- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
An Experimental Model of Neonatal Nociceptive Stimulation in Rats
Published: Vol 4, Iss 21, Nov 5, 2014 DOI: 10.21769/BioProtoc.1283 Views: 10641
Reviewed by: Soyun KimAntoine de MorreeAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Evaluating Working Memory on a T-maze in Male Rats
Ahmed M. Hussein [...] Volker Korz
Jul 20, 2018 10283 Views
Classic Labyrinth Test for Neurobehavioral Evaluation in Wistar Rats
Salim Gasmi
Sep 20, 2018 6881 Views

Consummatory Successive Negative Contrast in Rats
Ana María Jiménez-García [...] Ignacio Morón
Apr 5, 2019 5054 Views
Abstract
In order to survive, preterm and/or sick neonates need diagnostic and therapeutic measures that may cause discomfort, stress and pain during a critical period of intense growing and modeling of the central nervous system (Anand et al., 2013). Scientific interest in the long lasting effects of the Neonatal Intensive Care (NIC) experience, which provides a sensory experience completely different from the natural uterine environment, is growing (Jobe, 2014). The follow-up of critically ill newborn infants until adulthood indicated an association of early noxious stimuli with long lasting alterations in somatosensory and cognitive processing (Doesburg, 2013; Vinall et al., 2014; Vinall et al., 2013). However, one major limitation of the clinical studies is the difficulty to distinguish between long-term effects of pain suffered during neonatal intensive care and other confounding factors such as the presence of non-painful stress during hospital stay, the occurrence of acute and chronic morbidities, the post-natal environmental influences and family care. In this context, the understanding of the roles played by each factor and the interplay between these diverse variables require the use of animal models. The protocol described here is used to model the noxious stimulation in which premature newborns are subjected during treatment in the NIC. The current protocol models inflammatory nociceptive stimulation in neonatal rats, as previously demonstrated (Leslie et al., 2011; Lima et al., 2014; Malheiros et al., 2014). Complete Freund's adjuvant (CFA) is a solution of antigen emulsified in mineral oil and used as an immunopotentiator, causing a painful reaction that lasts 7-8 days after subcutaneous injection. It is effective in stimulating cell-mediated immunity. The rodent model of neonatal inflammatory stimulation with CFA is advantageous because at birth the formation of the central nervous system is incomplete in rat pups and corresponds to that of 24 week intra-uterine human preterm neonates (Anand et al., 1999), following similar patterns in the development of the pain system (Fitzgerald and Anand,1993). The first postnatal week in newborn rat pups corresponds to human premature infants from 24-36 weeks of gestation (Kim et al., 1996; Wilson, 1995), offering a suitable condition to model and compare preterm (rat pups on P1) to full term (rat pups on P8) infants subjected to noxious stimulation. In this paper, we present our methods to induce nociceptive inflammatory stimulation in neonatal rat pups as an attractive approach to study short- or long-term effects and the mechanisms underlying the behavioral repertoire of ex-premature infants or adolescents.
Keywords: PainMaterials and Reagents
- Pregnant Wistar rats are acquired around the 14th day of gestation
Note: It is important to determine the P0 (postnatal day 0).
- Complete Freund’s adjuvant (CFA) (25 µl) (Sigma-Aldrich, catalog number: F5881 )
Note: The CFA is diluted in 0,9% sterile saline just before use (2:1, CFA:saline).
Equipment
- Plastic cages (for maintaining the animals)
- Automatic temperature control system
- Insulin Syringe Ultra-Fine 6 mm (15/64”) x 31 G needle
- Electronic scale (sensitivity 0.1 g)
Procedure
Ethical statement: All procedures discussed here are in accordance with and were approved by the Independent Ethics Committee of the Universidade Federal de São Paulo.
- Obtain approval from local ethics committee.
- Pregnant Wistar rats from the 14th day of gestation have to be housed individually in plastic cages and placed in the animal facility, equipped with an automatic temperature control system (23 ± 2 ºC), ventilation, a 12-h light-dark cycle (lights on at 7:00 AM) and unrestricted access to food and water.
- The day of birth is designated as P0.
- Each litter is restricted to 8 pups within 24 h after birth, excluding those pups with body weights lower than 0.5 g.
- From P1 (postnatal day one) on, animals from all litters have their body weight recorded on a weekly basis, with an electronic scale (sensitivity 0.1 g), until the end of the experimental protocol.
