- EN - English
- CN - 中文
Mag-Net Strong Anion Exchange Enables Isolation of Ovarian Cancer Ascites Extracellular Vesicles for Proteomic Biomarker Discovery
Mag-Net 强阴离子交换技术用于分离卵巢癌腹水来源细胞外囊泡以开展蛋白质组学标志物发现
发布: 2026年03月20日第16卷第6期 DOI: 10.21769/BioProtoc.5639 浏览次数: 97
评审: Dipak Kumar PoriaAmbuja NavalkarShefali Singh

相关实验方案

适用于 LC–MS/MS 蛋白质组学分析的大体积细胞培养上清分泌组样品制备方法优化
Basil Baby Mattamana [...] Peter Allen Faull
2025年12月20日 1348 阅读
Abstract
Extracellular vesicles (EVs) are nanoscale particles secreted by all cells and present in all biological fluids, where they carry molecular cargo reflective of health and disease states. Their diagnostic potential is often obscured by the high abundance of non-EV proteins and lipoproteins (e.g., albumin, apolipoproteins) that complicate proteomic analysis of primary biofluids, such as ascites fluid. Conventional isolation strategies face a persistent trade-off between EV purity and yield. To overcome this, a magnetic bead-based protocol (Mag-Net) to enrich EVs according to electrochemical surface charge using strong anion-exchange chemistry (SAX) was adapted for proteomics. Our workflow is specifically adapted to ascites fluid from human or murine sources. This approach effectively separates EVs from high-abundance proteins and lipoproteins, enabling proteomic profiling from as little as 2 μL of ascites fluid. Demonstrated in both murine and human ovarian cancer models, Mag-Net offers a reproducible, scalable, and automation-ready solution for EV isolation from various biofluids.
Key features
• Extracellular vesicles (EVs) from murine and human ascites fluid are effectively enriched using Mag-Net beads.
• EVs are effectively captured and eluted from Mag-Net beads to support Raman spectroscopy, nanoparticle tracking analysis, and atomic force microscopy.
• EV isolation by Mag-Net provides robust proteomic depth obtained by mass spectrometry.
• Robust proteomic data can be obtained from input volumes ranging from 2 to 100 μL of ascites.
Keywords: Extracellular vesicles (细胞外囊泡)Graphical overview
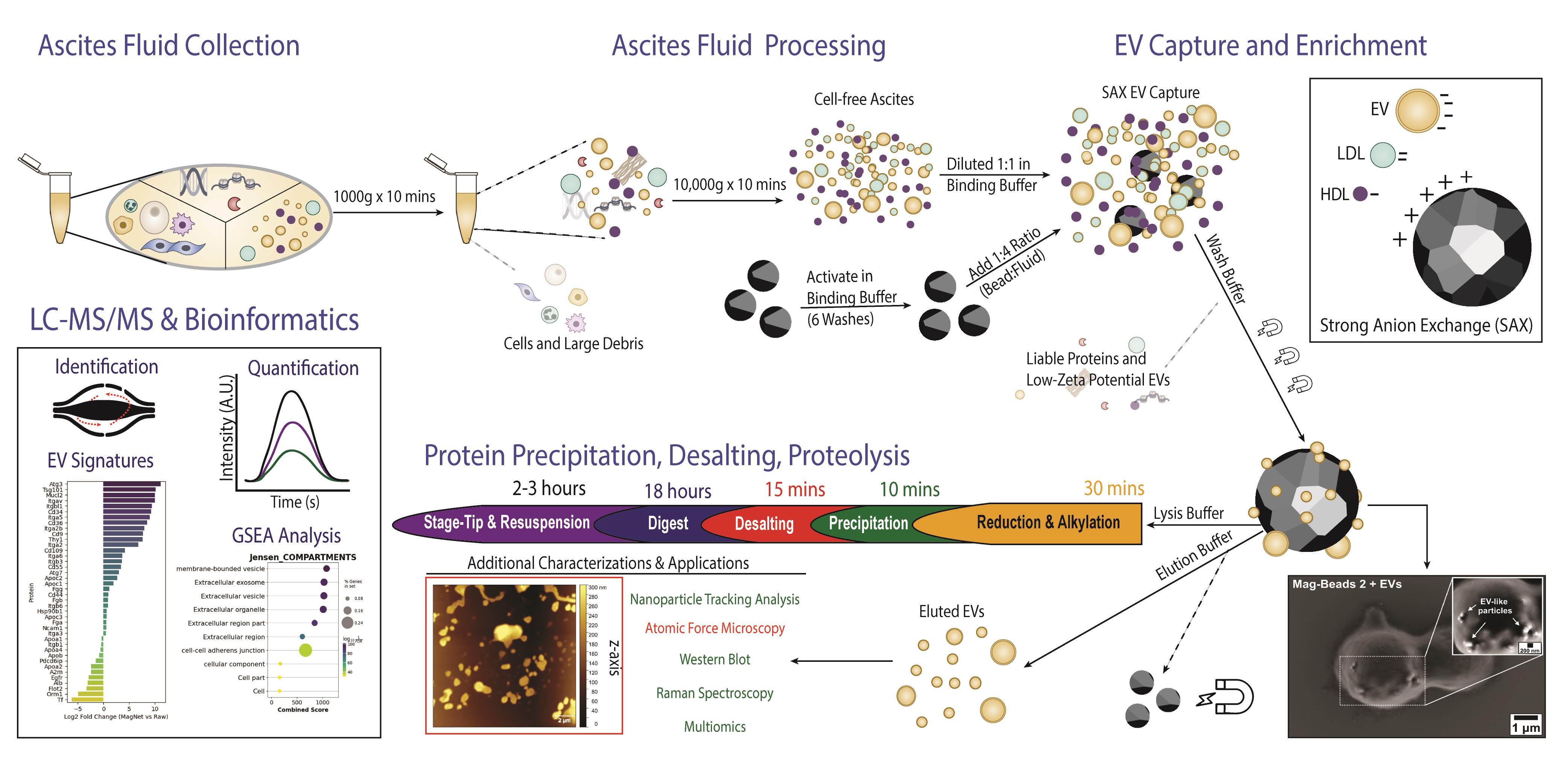
Isolation of extracellular vesicles from ascites fluid using magnetic beads functionalized with strong anion exchange. Ascites samples can be collected from human donors or murine models.
