figure 3. Set up for the glass wool in a microtube placed on a 15 ml tube
Figure 4. Harvest of T. harzianum spores - Use spores of T. harzianum (107 spores) to inoculate 250 ml flasks containing 100 ml of PDB. Maintain cultures at 25 °C and 200 rpm for 48 h. Harvest mycelia (approximately 250 mg) by filtration (filtration system: vacuum/pressure pump PALL, safety glass, rubber plugs, vacuum flask pirex and magnetic filter funnel). Wash the mycelia through the magnetic funnel with 100 ml of sterile water (Figure 5). Use the washed mycelia to inoculate the Phytatray boxes containing Arabidopsis plants grown for 21 days. Inoculate mycelia by lifting the stainless steel screen where the Arabidopsis seedlings are, with the help of sterile forceps (Figure 6). Place mycelia on MS media and shake (shaker Certomat® R) in order to promote dispersion.

Figure 5. Set up of the filtration system to harvest the spores
Figure 6. Procedure to inoculate the mycelia into the Phytatray boxes - Finally, keep hydroponic cultures for 20 days at 80 rpm and 22 °C in a plant growth chamber with 40% humidity under long daylight conditions (16 h light/8 h dark) (light intensity of 80 to 100 µE/µm2/s). Afterwards, roots are recovered using sterile scissors on a laminar flow cabinet. Wash the roots with sterile water to remove mycelia traces. Dry the roots on filter paper and finally freeze them in liquid nitrogen. Lyophilize using a Lyophilizer Virtis Advantage until the water from the roots is entirely removed. Collected samples are ready for nucleic acid extraction or qPCR procedures. Success of root invasion is analyzed by qPCR. The corresponding protocols are described in Alonso-Ramírez et al. (2014).
Recipes
- Sterilization solution
2.5% sodium hypochlorite
0.005% Triton X-100
dH2O - MS medium
9 g of Murashige & Skoog medium, including B5 vitamins, for 1 L
1% sucrose
Adjust pH to 5.7 with KOH
Qs. dH2O 1 L
Sterilized for 20 min at 120 °C/1 atm using an autoclave - Washing solution
70% ethanol
1% Triton X-100
dH2O
Acknowledgments
Research project funding was from Junta de Castilla y León (SA260A11-2) and Spanish national projects MICINN (AGL2009-13431-C02) and MINECO (AGL2012-40041-C02-01).
References
- Alonso-Ramirez, A., Poveda, J., Martin, I., Hermosa, R., Monte, E. and Nicolas, C. (2014). Salicylic acid prevents Trichoderma harzianum from entering the vascular system of roots. Mol Plant Pathol 15(8): 823-831.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Alonso-Ramírez, A., Poveda, J., Martín, I., Hermosa, R., Monte, E. and Nicolás, C. (2015). Trichoderma harzianum Root Colonization in Arabidopsis. Bio-protocol 5(13): e1512. DOI: 10.21769/BioProtoc.1512.
Category
Plant Science > Plant immunity > Host-microbe interactions
Microbiology > Microbe-host interactions > Fungus
Microbiology > Microbe-host interactions > In vivo model > Plant
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









