- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Identification of Helminth-induced Type 2 CD4+ T Cells and ILC2s
Published: Vol 4, Iss 11, Jun 5, 2014 DOI: 10.21769/BioProtoc.1141 Views: 9314
Reviewed by: Lee-Hwa TaiAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Protocol for Screening Host-Targeting Antivirals (HTAs) Using Human PBMCs and pDCs
Zhao Xuan Low [...] Pouya Hassandarvish
Mar 5, 2025 3068 Views

Novel Experimental Approach to Investigate Immune Control of Vascular Function: Co-culture of Murine Aortas With T Lymphocytes or Macrophages
Taylor C. Kress [...] Eric J. Belin de Chantemèle
Sep 5, 2025 3551 Views
Dual Phospho-CyTOF Workflows for Comparative JAK/STAT Signaling Analysis in Human Cryopreserved PBMCs and Whole Blood
Ilyssa E. Ramos [...] James M. Cherry
Nov 20, 2025 2351 Views
Abstract
After activation, T cells differentiate into different T helper (Th) subsets, namely Th1, Th2, and Th17. These different Th subsets are associated with the production of particular cytokines endowing them with different functions. In immunity against helminth infections the Th2 cell subset plays an important role. Th2 cells typically produce IL-4, IL-5, IL-13, and IL-9 resulting in antibody-isotype switching to IgE, eosinophilia, basophilia, mucin production, and smooth muscle cell hyperactivity. Here we analyze the development of the pathogen specific Th2 immune responses in mice after infection with the helminth parasite, Heligmosomoides polygyrus bakeri, the induction of innate lymphoid cells type 2 (ILC2) and the activation of the inflammasome in macrophages by excretory/secretory products of Heligmosomoides polygyrus bakeri.
Part I.Generation and collection of Heligmosomoides poylgyrus (Hp) excretory/secretory (ES) products (HES)
Materials and Reagents
- Adult L5 Heligmosomoides poylgyrus (Hp)
- 50 ml PBS (Life Technologies, Gibco®, catalog number: 10010-015 )
- Complete RPMI (GE Healthcare, catalog number: E15-840 )
- 10,000 MWCO cellulose membrane (EMD Millipore, Centriprep®)
- EndoTrap Blue 5/1 LPS-binding affinity column (Hyglos GmbH, catalog number: 311063 )
- HES wash solution I (see Recipes)
- HES wash solution II (see Recipes)
- HES culturing solution (see Recipes)
Equipment
- Aluminum foil
- 50 ml Falcon tube (BD, catalog number: 352070 )
- Pipette
- Falcon flask (TPP Techno Plastic Products, catalog number: 90076 )
- Nanodrop (Thermo Fisher Scientific)
- Centrifuge (Eppendorf, model: 5810R )
- Perti dish
- Microscope (OLYMPUS, model: CKX31 )
- Laminar airflow cabinet (Faster Ultrasafe, model: US218D )
Procedure
- Work sterile under using a laminar airflow cabinet.
- Wash adult L5 Hp extensively in sterile 50 ml PBS in a 50 ml Falcon tube.
- Mix by inverting the Falcon flask. Wait 1-5 min until L5 Hp sink to the bottom of the flask, then remove as much as possible of the supernatant by using a pipette.
- Repeat steps 1-2 for 3x.
- Transfer L5 Hp into 50 ml wash solution I as it was previously described by Camberis et al. (2003).
- Mix by inverting the Falcon tube. Wait 1-5 min until L5 Hp sink to the bottom of the flask, then remove as much as possible of the supernatant.
- Repeat steps 4-5 for 3x.
- Transfer settled L5 Hp into new 50 ml Falcon tube using a 10 ml pipette and gently suck the worms from the bottom of the 50 ml tube and fill up with to 50 ml with wash solution II.
- Mix by inverting and incubate 1 h at RT.
- Count L5 Hp worms by transfering to a perti dish using a dissecting microscope.
- Transfer settled L5 Hp worms into an appropriate cell culture flask at 100 worms/ml in HES culturing solution.
