- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Analysis of Sugar Component of a Hot Water Extract from Arabidopsis thaliana Pollen Tubes Using GC-EI-MS
Published: Vol 5, Iss 12, Jun 20, 2015 DOI: 10.21769/BioProtoc.1503 Views: 9204
Reviewed by: Samik BhattacharyaFeng LiAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Enzymatic Starch Quantification in Developing Flower Primordia of Sweet Cherry
Nestor Santolaria [...] Afif Hedhly
Apr 5, 2025 1935 Views
New Approach to Detect and Isolate Rhamnogalacturonan-II in Arabidopsis thaliana Seed Mucilage
Dayan Sanhueza and Susana Saez-Aguayo
Sep 5, 2025 1285 Views
Detailed Method for the Purification of Rhamnogalacturonan-I (RG-I) in Arabidopsis thaliana
Liang Zhang [...] Breeanna R. Urbanowicz
Feb 5, 2026 241 Views
Abstract
Extraction with hot water is the oldest and simplest method used to recover pectin from an alcohol insoluble residue extract, although this method has not been widely used for the cell wall analysis of pollen tube, a model used to study cell wall. This protocol described this method applied for pectin extraction from 6 h-old Arabidopsis pollen tubes followed by a sugar composition analysis by gas chromatography mass spectrometry.
Keywords: Pollen tubeMaterials and Reagents
- Arabidopsis day-0 flowers according to Boavida and McCormick (2007)
- Absolute ethanol (EtOH) for analysis (Merck Millipore, catalog number: 107017 )
- Trifluoroacetic acid 12.93N (TFA) (Sigma-Aldrich, catalog number: T62200 )
- Myo-inositol 2 mM (Sigma-Aldrich, catalog number: I5125 )
- Methanolic-HCl 3N (Sigma-Aldrich, catalog number: 33051 Supelco )
- Methanol (Thermo Fisher Scientific, catalog number: 10010280 )
- Silylation reagent (HMDS:TMCS:Pyridine, 3:1:9 SylonTM HTP) (Sigma-Aldrich, catalog number: 33038 Supelco )
- Cyclohexane (Acros Organics, catalog number: 279590010 )
- Monosaccharides standards
Arabinose (Sigma-Aldrich, catalog number: A3256 )
Fucose (Sigma-Aldrich, catalog number: F8150 )
Galactose (Sigma-Aldrich, catalog number: G0750 )
Galacturonic acid (Sigma-Aldrich, catalog number: 857289 )
Glucose (Merck Millipore, catalog number: 8337.0250 )
Glucuronic acid (Sigma-Aldrich, catalog number: G8645 )
Mannose (Sigma-Aldrich, catalog number: M4625 )
Rhamnose (Sigma-Aldrich, catalog number: R3875 )
Xylose (Sigma-Aldrich, catalog number: X2126 ) - Arabidopsis pollen germination medium (see Recipes)
- Trifluoroacetic acid (2 N) (see Recipes)
Equipment
- Inverted microscope Olympus CK2
- Centrifuge Allegra® X-15R Beckmann Coulter
- Hot-water bath (70 °C and 90 °C)
- 40-ml glass potter homogenizer
- Freeze-dryer
- Incubator (22 °C, 80 °C and 110 °C)
- Sample concentrator under air flow (Techne, catalog number: FSC400D )
- GC-EI-MS instrument is composed of a Zebron Z5-MSi (30 m, 0.25 mm id, 0.25 µm film thickness, Phenomenex) capillary column for chromatographic separations, a Hewlett-Packard 6890 series gas chromatograph coupled to an Autospec mass spectrometer of EBE geometry (Micromass, Manchester, UK) equipped with a Opus 3.1 data system
- 1.5 ml Eppendorf tube
- Vortex
- Pair of tweezers
- 50-ml Falcon
- Microliter syringe (10 µl, Hamilton)
Procedure
- In vitro Arabidopsis pollen tubes growth and fixation
The protocol described below is similar to the one available at Wang and Jiang (2014) with several differences.- Grow in vitro Arabidopsis pollen according to the method described by Boavida and McCormick (2007).
- Harvest 40 freshly open Arabidopsis thaliana flowers (morning is the best).
- Submerge in 1 ml of pollen germination medium in a 1.5 ml Eppendorf tube.
- Shake the tube vigorously manually and using a vortex for 5 min to release the pollen grains from the anthers.
- Check the pollen density with an inverted microscope.
- Remove all the flower debris with a pair of tweezers.
- Spin-down the pollen suspension at 3,200 x g for 7 min (a yellowish pellet should be visible).
- Remove the supernatant.
