- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Application of Silicone Tubing for Robust, Simple, High-throughput, and Time-resolved Analysis of Plant Volatiles in Field Experiments
Published: Vol 5, Iss 3, Feb 5, 2015 DOI: 10.21769/BioProtoc.1391 Views: 15064
Reviewed by: Tie LiuAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Heterologous Production of Artemisinin in Physcomitrium patens by Direct in vivo Assembly of Multiple DNA Fragments
Nur Kusaira Khairul Ikram [...] Henrik Toft Simonsen
Jul 20, 2023 2363 Views
Utilizing FRET-based Biosensors to Measure Cellular Phosphate Levels in Mycorrhizal Roots of Brachypodium distachyon
Shiqi Zhang [...] Maria J. Harrison
Jan 20, 2025 2435 Views
High-Performance Liquid Chromatography Quantification of Glyphosate, Aminomethylphosphonic Acid, and Ascorbate in Culture Medium and Microalgal Cells
Juan Manuel Ostera [...] Gabriela Malanga
Apr 5, 2025 1227 Views
Abstract
Plant volatiles (PVs) mediate manifold interactions between plants and their biotic and abiotic environments (Dicke and Baldwin, 2010; Holopainen and Gershenzon, 2010). An understanding of the physiological and ecological functions of PVs must therefore be based on measurements of PV emissions under natural conditions. Yet sampling PVs in natural environments is difficult, limited by the need to transport, maintain, and power instruments, or else to employ expensive sorbent devices in replicate. Thus PVs are usually measured in the artificial environments of laboratories or climate chambers. However, polydimethysiloxane (PDMS), a sorbent commonly used for PV sampling (Van Pinxteren et al., 2010; Seethapathy and Górecki, 2012), is available as silicone tubing (ST) for as little as 0.60 €/m (versus 100-550 € apiece for standard PDMS sorbent devices). Small (mm-cm) ST pieces can be placed in any experimental setting and used for headspace sampling with little manipulation of the organism or headspace. ST pieces have absorption kinetics and capacities sufficient to sample plant headspaces on a timescale of minutes to hours, producing biologically meaningful “snapshots” of PV blends. When combined with thermal desorption (TD)-GC-MS analysis - a 40-year-old and widely available technology - ST pieces yield reproducible, sensitive, spatiotemporally resolved, quantitative data from headspace samples taken in natural environments (Kallenbach et al., 2014).
Keywords: PolydimethylsiloxaneMaterials and Reagents
- Acetonitrile (gradient grade)
- Methanol (gradient grade)
- Nitrogen (gaseous)
- Scentless soap (Labosol S)
- Argon (gaseous)
Note: Argon provides a clean, inert atmosphere which is heavier than air, for long-term storage of conditioned ST pieces. For shorter time periods, storage under nitrogen is adequate.
Equipment
- Silicone laboratory tubing (ST, 1 mm i.d. x 1.8 mm o.d.) (Carl Roth, catalog number: 9555.1 )
- 4 ml screw-neck glass vials (brown) (Macherey-Nagel, catalog number: 702973 ) and screw caps (N13 PP) (Macherey-Nagel, catalog number: 702051 )
- 1.5 ml screw-neck glass vials (Macherey-Nagel, clear, catalog number: 702282 ; brown, catalog number: 702293 ) and screw caps (N9 polypropylene [PP], Macherey-Nagel, catalog number: 702028)
- Polytetrafluoroethylene (PTFE) seal tape (RS, catalog number: 231-964 )
- Paddle wire
- Retractable-blade utility knife or single-edged razor blade
- Cutting mask (custom-made) for uniformly cutting 5 mm PDMS pieces, made out of polyoxymethylene (POM) (Figure 1A)
Note: PDMS pieces can also be cut manually, but a cutting mask increases uniformity and saves time when cutting large numbers of PDMS pieces.
- Laboratory borosilicate glass bottle (volume: 100-250 ml) (Carl Roth, catalog numbers: A358.1 or A359.1 )
- Stainless steel spatula (Carl Roth, catalog number: 1902.1 )
- Stainless steel forceps (Carl Roth, catalog number: 2687.1 ) and aluminum foil (Carl Roth, catalog number: 0192.1 )
- Glass column (50 ml) with screw-cap ends, containing a size P0 glass frit (Figure 1C)
Note: The glass frit should be sufficiently small to cause a turbulent nitrogen gas flow.
