- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
3D Mammary Colony-Forming Cell Assay
Published: Vol 4, Iss 7, Apr 5, 2014 DOI: 10.21769/BioProtoc.1087 Views: 17245
Reviewed by: Vanesa Olivares-IllanaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
A Simplified 3D-Plasma Culture Method for Generating Minimally Manipulated Autologous Equine Muscle-Derived Progenitor Cells
Hélène Graide [...] Didier Serteyn
Dec 5, 2025 1295 Views
Optimization of Adipogenic Differentiation Protocol for Murine and Human Cell Culture Models
Junwan Fan [...] Wenyan He
Jan 20, 2026 314 Views
Simple and Rapid Model to Generate Differentiated Endometrial Floating Organoids
Adriana Bajetto [...] Tullio Florio
Feb 5, 2026 259 Views
Abstract
The mammary epithelium consists of multiple phenotypically and functionally distinct cell populations, which are organized as a hierarchy of stem cells, progenitors and terminally differentiated cells.
Identification of the mechanisms regulating the growth and differentiation of mammary stem and progenitor cells is of great interest not only to better understand the mammary gland development but also to clarify the origins of breast cancer, as these cells seem to be the likely targets of malignant transformation within the mammary epithelium. Hence, a variety of approaches have been developed for quantifying and studying these specific mammary cell subsets.
Given their high proliferative capacity, mammary progenitor cells are able to form colonies in vitro in low-density cultures. Here we describe how to perform a three dimensional (3D) Mammary Colony-Forming Cell (Ma-CFC) Assay, an in vitro functional assay suitable for the detection and analysis of mammary progenitor cells in feeder-free culture conditions.
Briefly, this protocol involves the seeding of mammary single cells, at clonal density, onto a semi-solid matrix (Matrigel), thus allowing mammary progenitors to proliferate and give rise to discrete 3D colonies. The number and the cell composition of the resulting colonies will vary according to the frequency and the differentiation potential of the progenitors, respectively.
Materials and Reagents
- Single-cell suspension of primary mouse mammary cells [see Debnath et al. (2003) for details about the dissociation of mouse mammary glands into single cells]
- EpiCult®-B Basal Medium Mouse (STEMCELL Technologies, catalog number: 05611 )
- EpiCult®-B Proliferation Supplements (STEMCELL Technologies, catalog number: 05612 )
- Trypan blue
- Recombinant human Epidermal Growth Factor (EGF) (Sigma-Aldrich, catalog number: E9644 )
- Recombinant human basic Fibroblast Growth Factor (bFGF) (Life Technologies, catalog number: PHG0023 )
- Heparin sodium salt (Sigma-Aldrich, catalog number: H3149 )
- Fetal Bovine Serum (FBS) (heat-inactivated) (Life Technologies, catalog number: 10270-106 )
- Penicillin-Streptomycin (Pen/Strep) (Life Technologies, catalog number: 15070-063 )
- Growth Factor Reduced (GFR) BD MatrigelTM Matrix [protein concentration > 8 mg/ml, endotoxin levels < 2 Endotoxin Units (EU)/ml] (BD Biosciences, catalog number: 354230 )
Note: BD MatrigelTM Matrix is supplied as a frozen solution. Thaw it on ice overnight and stored as 1 ml aliquots at -20 °C. Leave Matrigel aliquots to thaw on ice for 2 h before use.
- Complete EpiCult-B Medium (see Recipes)
Equipment
- BD Falcon 8-well culture slides (BD Biosciences, catalog number: 354108 )
- Refrigerated centrifuge with swinging bucket rotor
- Humidified 37 °C, 5% CO2 cell culture incubator
- Inverted tissue culture microscope
- Zeiss Observer Z.1 microscope (5x/0.12 objective)
Software
- AxioVision Rel 4.8 software
- ImageJ
Procedure
- The 8-well chamber slide is placed on ice and loaded with 80 µl of Matrigel per well. The Matrigel drop is positioned in the centre of each well, taking care to avoid air-bubble formation.
Note: To prevent gelation, it is recommended to handle Matrigel on ice.
- Next, the chamber slide is secured onto a pre-cooled adapter for swing-bucket rotor and centrifuged at 300 x g (with a slow acceleration ramp) for 10 min at 4 °C. The Matrigel layer is now flat and uniformly distributed onto the bottom of the chamber wells.
Note: It is convenient to use some plastic paraffin film to keep the lid of the culture slide in place during step 2.
- The chamber slide is then transferred to a cell culture incubator to allow Matrigel solidification for at least 15 min.
- Meanwhile, freshly prepared single mouse mammary cells are resuspended in Complete EpiCult-B Medium and counted.
Note: Trypan Blue stain is one of several methods recommended for viable cell counting.
- The cell suspension is diluted in Complete EpiCult-B Medium to obtain a final concentration of 25,000 viable cells/ml.