- To compare the effects of CFA injection during neural development, on postnatal days 1, 8 or 21 rat pups receive a single intraplantar subcutaneous injection of an inflammatory agent, the complete Freund’s adjuvant (CFA; 25 µl) into the left hindpaw.
Note: Take care to avoid spending more than 2 min with each pup. This would cause higher stress levels in the dam.
- All animals in a single litter are assigned to a particular group (CFA-stimulated at P1, P8, P21 or control), to avoid the influence that handling of pups at different ages could have on maternal care towards the remainder of the litter.
- After CFA injection, the pups return to their home-cage with their dams.
- As a result, paws of stimulated pups show swelling, edema and redness, starting a few hours after injection, lasting 5-7 days. The edema can be visually observed, as in Figure 1.
- Control animals are subjected to needle insertion like the experimental pups, but without fluid infusion.
- All litters are weaned on P21, and from then on, housed in same-sex groups of 5 animals of the same experimental group at most.
- To avoid litter effects, pups from different litter were randomly assigned to different groups.
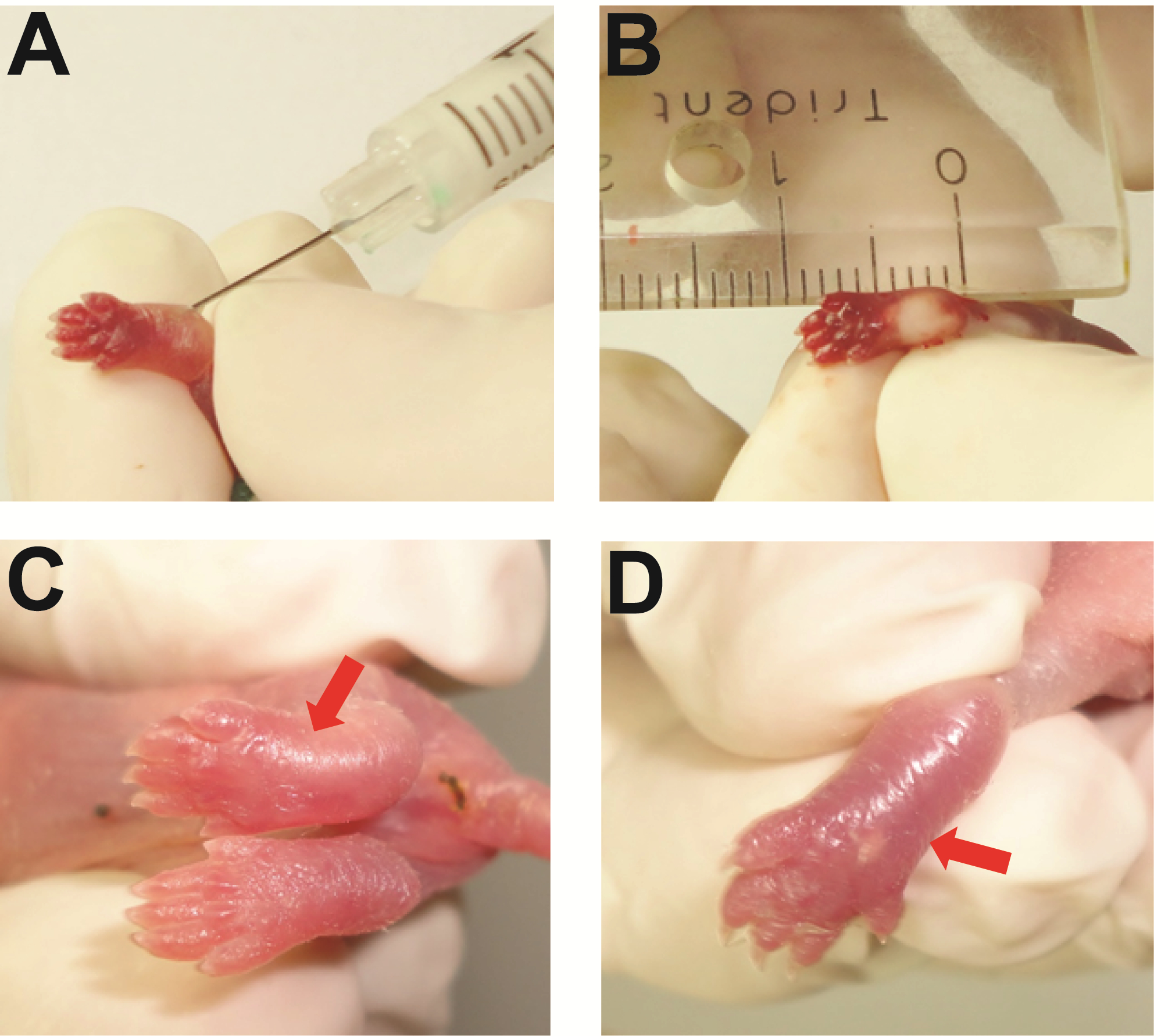
Figure 1. In A the intraplantar subcutaneous injection of CFA on postnatal day 1. Just after the injection B the bleaching and swelling under the skin of the left paw is visible. At day 3 C and day 6 D after CFA injection, the left paw is still swollen (red arrow) compared with the right paw.