- Polyether ether ketone (PEEK) screw-caps with PTFE lining and stainless steel hose connections, for glass column (Figure 1D)
- Flow meter (5-10 L volume)
- PTFE gas hoses
- Heating oven (max. temperature at least 210 °C)
- Polyethylene tetraphthalate (PET) trapping containers (30-600 ml, Solo or Iso-pack)
Note: The trapping container volume should be as small as possible without significantly changing environmental conditions (e.g. humidity, temperature). This can be tested by enclosing a test sample for the planned trapping period and checking for any visual change (e.g. wilting, condensation) as well as by placing environmental sensors (e.g. temperature, humidity) inside and outside of the trapping container.
- Thermo desorption (TD) unit (Shimadzu, model: TD-20 )
- 89 mm TD sampler tubes (Supelco, catalog number: 28714-U ) and caps with O-rings (Shimadzu, caps, catalog number: 2235461791 and O-rings, catalog number: 2235716691 )
Note: Caps can also be milled from brass as long as the exact dimensions are kept.
- GC-MS (Shimadzu, model: GC-MS-QP2010Ultra )
- 5MS GC column (30 m long, 0.25 mm i.d., 0.25 µm film thickness) (Restek Rtx-5MS)
- WAX column (30 m long, 0.25 mm i.d., 0.25 µm film thickness) (Phenomenex ZB-Wax Plus)
Procedure
- Preparation of PET trapping containers (please see Figure 2 for examples)
- Make a hole of a standard size in the lid of each trapping container (if necessary; 600 ml Iso-pack cups have lids with holes), so that the sample can be easily inserted without plant damage.
- In each cup, make a hole in the bottom of a standard size, to allow movement of air through the container and to enable exchange of ST pieces without opening trapping containers during sampling.
- Thread paddle wire through small holes in each cup to use for fixing trapping containers to supports.
Note that this will block the inner surface of the ST pieces from exposure to the headspace and could thus potentially increase equilibration time, although we have not found such use of wire to significantly change required exposure times.
- Wash cups with scentless soap (Neolab Labosol S) and water and air-dry before and after use.
Note: Paddle wire may be removed before washing to avoid rusting; otherwise, drying should be accelerated by shaking or patting dry, heating at a low temperature (30-50 °C), or airflow.
- Make a hole of a standard size in the lid of each trapping container (if necessary; 600 ml Iso-pack cups have lids with holes), so that the sample can be easily inserted without plant damage.
- Preparation of silicone tubing (ST)
- Cut ST into uniform 5 mm pieces using the knife or scalpel and handmade cutting mask (Figure 1A).
- Collect ST pieces in a laboratory glass bottle and add 4/1 (v/v) acetonitrile/methanol until the ST pieces are fully covered (Figure 1B).
- Soak for approx. 3 h at room temperature.
- Decant the solvent and with the spatula, pour the ST pieces into the glass column with glass frit (Figure 1C).
Note: Dry ST pieces are electrostatic and will stick to glass walls. Pouring with some solvent facilitates the transfer. This should be done over a glass beaker to collect solvent.
- Connect the glass columns to nitrogen gas flow at 5 L/min to flush solvent and contaminants.
- Transfer the closed glass columns into a modified heating oven (Figure 1D).
Note: We laid PTFE gas hose through the ventilation of the heating oven to have a nitrogen connection inside the oven.
- Heat under nitrogen flow (5 L/min) for 1.5 h at 210 °C.
Note: The temperature should be held for 1.5 h. Determine how long your oven requires to reach 210 °C and add this pre-heating time to the total time, e.g. 3 h: 1.5 h heating and 1.5 h at 210 °C. You may, at the same time, place the forceps and spatula on a piece of aluminum foil in the oven, for cleaning.
- Switch off the heating and cool to room temperature under nitrogen flow (5 L/min).
- Transfer the ST pieces into brown 4 ml glass vials.
Note: The dry ST pieces are electrostatic. Small numbers are best transferred by using forceps; larger numbers of ST pieces are best transferred by first pouring them on aluminum foil and sliding them from the foil into the vial, assisted with forceps or gloved fingers.