- A stock of Complete EpiCult-B Medium supplemented with 5% Matrigel is prepared by adding 50 µl of Matrigel per ml of Complete EpiCult-B Medium and mixing by pipetting repeatedly.
- The cell suspension from step 5 is mixed with the medium from step 6 in a 1:1 ratio. 400 µl of this mixture, which correspond to 5,000 cells in Complete EpiCult-B Medium 2.5% Matrigel, are gently plated per well onto the solidified Matrigel from step 3 (Figure 1).
- The chamber slide is placed in the cell culture incubator. The cells will settle onto the solid Matrigel layer.
- The overlay liquid medium is replaced with fresh Complete EpiCult-B Medium supplemented with 2.5% Matrigel every 3 days. To avoid damaging the bottom gel layer, the medium is aspirated from the corner of each well and the fresh medium is gently dispensed along the walls.
- Colonies will be generated after 10-14 days of culture. Representative images are shown in Figure 2.
- The number of colonies can be determined, using an inverted microscope, either by manual counting or with the support of image analysis programs, such as Image J software.
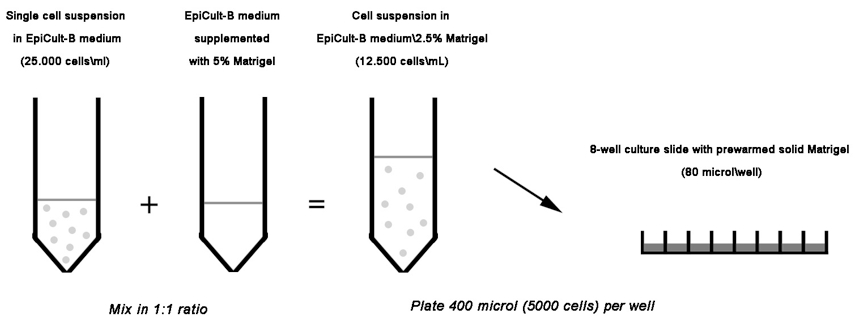
Figure 1. Schematic diagram depicting the procedure described above as step 7
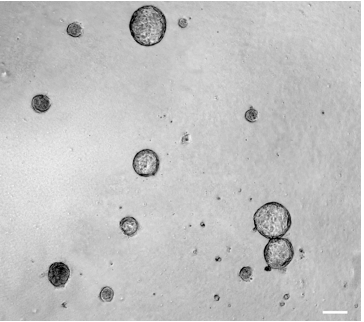
Figure 2. 3D mammary colony-forming cell assay. Phase-contrast image of colonies derived from 3D culture (day 10) of mouse mammary single cells. This image was obtained by using a Zeiss Observer Z.1 microscope (5x/0.12 objective) and the AxioVision Rel 4.8 software. Scale bar, 100 µm
Recipes
- Complete EpiCult-B Medium (50 ml)
EpiCult-B Basal Medium: 42 ml
EpiCult®-B Proliferation Supplements: 5 ml
FBS: 2.5 ml
Pen/Strep: 0.5 ml
EGF (0.2 mg/ml): 2.5 µl
bFGF (0.2 mg/ml): 2.5 µl
Heparin (50 mg/ml): 4 µl
Filter-sterilize through a 0.2 µm filter
Stored at 4 °C and use within one week
Complete EpiCult-B Medium must be supplemented with Matrigel immediately before use
Acknowledgments
We thank Dr Agata Tinnirello and Prof Senthil Muthuswamy for technical advice on the 3D overlay cell-culture method (previously described by Debnath et al. (2003). This work was funded by AIRC (Grant IG2008 and IG2011), MIUR (FIRB Giovani 2008), PiSTEM and Progetto Sanità Finalizzata. G. Tornillo was supported by a FIRC fellowship.
References
- Debnath, J., Muthuswamy, S. K. and Brugge, J. S. (2003). Morphogenesis and oncogenesis of MCF-10A mammary epithelial acini grown in three-dimensional basement membrane cultures. Methods 30(3): 256-268.
- Tornillo, G., Elia, A. R., Castellano, I., Spadaro, M., Bernabei, P., Bisaro, B., Camacho-Leal Mdel, P., Pincini, A., Provero, P., Sapino, A., Turco, E., Defilippi, P. and Cabodi, S. (2013). p130Cas alters the differentiation potential of mammary luminal progenitors by deregulating c-Kit activity. Stem Cells 31(7): 1422-1433.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Tornillo, G. and Cabodi, S. (2014). 3D Mammary Colony-Forming Cell Assay. Bio-protocol 4(7): e1087. DOI: 10.21769/BioProtoc.1087.
Category
Stem Cell > Adult stem cell > Maintenance and differentiation
Cell Biology > Cell isolation and culture > 3D cell culture
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