- Incubate at 37 °C in cell culture isolator with 5% CO2.
- Collected supernatant every 2 day using a pipette for a period of 2 weeks, followed by sterile filtration and concentration of the supernatant by centrifugation through a 10,000 MWCO cellulose membrane using a swinging bucket rotor at 4,000 x g, 25 °C, and a starting volume of 15 ml. Refresh the medium in the worm culture flask with the same volume of culturing solution. Measure the protein concentration in the concentrated HES using a NanoDrop.
- Remove LPS contamination from HES culture supernatants using an EndoTrap Blue LPS-binding affinity column according to the manufacturer instructions.
Recipes
- HES wash solution I
500 ml PBS
2 ml gentamycin reagent solution (50 mg/ml)
10 ml 100x penicillin-streptomycin solution (10,000 U/ml) - HES wash solution II
500 ml RPMI 1640
2 ml gentamycin reagent solution (50 mg/ml)
10 ml 100x penicillin-streptomycin solution (10,000 U/ml) - HES culturing solution
500 ml RPMI 1640
1 ml gentamycin reagent solution (50 mg/ml)
5 ml 100x penicillin-streptomycin solution (10,000 U/ml)
50 ml 10% glucose
Part II.Identification of helminth-induced type 2 CD4+ T cells
Materials and Reagents
- Mesenteric lymph nodes (MLNs) from Hp infected mice
- Trypan blue (Life Technologies, Gibco®, catalog number: 15250-061 )
- Complete RPMI (GE Healthcare, catalog number: E15-840 )
- PMA (phorbol myristate acetate) (Sigma-Aldrich, catalog number: P8139 )
- Ionomycin (Sigma-Aldrich, catalog number: I9657 )
- Brefeldin A (Sigma-Aldrich, catalog number: B7651 )
- PBS (Life Technologies, Gibco®, catalog number: 10010-015 )
- BSA (Roche Diagnostics, catalog number: 10735108001 )
- Live /dead fixable aqua dead cell stain kit (Life Technologies, InvitrogenTM, catalog number: L34957 )
- PFA (Sigma-Aldrich, catalog number: P6148 )
- Saponin (Sigma-Aldrich, catalog number: 47036 )
- EDTA (AppliChem GmbH, catalog number: A3145 )
- FACS buffer (see Recipes)
Equipment
- Aluminum foil
- 70 μm cell strainer (Thermo Fisher Scientific, catalog number: 22363548 )
- 50 ml Falcon tube (BD, catalog number: 352070 )
- Neubauer counting chamber (Assistent®)
- Incubator (Thermo Fisher Scientific, HeracellTM 150i )
- 96 well cell culture plates (Corning, Costar®, catalog number: 3799 )
- Centrifuge (Eppendorf, model: 5810R )
- Microscope (OLYMPUS, model: CKX31 )
- BD LSR II flow cytometer (BD Biosciences)
Procedure
- Work on ice and under sterile conditions.
- Remove mesenteric lymph nodes (MLNs) from Hp infected mice and place in complete RPMI on ice (make sure to remove all fat tissue of the MLNs before you store them in complete RPMI).
- Smash MLNs trough a 70 μm cell strainer placed on an 50 ml Falcon tube and rinse with 20 ml complete RPMI.
- Count live cells using trypan blue and a hemacytometer (Neubauer counting chamber) in smashed MLN cell suspensions.
- Incubate MLN cell suspensions at 5 x 105 live cells per 200 μl complete RPMI supplemented with 5 μg/ml HES in round bottom 96 cell culture well plates in triplicates at 37 °C with 5% CO2.
- Leave cells for 72 h in the incubator.
- Centrifuge 96 well plates at 400 x g for 5 min at 4 °C.
- Discard supernatant (or freeze on -20 °C for further cytokine analysis).
- Resuspend cells in 100 μl complete RPMI supplemented with PMA (10-7 M) and Ionomycin (1 μg/ml).