- Flush the vials with argon, screw closed, and seal the closed vials with PTFE tape (Figure 1E).

Figure 1. Working steps for preparation and conditioning of silicone tubing (ST). Photographs by Danny Kessler and Mario Kallenbach.
- Cut ST into uniform 5 mm pieces using the knife or scalpel and handmade cutting mask (Figure 1A).
- Sampling of PVs from leaf and flower headspaces with ST pieces
- Place the lids on the leaf or flower assigned for trapping as shown in Figure 2A-B.
- Place an appropriate number of ST pieces into the trapping container (Figure 2A-B).
Note: Each ST piece is analyzed only once. We use two to five ST pieces as technical replicates for each sample. It is essential to use the same number of technical replicates for every sample; the total volume of ST in the sample headspace will determine the amount of PVs collected on each individual ST piece.
- Close the trapping containers and collect the volatile headspace (Figure 2C).
Note: In field experiments we stabilize the trapping containers with sticks, tent herrings, stones, etc.
- After the trapping, store the ST pieces in tightly-closed 1.5 ml glass vials.
Note: Technical replicates may be stored in a common vial. This will increase their homogeneity. The storage temperature should be adapted to the compounds of interest. For short term storage (up to several weeks, see Kallenbach et al., 2014) and/or stable substances, storage at room temperature is sufficient; otherwise, storage at -20 °C is recommended.
- Store the ST pieces in vials at room temperature for at least 1 day prior to analysis to allow equilibration between ST pieces and the surrounding atmosphere in the glass vial.
Note: Trapping on ST is an absorptive method with an enrichment of headspace volatiles into the polydimethylsiloxane (PDMS). Therefore, an equilibration between PDMS and the surrounding atmosphere always occurs. Thus for samples which should be directly comparable, we always store similar numbers of technical replicates in similar vial sizes to have an equal ratio of PDMS volume to atmosphere volume.

Figure 2. Experimental setup for headspace volatile sampling from leaves and flowers. Red arrows highlight the positions of ST tubing. Photographs by Danny Kessler and Mario Kallenbach.
- Place the lids on the leaf or flower assigned for trapping as shown in Figure 2A-B.
- TD-GC-MS analysis
- Place an individual ST piece into a TD sampler tube (Figure 3A).
Note: To increase sensitivity, multiple ST pieces may be desorbed in the same TD sampler tube. This will lead to larger silicone peaks in GC-MS chromatograms, and thus it may be necessary to modify the MS method to either switch off the ionizing filament during the elution of known silicone peaks, or else reduce the voltage of the detector during these elution times in order to avoid saturating the detector.
- Metal springs, supplied with the TD sampler tubes, prevent ST pieces from being flushed out of the sampler tube (red arrow in Figure 3B).
- Desorb and analyze the headspace volatile samples by TD-GC-MS (Figure 3C) using the settings described in Table 1 and accompanying method optimization notes.
Note: Blank tubes should be desorbed periodically, e.g. every 10 samples, to check for contamination.
- If required necessary (i.e. in case of detected contamination is detected in blank tubes), clean TD sampler tubes and lids (containing O-rings). Tubes can be cleaned by soaking in acetone for a short period (<1 h is sufficient) – tubes in acetone may also be placed in a sonic bath (15-20 min at room temperature is sufficient) - draining, and heating to 210 °C. Lids can be cleaned by soaking in methanol under the same conditions as tubes, draining, and air-drying under a cover of lint-free tissue paper to avoid dust.
Note: More aggressive solvents and higher temperatures will damage O-rings, while lids will oxidize if cleaned with water.
- Standard solutions can be analyzed by injecting 1 µl of standard at an appropriate concentration (usually ca. 0.1-10 ng/µl, but the range will depend on sensitivity settings of the MS, split setting of the injector, and compound), dissolved in dichloromethane or hexane, onto the inner wall of a clean ST placed in a TD sampler tube. The solvent and standard will be quickly absorbed by the ST.
Note: This method of analyzing standards may not work well for low-volatility compounds with high affinity for PDMS, e.g., long linear alkanes. For such standards, it may be preferable to inject the 1 µl of standard solution directly onto the inner glass wall of the TD sampler tube and quickly close the tube.