- Add 250 µl of pollen germination medium and resuspend the pellet.
- Place the tube horizontally in the incubator at 22 °C in the dark for 6 h.
- After 6 h, check for pollen germination and pollen tube growth with the inverted microscope (Figure 1).
- Harvest 40 freshly open Arabidopsis thaliana flowers (morning is the best).
- Fix the pollen culture by adding 750 µl absolute EtOH (final concentration 75% EtOH) and keep at 4 °C (no longer than one month).
Note: Around 90 pollen cultures (3,600 flowers) are needed for the detection of minor sugars such as the Kdo (2-keto-3-deoxy-D-manno-octulosonic acid), one of the constituents of rhamnogalacturonan-II (RG-II). For other major cell wall components, pollen tubes from 400 flowers are generally sufficient.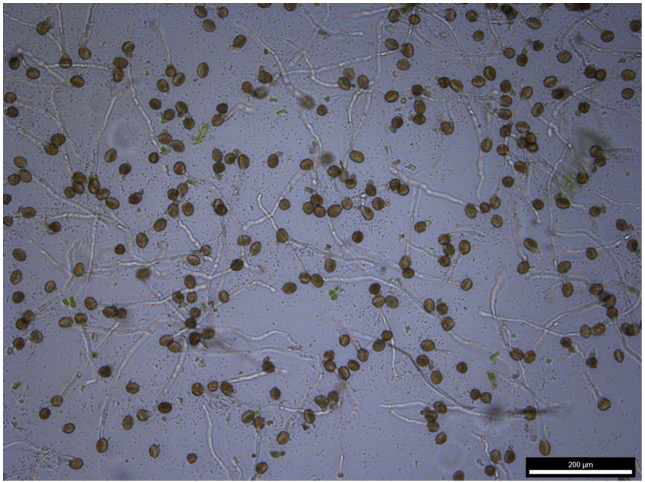
Figure 1. 6 h-old Arabidopsis pollen tubes. Note that the pollen grain density is important to obtain a good germination rate.
- Grow in vitro Arabidopsis pollen according to the method described by Boavida and McCormick (2007).
- Pectin extraction and analysis
- Hot water pectin extraction:
- Pool all the pollen cultures in a 50-ml Falcon® tube and centrifuge at 4,700 x g for 5 min. Repeat the centrifugation step if the total volume of the pollen cultures exceeds 45 ml.
- Discard the supernatant and wash with 15 mL EtOH 70% to remove salt and sucrose from the germination medium.
- Incubate in hot-water bath at 70 °C for 15 min.
- Grind the material manually with a 40-ml glass potter homogenizer until no debris are visible.
- Centrifuge at 4,700 x g for 5 min and discard the supernatant.
- Allow the pellet to dry until no supernatant is visible (it might take no more than 2 h using a stream of dry air).
- Resuspend the pellet in 5 ml distilled water.
- Incubate in a hot-water bath at 90 °C for 1 h.
- Centrifuge at 4,700 x g for 5 min.
- Freeze-dry the supernatant. No liquid must be visible (it might take one day).
- Pool all the pollen cultures in a 50-ml Falcon® tube and centrifuge at 4,700 x g for 5 min. Repeat the centrifugation step if the total volume of the pollen cultures exceeds 45 ml.
- Sample preparation for gas chromatography analysis:
Note: Glassware tubes are preferentially used for the sample preparation. A standard sample containing the most common cell wall monosaccharides (Ara, Fuc, Gal, GalUA, Glc, GlcUA, Man, Rha, Xyl) is prepared at the same time without the TFA hydrolysis step.- Hydrolyze the extract with 250 µl TFA 2 N for 2 h at 110 °C.
- Add 50 µl of myo-inositol 2 mM in the sample. Myo-inositol is used as an internal standard.
- Perform a methanolysis for 16 h at 80 °C using 250 µl of dried 1 M methanolic-HCl (dilute the 3 M stock solution with methanol).
- Wash the sample twice using 500 µl methanol.
- Convert the methyl glycosides into their trimethylsilyl derivatives at 110 °C for 20 min with 200 µl of the silylation reagent (HMDS: TMCS: Pyridine, 3:1:9, Supelco).
- Wash twice the sample using 500 µl cyclohexane.
Note: Air dry the sample using a stream of dry air with a sample concentrator between each step (a nitrogen flow can be used as well). Add 1 ml cyclohexane and vortex the tube.
- Hydrolyze the extract with 250 µl TFA 2 N for 2 h at 110 °C.
- Monosaccharides analysis:
- Inject 0.5 µl of the sample in the GC-EI-MS instrument, using a 10 µl syringe, in splitless mode. Helium is the carrier gas and the flow-rate is 0.8 ml/min.