- Incubate for additional 2 h at 37 °C with 5% CO2.
- Add 100 μl of complete RPMI supplemented with 20 μg/ml Brefeldin A.
- Incubate for additional 2 h at 37 °C with 5% CO2.
- Centrifuge 96 well plates at 400 x g for 5 min at 4 °C.
- Combine triplicates into a single well by resuspending one after the other with the same 200 μl PBS with 0.2% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C.
- Resuspend with 200 μl PBS with 0.2% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C.
- Resuspend with 50 μl/well 24G2 as FcR block (obtained from cell line culture supernatants) (1:800 dilution from stock 1 mg/ml stock solution) for 10 min.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 100 μl PBS with 0.5% BSA supplemented with live /dead fixable aqua dead cell stain kit, according to the manufacturer instructions.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 50 μl PBS with 0.5% BSA including the cell surface antibodies labeled with for FACS analysis.
- Incubate in the dark, e.g. cover with aluminum foil for 30 min.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 200 μl PBS with 0.5% BSA.
- Repeat step 24.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in PBS with 2% PFA for 20 min for fixation.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 200 μl PBS with 0.5% BSA.
- Repeat step 27.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 100 μl saponin (10x diluted in PBS with 0.2% BSA) to permeabilise cells.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 50 μl saponin supplemented with intracellular cytokine antibodies labeled with fluorochrom for FACS analysis.
- Incubate in the dark, e.g. cover with aluminum foil for 40 min.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl saponin.
- Repeat step 32.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Repeat step 34.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 75 μl FACS buffer.
- Filter cells in a 70 μm cell strainer.
- Acquire cells at the LSRII.
Recipes
- FACS buffer
500 ml PBS
25 ml BSA (10%)
20 ml EDTA (20 mM)
Part III.Identification of helminth-induced ILC2s
Materials and Reagents
- Mesenteric lymph nodes (MLNs) and spleens from Hp infected mice
- Complete RPMI (GE Healthcare, catalog number: E15-840 )
- PBS (Life Technologies, Gibco®, catalog number: 10010-015 )
- BSA (Roche Diagnostics, catalog number: 10735108001 )
- Live /dead fixable aqua dead cell stain kit (Life Technologies, InvitrogenTM, catalog number: L34957 )
- Biotin labeled cell surface antibodies (Biolegend)
- C-kit (CD117) Pacific Blue (clone: 2B8 ) (Biolegend)
- sca-1 (Ly-6A/E) PE-Cy7 (clone: D7) (eBioscience)
- T1/ST2 FITC (clone: DJ8) (MD Bioproducts)
- ICOS (CD278) PerCp-Cy5.5 (clone: C398.4A ) (Biolegend)
- Streptavidin PE-TexasRed (BD Biosciences)
- FACS buffer (see Recipes)
Equipment
- 70 μm cell strainer
- 50 ml Falcon tube
- 3 ml syringe plunger
- 96 well plates
- BD LSR II flow cytometer (BD Biosciences)
Procedure
- Work on ice.
- Remove mesenteric lymph nodes (MLNs) and spleens from Hp infected mice and place them in complete RPMI on ice (make sure to remove all fat tissue of the MLNs and spleens before you store them in complete RPMI).
- Smash MLNs and spleens trough a 70 μm cell strainer placed on an 50 ml Falcon tube by using a 3 ml syringe plunger and rinse with 20 ml complete RPMI.
- Count live cells in smashed cell suspensions.
- Transfer up to 1 x 106 cells in 200 μl PBS with 0.2% BSA in a single round bottom 96 well.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation.
- Resuspend with 200 μl PBS with 0.2% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation.
- Resuspend with 50 μl/well 24G2 (1:800 dilution from stock 1 mg/ml stock solution) for 10 min.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C and resuspend in 100 μl PBS with 0.5% BSA supplemented with live /dead fixable aqua dead cell stain kit, according to the manufacturer instructions.
