- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
VZV Replication Assays
Published: Vol 4, Iss 16, Aug 20, 2014 DOI: 10.21769/BioProtoc.1210 Views: 11841
Reviewed by: Pinchas TsukermanAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
In vitro Drug Susceptibility Assay for HBV Using Southern Blotting
Sung Hyun Ahn [...] Kyun-Hwan Kim
Apr 20, 2015 11349 Views
Isolation of Exosomes from Semen for in vitro Uptake and HIV-1 Infection Assays
Marisa N. Madison [...] Chioma M. Okeoma
Apr 5, 2017 12876 Views
Sleeping Beauty Transposon-based System for Rapid Generation of HBV-replicating Stable Cell Lines
Jin-Wei Zheng [...] Quan Yuan
Jul 5, 2018 9741 Views
Abstract
Varicella zoster virus (VZV) is a human herpesvirus which causes Varicella (chickenpox) upon primary infection and Zoster (shingles) following reactivation from latency (von Bokay, 1909). Whilst VZV is extensively studied, inherent features of VZV replication, such as cell-association of virus particles during in vitro culture and a restricted host range (limited to humans and some other primates) mean the cellular and viral mechanisms underlying VZV reactivation and pathogenesis remain largely uncharacterised. Much remains to be learnt about VZV, interactions with its host, and the development of disease. This protocol describes a basic VZV replication assay using a recombinant VZV-GFP reporter virus. As VZV is highly cell-associated in tissue culture, the reporter virus inoculum described here is a preparation of infected cells. This reporter virus-infected cell line can be used in combination with siRNA gene depletion or cDNA overexpression transfection protocols to determine the effect of individual cellular genes on virus replication.
Materials and Reagents
- VZV-permissive human cells (e.g. MeWo cells) (ATCC, catalog number: HTB-65 )
- Minimal essential medium eagle with Earle’s BBS, with L-glutamine (Lonza, catalog number: 12-611F )
- Fetal bovine serum (FBS) (LabTech, catalog number: FCS-SA-10454 )
- Penicillin: streptomycin (5,000 units/ml each) (Lonza, catalog number: DE17-603E )
- Non-essential amino acids (NEAA) (Life Technologies, catalog number: 11140035 )
- 1x Trypsin-EDTA liquid (0.05% Trypsin, 0.53 mM EDTA-4Na) (Life Technologies, catalog number: 25300096 )
- Phosphate buffered saline without Magnesium or Calcium (Lonza, catalog number: 17-516F )
- Recombinant VZV-GFP cell-associated virus stock (Zerboni, 2000)
- MeWo growth medium (see Recipes)
Equipment
- 75 cm2 filter cap tissue culture flasks (Sigma-Aldrich, catalog number: C7106-120EA )
- 96-well black tissue culture-treated plates (48/case) (VWR International, catalog number: 734-1609 )
- 50 ml centrifuge tube (e.g. Corning, catalog number: 430290 )
- Disposable hemocytometer (KOVA Glasstic slide 10 with counting grid) (HYCOR Biomedical, catalog number: 87144E )
- Fluorescent plate reader (e.g. POLARstar OPTIMA, BMG LABTECH)
- Class II Microbiological safety cabinet
- Humidified cell culture incubator (37 °C, 5% CO2)
- Pipette aid (Alpha laboratories, catalog number: 4-131-201-E )
- Centrifuge (e.g. Eppendorf, catalog number: 5811-000.010 )
- Light microscope
Procedure
- Take a semi-confluent (~50%) T75 flask of MeWo cells that has been split the previous day (see Note 1).
- Remove media, rinse gently in sterile phosphate buffered saline (PBS), and discard wash.
- Dislodge cells by adding 3 ml trypsin. Incubate for around 5 min at 37 °C or until cells are fully dislodged.
- Inactivate trypsin by adding 7 ml MeWo growth medium.
- Pipette up and down to create a single-cell suspension.
- Remove 12 μl and count cell density in a disposable hemocytometer.
- Dilute cells to a density of 2 x 105 cells/ml in MeWo growth medium and seed 100 μl per well in a black, flat-bottomed 96-well plate to give 2 x 104 cells/well (see Note 2).
- Leave cells to adhere overnight in a humidified incubator with 5% CO2 at 37 °C.
- Remove a vial of VZV-GFP of known titre from liquid nitrogen storage and thaw immediately in a water bath at 37 °C until just thawed (see Note 3).
- Transfer thawed cells to a 50 ml centrifuge tube and slowly add 30 ml growth medium (pre-warmed to 37 °C) drop by drop to gradually increase the temperature of the cells.
- Centrifuge for 10 min at 200 x g to pellet cells.
- Gently remove the entire washing medium and resuspend in 1 ml growth media.
- Dilute the cells in sufficient growth media to result in 1,000 IU/ml (see Note 4).
- Remove plates from the incubator, and remove media by inverting plate and shaking over a container of suitable disinfectant.
- Add 100 μl of the washed and resuspended VZV-GFP cell inoculum to each well to be infected, and 100 μl growth medium to uninfected control wells (see Note 5).
- Replace the lid on the plate and return the plate back to the incubator. Monitor virus replication as a measure of GFP fluorescence from ~22 h post-infection until replication plateaus (see Note 6).
- For comparison of virus replication in untreated and treated cells (protein overexpression, gene depletion, drug treatment etc.) calculate the replication slope over the linear growth phase and normalise treated cells to untreated (“normal” replication; Figure 1).