- Start the temperature programming at 120 °C for 2 min, ramp to 160 °C at 10 °C/min, then to 220 °C at 1.5 °C/min and finally ramp to 280 °C at 15 °C/min (maintain at 270 °C for 1 min). The temperatures of the injector, the interface and the lines are 250 °C. Record the EI mass spectra using an electron energy of 70 eV, an acceleration voltage of 8 kV and a resolving power of 1,000. The trap current of 200 µA and the magnet scan rate is 1 s/decade over a m/z range 600-38. The temperature of ion source is 250 °C.
- The data are processed with the software STAR Workstation Varian 5.52. First of all, it is important to check that peak and peak areas (A) are assigned correctly for each spectrum by comparing with the standard sample before proceeding. The response factor (Rf) for each monosaccharide has been determined previously by calibration of the instrument for each standard sugar. Peak area is integrated on the total chromatogram for each sugar and for the internal standard (inositol). The amount of each sugar is calculated by the formula:

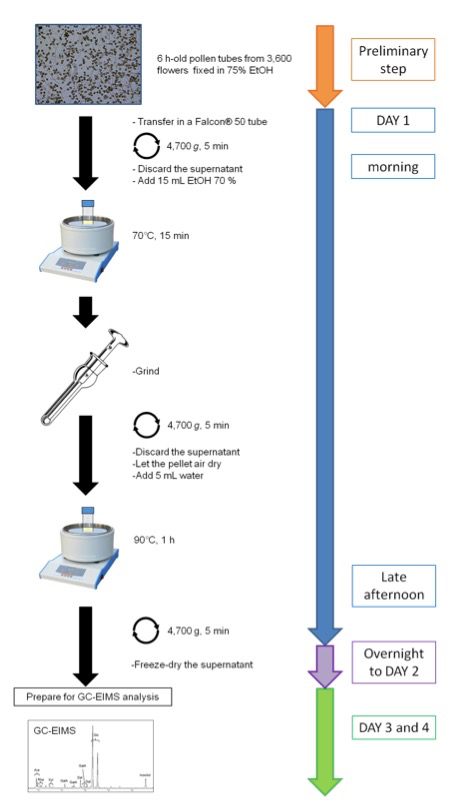
Figure 2. Schematic representation of the protocol for sugar composition of a hot water extract from Arabidopsis thaliana pollen tubes- Recipes Arabidopsis thaliana pollen germination medium
5 mM CaCl2
0.01% H3BO3
1 mM MgSO4
5 mM KCl
10% (w/v) sucrose
pH 7.5 - Trifluoroacetic acid (2 N)
0.15 volume of Trifluoroacetic acid 12.93 N
0.85 volume of fresh distilled water
Note: Use glassware.
- Inject 0.5 µl of the sample in the GC-EI-MS instrument, using a 10 µl syringe, in splitless mode. Helium is the carrier gas and the flow-rate is 0.8 ml/min.
- Hot water pectin extraction:
Acknowledgments
This protocol was initially described in Dumont et al. (2014). This work was supported by the University of Rouen, the region Haute-Normandie, and the “Trans Channel Wallnet” project that has been selected by the INTERREG IVA program France (Channel) – England European cross-border cooperation programme, which is co-financed by the ERDF. The authors also thank the Labex SynOrg (ANR-11-LABX-0029, CRUNCh network, and the European Regional Development Fund (ERDF 31708).
References
- Boavida, L. C. and McCormick, S. (2007). TECHNICAL ADVANCE: Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J 52(3): 570-582.
- Dumont, M., Lehner, A., Bouton, S., Kiefer-Meyer, M. C., Voxeur, A., Pelloux, J., Lerouge, P. and Mollet, J. C. (2014). The cell wall pectic polymer rhamnogalacturonan-II is required for proper pollen tube elongation: implications of a putative sialyltransferase-like protein. Ann Bot 114(6): 1177-1188.
- Wang, H. and Jiang, L. (2014). Immunofluorescence labeling of pollen tubes. Bio-protocol 4(10): e1131.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Dumont, M., Lehner, A., Loutelier-Bourhis, C., Mollet, J. and Lerouge, P. (2015). Analysis of Sugar Component of a Hot Water Extract from Arabidopsis thaliana Pollen Tubes Using GC-EI-MS. Bio-protocol 5(12): e1503. DOI: 10.21769/BioProtoc.1503.
Category
Plant Science > Plant biochemistry > Carbohydrate
Biochemistry > Carbohydrate > Polysaccharide
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link