- Incubate in the dark, e.g. cover with aluminum foil for 20 min.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 50 μl PBS with 0.5% BSA including the biotin labeled cell surface antibodies (CD3, CD4, CD11b, CD11c, CD19, Nk1.1, Ly6G and IgE) for the lineage negative gating and c-kit (Pacific Blue), sca-1 (PE-Cy7), T1/ST2 (FITC), ICOS (PerCp/Cy5.5).
- Incubate in the dark, e.g. cover with aluminum foil for 30 min.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 50 μl PBS with 0.5% BSA including the streptavidin Texas red labeled antibody.
- Incubate in the dark, e.g. cover with aluminum foil for 30 min.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 50 μl PBS with 2% PFA for 20 min for fixation.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 200 μl PBS with 0.5% BSA.
- Repeat step 20.
- Centrifuge at 400 x g for 5 min at 4 °C. Discard supernatant after centrifugation and resuspend in 75 μl FACS buffer.
- Filter cells in a 70 μm cell strainer.
- Acquire cells at the LSRII.
- Gating strategy is shown in Figure 1:
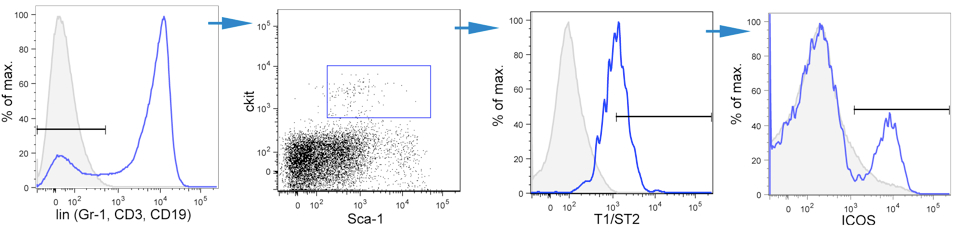
Figure 1. Gating strategy used to identify ILC2s. ILC2 cells were identified as lineage-negative (Gr-1, CD3, CD19), Sca-1, c-kit, T1/ST2 and ICOS positive live cells present in the spleen of mice at 6 days post infection with the helminth parasite Heligmosomoides polygyrus. Y-axis represents counts if not otherwise indicated.
Recipes
- FACS buffer
500 ml PBS
25 ml BSA (10%)
20 ml EDTA (20 mM)
Acknowledgments
This protocol is an extended version of the one described in Zaiss et al. (2013). The research has received funding from the European community seventh framework program [FP7/2009–2014] under EC-GA no[241642] and part of this work was funded by grants from the Swiss National Science Foundation and the Institute of Arthritis Research. KMM is supported by EMBO long-term fellowship and the Australian NHMRC post-doctoral fellowship.
Competing interests
The authors declare no conflict of interest or competing interest.
References
- Camberis, M., Le Gros, G. and Urban, J., Jr. (2003). Animal model of Nippostrongylus brasiliensis and Heligmosomoides polygyrus. Curr Protoc Immunol Chapter 19: Unit 19 12.
- Zaiss, M. M., Maslowski, K. M., Mosconi, I., Guenat, N., Marsland, B. J. and Harris, N. L. (2013). IL-1beta suppresses innate IL-25 and IL-33 production and maintains helminth chronicity. PLoS Pathog 9(8): e1003531.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Readers should cite both the Bio-protocol article and the original research article where this protocol was used:
- Zaiss, M. M. and Maslowski, K. M. (2014). Identification of Helminth-induced Type 2 CD4+ T Cells and ILC2s. Bio-protocol 4(11): e1141. DOI: 10.21769/BioProtoc.1141.
- Zaiss, M. M., Maslowski, K. M., Mosconi, I., Guenat, N., Marsland, B. J. and Harris, N. L. (2013). IL-1beta suppresses innate IL-25 and IL-33 production and maintains helminth chronicity. PLoS Pathog 9(8): e1003531.
Category
Immunology > Immune cell function > Cytokine
Immunology > Host defense > Murine
Immunology > Immune cell function > Lymphocyte
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