Notes
All procedures are performed before 10 AM when the levels of corticosterone are low.
Acknowledgments
This work was supported by FAPESP, by a grant to LC (09/53646-0).
References
- Anand, K. J., Coskun, V., Thrivikraman, K. V., Nemeroff, C. B. and Plotsky, P. M. (1999). Long-term behavioral effects of repetitive pain in neonatal rat pups. Physiol Behav 66(4): 627-637.
- Anand, K. J., Palmer, F. B. and Papanicolaou, A. C. (2013). Repetitive neonatal pain and neurocognitive abilities in ex-preterm children. Pain 154(10): 1899-1901.
- Doesburg, S. M., Chau, C. M., Cheung, T. P., Moiseev, A., Ribary, U., Herdman, A. T., Miller, S. P., Cepeda, I. L., Synnes, A. and Grunau, R. E. (2013). Neonatal pain-related stress, functional cortical activity and visual-perceptual abilities in school-age children born at extremely low gestational age. Pain 154(10): 1946-1952.
- Fitzgerald, M. and Anand, K. (1993). The developmental neuroanantomy and neurophysiology of pain. In: Schechter, N., Berde, C. and Yaster, M. (eds). Pain management in infants, children and adolescents. Baltimore: Williams & Williams, pp. 11-32.
- Jobe, A. H. (2014). A risk of sensory deprivation in the neonatal intensive care unit. J Pediatr 164(6): 1265-1267.
- Kim, J. J., Foy, M. R. and Thompson, R. F. (1996). Behavioral stress modifies hippocampal plasticity through N-methyl-D-aspartate receptor activation. Proc Natl Acad Sci U S A 93(10): 4750-4753.
- Leslie, A. T., Akers, K. G., Martinez-Canabal, A., Mello, L. E., Covolan, L. and Guinsburg, R. (2011). Neonatal inflammatory pain increases hippocampal neurogenesis in rat pups. Neurosci Lett 501(2): 78-82.
- Lima, M., Malheiros, J., Negrigo, A., Tescarollo, F., Medeiros, M., Suchecki, D., Tannus, A., Guinsburg, R. and Covolan, L. (2014). Sex-related long-term behavioral and hippocampal cellular alterations after nociceptive stimulation throughout postnatal development in rats. Neuropharmacology 77: 268-276.
- Malheiros, J. M., Lima, M., Avanzi, R. D., Gomes da Silva, S., Suchecki, D., Guinsburg, R. and Covolan, L. (2014). Repetitive noxious neonatal stimuli increases dentate gyrus cell proliferation and hippocampal brain-derived neurotrophic factor levels. Hippocampus 24(4): 415-423.
- Vinall, J., Miller, S. P., Bjornson, B. H., Fitzpatrick, K. P., Poskitt, K. J., Brant, R., Synnes, A. R., Cepeda, I. L. and Grunau, R. E. (2014). Invasive procedures in preterm children: brain and cognitive development at school age. Pediatrics 133(3): 412-421.
- Vinall, J., Miller, S. P., Synnes, A. R. and Grunau, R. E. (2013). Parent behaviors moderate the relationship between neonatal pain and internalizing behaviors at 18 months corrected age in children born very prematurely. Pain 154(9): 1831-1839.
- Wilson, D. A. (1995). NMDA receptors mediate expression of one form of functional plasticity induced by olfactory deprivation. Brain Res 677(2): 238-242.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Malheiros, J. M., Amaral, C., Leslie, A. T. S., Guinsburg, R. and Covolan, L. (2014). An Experimental Model of Neonatal Nociceptive Stimulation in Rats. Bio-protocol 4(21): e1283. DOI: 10.21769/BioProtoc.1283.
Category
Neuroscience > Behavioral neuroscience > Animal model > Rat
Neuroscience > Nervous system disorders > Animal model
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