Figure 3. Loading of ST pieces into TD sampler tubes and TD-GC-MS-equipment. Photographs by Danny Kessler and Mario Kallenbach.
Table 1. TD-GC-MS settings for leaf and flower headspace analysis
A. TD sampling and injection
TD block
200 °C
TD line
230 °C
GC interface
230 °C
Sampling
N2 flow at 60 ml/min for 8 min at 230 °C
Cryotrap cold temp.
-20 °C
Cryotrap heat temp.
230 °C for 10 min
B. GC separation
Carrier gas
He at 40 cm/sec
Split ratio
1 to 20
Leaf headspace
Column
Rtx-5MS, 30 m long, 0.25 mm i.d., 0.25 µm film thickness
Oven program
40 °C for 5 min, ramp to 185 °C with 5 °C/min,
ramp to 280 °C with 30 °C/min, hold for 0.83 min
Total run time
38 min
Flower headspace
Column
ZB-Wax Plus, 30 m long, 0.25 mm i.d., 0.25 µm film thickness
Oven program
60 °C for 1 min, ramp to 150 °C at 30 °C/min, ramp to 200 °C
at 10 °C/min, ramp to 230 °C at 30 °C/min, hold for 1 min
Total run time
11 min
C. MS detection
Transfer line temperature
240 °C
Ion source temperature
220 °C
Scan range
Full scan from 33 to 400 m/z
Scan speed
0.3 sec/full scan
Method optimization notes: For humid or wet samples, or samples containing large amounts of low-volatility compounds, the TD program can be made more stringent by increasing the N2 flow to 100-150 ml/min and by increasing the sampling time. GC methods can be adjusted as appropriate for specific analytes and columns by altering split ratio, ramping rates, temperatures, and holding times. As described under TD-GC-MS analysis, a timed MS program may be used to reduce or eliminate signal from very abundant peaks or contaminants. The Shimadzu QP2010Ultra MS can be tuned to three different sensitivity ranges for less or more dilute samples. For a few PVs which degrade or re-arrange at high temperatures, it may be desirable to reduce desorption, interface, transfer line, and ion source temperatures to 150-200 °C. All such method adjustments should only be undertaken with the assistance of an experienced TD-GC-MS user.
- Place an individual ST piece into a TD sampler tube (Figure 3A).