Background
Ascites fluid is a hallmark of several cancers [1], including ovarian cancer [2–4], and represents a valuable yet underutilized source of molecular information [5,6]. Given the absence of reliable biomarkers for early detection, comprehensive proteomic profiling of the tumor microenvironment can help uncover tumor-specific or tumor-associated proteins that reflect disease state and progression [7]. Similar to plasma, ascites is characterized by a high dynamic range of protein abundances. These are derived from diverse cellular sources [1,7,8], posing challenges for in-depth proteomic analysis without depletion of high-abundant species or enrichment of biological sub-compartments. Extracellular vesicles (EVs), which encapsulate molecular cargo representative of their cell of origin, provide a concentrated and disease-relevant snapshot of the tumor milieu [9–11]. Therefore, efficient and selective enrichment of EVs can augment the depth of omics technology and the ultimate success of biomarker discovery efforts [7,12–15].
To address the limitations of conventional EV isolation methods, such as ultracentrifugation (UC) or size-exclusion chromatography (SEC) [12,14–20], we employ a magnetic bead-based approach (Mag-Net) utilizing strong anion-exchange (SAX) chemistry [21]. Mag-Net beads enable scalable input volumes, effective depletion of high-abundance proteins and lipoproteins, and tunable buffer chemistry to exploit EV surface charge (zeta potential) for selective separation [22]. This charge-based isolation strategy is compatible with both manual and automated workflows, and its modular design allows integration with multiomic analyses [23]. The viscosity, lipid, and mucin content of ascites fluid challenge EV isolation [4]; thus, an efficient EV purification from this complex matrix will benefit clinical or diagnostic fields. We have shown that bead-bound EVs isolated from ascites can be divided to support both on-bead proteomic and off-bead EV characterization workflows [23]. Herein, we describe an adaptable protocol optimized for murine and human ascites fluid, enabling efficient EV isolation and proteomic analysis from starting volumes ranging between 2 and 100 μL of input material.