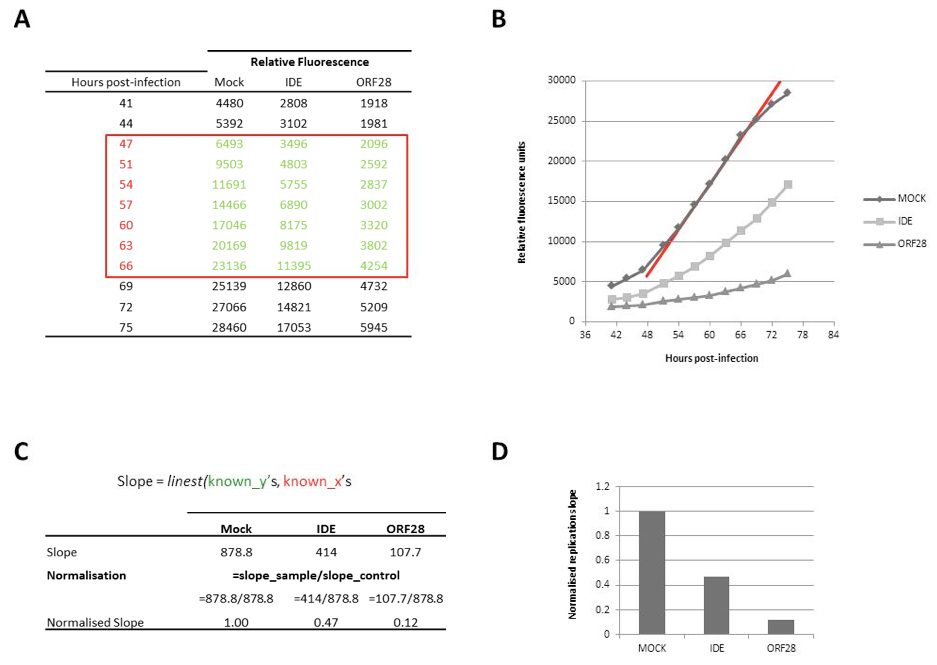
Figure 1. Calculation of replication slopes. A. Virus growth is monitored over multiple rounds of replication as a function of GFP fluorescence. B. Relative fluorescence units for mock transfected and positive controls-transfected (IDE, a cellular receptor for VZV; ORF28, the DNA polymerase for VZV) cells is plotted against hours post-infection to generate growth curve. C. The slope of replication over the linear phase of growth is calculated in Excel using the linest equation, and normalized to control (mock) transfected cells. D. Replication slopes are compared in a bar chart to see the effect of siRNA gene depletion on VZV replication.
Notes
- Virus replication proceeds more efficiently in cells that have been freshly passaged before seeding into assay plates.
- Using a black assay plate will help reduce background and crosstalk between wells.
- When grown in culture VZV virus particles remain associated with the cell membrane. As such, VZV-infected MeWo cells are used as the inoculum for VZV replication assays. These cells are stored in liquid nitrogen and need to be thawed and washed before use. Cells are extremely sensitive to temperature changes. When thawing it is essential that they are thawed quickly at 37 °C until only just thawed (1-2 min). Do not leave cells for longer periods of time as this will considerably reduce cell viability and therefore titre of replication-competent virus.
- It is important to do the infection with a known quantity of virus (multiplicity of infection; MOI) to generate growth curve over a reasonable time-scale. The titre [number of infectious units per ml of virus (IU/ml)] of VZV-GFP-infected MeWo cells is determined by a standard plaque assay, where a virus stock is added to a Mewo cell monolayer and overlaid with agarose. This results in the infection only of adjacent cells, which subsequently die to leave an empty patch within the cell monolayer. These patches, or ‘plaques’ can be counted to quantify the virus as ‘plaque-forming units’, or infectious units (IU), per ml inoculum. In our experience 100 IU per well of a 96-well plate produces a suitable growth curve for replication analyses and comparisons. For example, a virus stock with a titre of 6.6 x 104 IU/ml would contain 6,600 IU in 100 µl. A 1 ml aliquot of virus inoculum should therefore be resuspended in 66 ml media.
- Tissue culture plasticware, cells and growth medium all have some level of fluorescence. When utilising fluorescent reporter genes it is therefore essential to have appropriate controls to provide background fluorescence readings. For this assay, the fluorescence from mock-infected cells is used as background.
- Replication is monitored over regular intervals to enable a complete growth curve (fluorescence over time) to be plotted. When comparing VZV replication between untreated and treated samples we use the slope of replication over the linear growth phase. It is therefore important to have as many measurements over this phase as possible (with a minimum of 6 for statistical reliability of the slope calculation). A test replication assay with your own equipment will allow the time of linear growth to be established, and assay timings can be adjusted to ensure measurements can be taken over this period.
Recipes
- MeWo growth medium
Eagle’s minimum essential medium (EMEM)
5% FCS
1% p-s
1% NEAA
Acknowledgments
We are grateful to Prof. Ann Arvin (Stanford School of Medicine) for the kind gift of the VZV-GFP clone, and to Dr. Armin Baiker for the propagation of a cell-associated VZV-GFP virus stock. The protocol was optimised by Dr Lakshmi N. Kaza and Dr Samantha Griffiths. The authors gratefully acknowledge the MRC for funding (G0501453 J.H.).
References
- von Bokay, J. (1909). Uëber den aëtiologischen Zusammenhang der Varizellen mit gewissen Fällen von Herpes Zoster. Wien Klin Wochenschr 22: 1323-1326.
- Zerboni, L., Sommer, M., Ware, C. F. and Arvin, A. M. (2000). Varicella-zoster virus infection of a human CD4-positive T-cell line. Virology 270(2): 278-285.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Griffiths, S. J. and Haas, J. (2014). VZV Replication Assays. Bio-protocol 4(16): e1210. DOI: 10.21769/BioProtoc.1210.
Category
Microbiology > Microbe-host interactions > In vitro model > Cell line
Microbiology > Microbe-host interactions > Virus
Microbiology > Microbial cell biology > Cell isolation and culture
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link