Representative data
- Chromatograms of Nicotiana attenuata leaf and flower headspace collections.
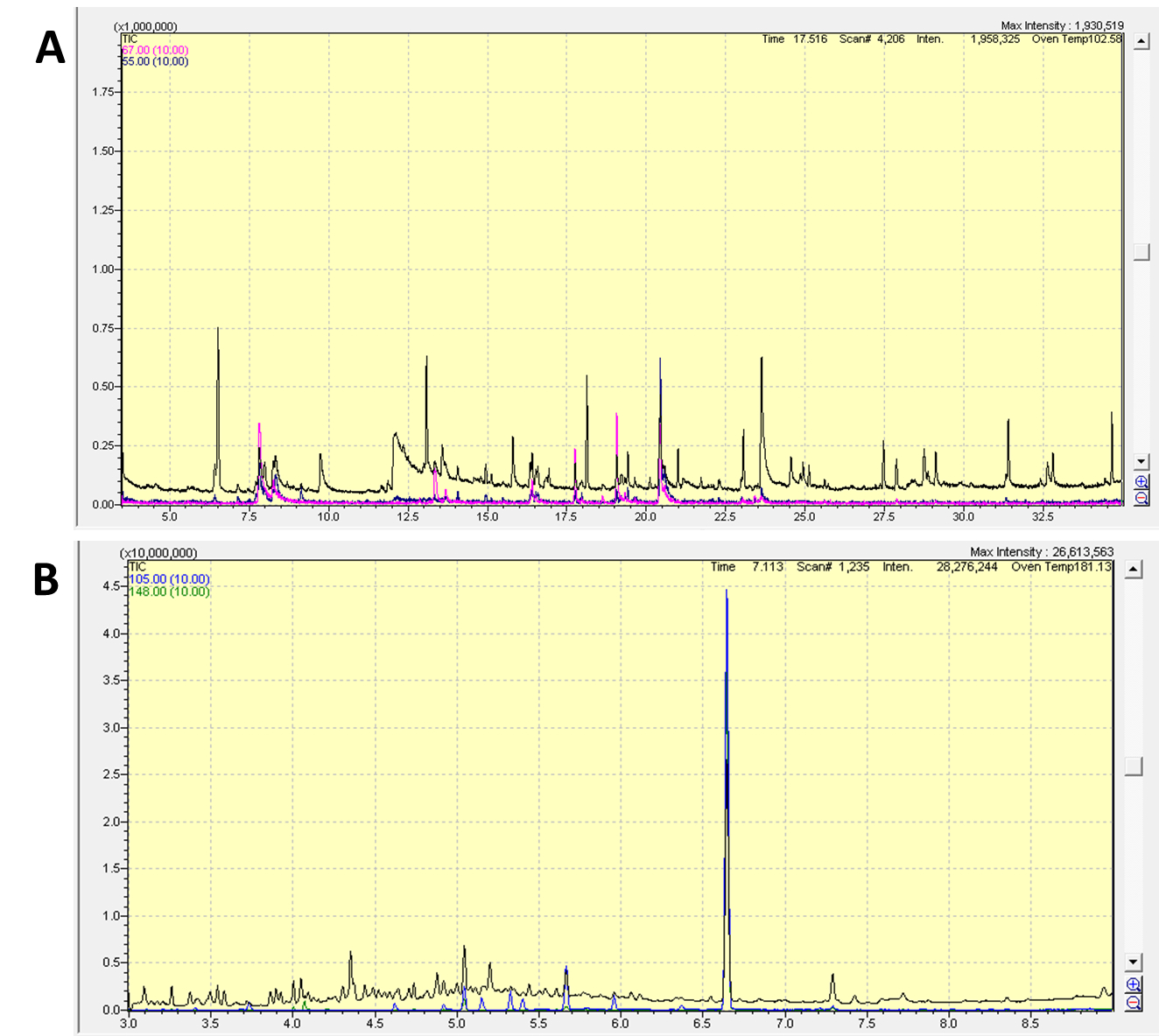
Figure 4. Example chromatograms of a headspace volatile collection from a N. attenuata leaf A and a flower B. The total ion chromatogram (TIC) and representative ion traces (A. m/z 55 and 67 to represent green leaf volatiles; B. m/z 105 and 148 to represent benzyl acetone) are shown. Peak identities can be found in Kallenbach et al. (2014).
Acknowledgments
The presented protocol was adopted from Kallenbach et al. (2014). This work was funded by the Max-Planck-Society and by Advanced Grant no. 293926 of the European Research Council.
References
- Dicke, M. and Baldwin, I. T. (2010). The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help’. Trends Plant Sci 15(3): 167-175.
- Holopainen, J. K. and Gershenzon, J. (2010). Multiple stress factors and the emission of plant VOCs. Trends Plant Sci 15(3): 176-184.
- Kallenbach, M., Oh, Y., Eilers, E. J., Veit, D., Baldwin, I. T. and Schuman, M. C. (2014). A robust, simple, high-throughput technique for time-resolved plant volatile analysis in field experiments. Plant J 78(6): 1060-1072.
- Seethapathy, S. and Gorecki, T. (2012). Applications of polydimethylsiloxane in analytical chemistry: a review. Anal Chim Acta 750: 48-62.
- van Pinxteren, M., Paschke, A. and Popp, P. (2010). Silicone rod and silicone tube sorptive extraction. J Chromatogr A 1217(16): 2589-2598.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Kallenbach, M., Veit, D., Eilers, E. J. and Schuman, M. C. (2015). Application of Silicone Tubing for Robust, Simple, High-throughput, and Time-resolved Analysis of Plant Volatiles in Field Experiments. Bio-protocol 5(3): e1391. DOI: 10.21769/BioProtoc.1391.
Category
Plant Science > Plant biochemistry > Other compound
Plant Science > Plant metabolism > Other compound
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Dear Mario,I am about to use your protocol to collect some BVOCs,...
Share
Bluesky
X
Copy link