Materials and reagents
Biological materials
1. Murine ascites fluid (ID8 model of ovarian cancer, collected in-house)
2. Human ascites fluid (epithelial ovarian cancer, collected in-house)
Reagents
1. MagReSynTM SAX magnetic beads (ReSyn, catalog number: MR-SAX002)
2. LC–MS-grade water (Fisher Scientific, CAS: 7732-18-5)
3. LC–MS-grade acetonitrile (Fisher Scientific, CAS: 75-05-8)
4. Tris-Bis propane (Sigma-Aldrich, CAS: 64431-96-5)
5. Sodium chloride (NaCl) (Sigma-Aldrich, CAS: 7647-14-5)
6. Urea (Sigma-Aldrich, CAS: 15-37-6)
7. Tris (Sigma-Aldrich, CAS: 77-86-1)
8. N-40 (ThermoFisher, CAS: 9016-45-9)
9. 10 N HCl (Fisher Scientific, CAS: 7647-01-0)
10. Trifluoroacetic acid (TFA) (Sigma-Aldrich, CAS: 76-05-1)
11. Tween-20 (Sigma-Aldrich, CAS: 9005-64-5)
12. n-Dodecyl β-D-maltoside (Sigma-Aldrich, CAS: 69227-93-6)
13. SDS pellets (Sigma-Aldrich, CAS: 151-21-3)
14. TCEP (Thermo Fisher, catalog number: T2556)
15. IAA (Bio-Rad, catalog number: 1632109)
16. Ammonium bicarbonate (Sigma-Aldrich, catalog number: 1066-33-7)
17. Sequencing-grade trypsin (Promega, catalog number: V5113)
18. Sequencing-grade LysC (FujiFilm Bioscience, catalog number: 125-05061)
19. LC–MS-grade 0.1% in water (Fisher Scientific, catalog number: LS1184)
20. UPLC-grade 0.1% in acetonitrile (Fisher Scientific, catalog number: LS1201)
21. C18 Disks (Empore, catalog number: 66883-U)
22. BCA Peptide Quantification kit (Thermo Fisher, catalog number: 23225)
Solutions
1. Binding buffer (BB) (see Recipes)
2. Wash buffer (WB) (see Recipes)
3. Elution buffer (EB) (see Recipes)
4. 1% DDM stock (see Recipes)
5. Digestion buffer (DB) (see Recipes)
6. Peptide resuspension solution (see Recipes)
Recipes
1. Binding buffer (BB), pH 6.3
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Deionized water (LC–MS grade) | n/a | 50 mL |
| Bis-Tris propane | 100 mM | 1.412 g |
| NaCl | 150 mM | 439 mg |
| 5 N HCl* | to pH 6.3 | Estimated 700–750 μL** |
| Total | n/a | 50 mL |
*Prepared from 10 N stock. Decreased molarity of HCl prevents polymer leeching from plastics.
**It is important to add in a drop-wise fashion or small volumes (<100 μL). Buffer is basic (>9.5) without titration of HCl.
Note: Store at room temperature and use within 2 days.
2. Wash buffer (WB), pH 6.3
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Deionized water (LC–MS grade) | n/a | 50 mL |
| Bis-Tris propane | 50 mM | 0.706 g |
| NaCl | 150 mM | 439 mg |
| 5 N HCl* | to pH 6.3 | Estimated 200–300 μL** |
| Total | n/a | 50 mL |
*Prepared from 10 N stock. Decreased molarity of HCl prevents polymer leeching from plastics.
**It is important to add in a drop-wise fashion or small volumes (<100 μL). Buffer is basic (>9.5) without titration of HCl.
Note: Store at room temperature and use within 2 days.
3. Elution buffer (EB), pH 6.3
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Deionized water (LC–MS grade) | n/a | 45 mL |
| Bis-Tris propane | 50 mM | 0.706 g |
| NaCl | 1 M | 2.92 g |
| Tween-20 (1%) | 0.1% | 5 mL |
| 5 N HCl* | to pH 6.3 | Estimated 200–300 μL** |
| Total | n/a | 50 mL |
*Prepared from 10 N stock. Decreased molarity of HCl prevents polymer leeching from plastics.
**It is important to add in a drop-wise fashion or small volumes (<100 μL). Buffer is basic (>9.5) without titration of HCl.
Note: Store at room temperature and use within 2 days.
4. 1% DDM stock
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| n-Dodecyl β-D-maltoside stock | n/a | 100 mg |
| Deionized water (LC–MS grade) | n/a | 10 mL |
| Total | 1% (w/v) | 10 mL |
Can be aliquoted and stored at -20 °C for 2 weeks.
5. Digestion buffer (DB), pH 8.0
| Reagent | Final concentration | Quantity or volume |
|---|---|---|
| Deionized water (LC–MS grade) | n/a | 9.8 mL |
| Ammonium bicarbonate | 50 mM | 39.5 mg |
| 1% DDM stock | 0.02% (w/v) | 200 μL |
| Total | n/a | 10 mL |
Note: Store at 4 °C and use within 2 days.
Laboratory supplies
1. 10 μL low-bind pipette tips (Avantor, catalog number: 76322-528)
2. 200 μL low-bind pipette tips (Avantor, catalog number: 76322-150)
3. 1,000 μL low-bind pipette tips (Avantor, catalog number: 76322-154)
4. 2.0 mL round-bottom Eppendorf tubes (Eppendorf, catalog number: 0030108132)
5. 50 mL Falcon tubes (Avantor, catalog number: 525-0610)
6. 15 mL Falcon tubes (Avantor, catalog number: 525-0604)
7. 10 mL stereological pipettes (Corning, catalog number: 4488)
8. 500 mL sterile glass container (Pyrex, Milipore Sigma, catalog number: CLS1000600-6EA)
Equipment
1. Analytical scale (Metteler-Toledo, model: MA204, Fisher Scientific, catalog number: 01-804-204)
2. Benchtop vortex (OHAS, model: Votrex Genie 2, Fisher Scientific, catalog number: 1-804-423)
3. pH meter (Mettler-Toledo, model: FiveEasy F20, Fisher Scientific, catalog number: 01-912-346)
4. -20 °C freezer
5. -80 °C freezer
6. 4 °C refrigerator
7. EppendorfTM ThermoMixerTM C (Eppendorf, catalog number: EP5382000023)
8. Magnetic rack (Luna Nanotech, catalog number: MGR-016)
9. UPLC- Vanquish NeoTM (Thermo Fisher)
10. Mass spectrometer EclipseTM Tribrid Orbitrap (Thermo Fisher)
11. Scanning electron microscopy (Zeiss, model: LEO 1530)
12. Nanoparticle tracking analysis (Particle Metrix, Zetaview)
13. Atomic force microscopy (Bruker, BioScope Catalyst with NCLR-50 Cantilever)
Software and datasets
1. DIA-NN (v1.9)
2. MSCovert (v3.0)
3. Gwyiddon Nanoscope (v8.5)
4. ZetaView Analyze (v1.4)
5. Zeiss SmartSEM (v6.0)
Procedure
文章信息
稿件历史记录
提交日期: Dec 16, 2025
接收日期: Feb 1, 2026
在线发布日期: Mar 5, 2026
出版日期: Mar 20, 2026
版权信息
© 2026 The Author(s); This is an open access article under the CC BY license (https://creativecommons.org/licenses/by/4.0/).
如何引用
Cooper, T. T. (2026). Mag-Net Strong Anion Exchange Enables Isolation of Ovarian Cancer Ascites Extracellular Vesicles for Proteomic Biomarker Discovery. Bio-protocol 16(6): e5639. DOI: 10.21769/BioProtoc.5639.
分类
癌症生物学 > 通用技术 > 生物化学试验
生物化学 > 蛋白质 > 分离和纯化
系统生物学 > 蛋白质组学 > 分泌蛋白质组
您对这篇实验方法有问题吗?
在此处发布您的问题,我们将邀请本文作者来回答。同时,我们会将您的问题发布到Bio-protocol Exchange,以便寻求社区成员的帮助。
Share
Bluesky
X
Copy link