- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Protease Activity Assay in Fly Intestines
Published: Vol 7, Iss 18, Sep 20, 2017 DOI: 10.21769/BioProtoc.2560 Views: 9618
Reviewed by: Jihyun KimTzvetina BrumbarovaAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

Enzymatic Assays and Enzyme Histochemistry of Tuta absoluta Feeding on Tomato Leaves
Rim Hamza [...] Luis A. Cañas
Sep 5, 2018 7627 Views
In situ Dephosphorylation Assay with Recombinant Nil Phosphatase
Nilay Nandi [...] Helmut Krämer
Sep 20, 2022 2151 Views
Detecting the Activation of Endogenous Small GTPases via Fluorescent Signals Utilizing a Split mNeonGreen: Small GTPase ActIvitY ANalyzing (SAIYAN) System
Miharu Maeda and Kota Saito
Jan 5, 2026 496 Views
Abstract
The intestine is a central organ required for the digestion of food, the absorption of nutrients and for fighting against aggressors ingested along with the food. Impairment of gut physiology following mucosal damages impacts its digestive capacities that consequently will affect growth, wellbeing or even survival of the individual. Hence, the assessment of intestinal functions encompasses, among others, the monitoring of its integrity, its cellular renewing, its immune defenses, the production of enteroendocrine hormones and its digestive capacities. Here, we describe in detail how to assess the activity of the proteases secreted in the intestinal lumen of adult Drosophila melanogaster flies. This method can also be used for larval intestines. The present protocol is adapted and improved from the Sigma-Aldrich’s protocol proposed in the ‘Protease Fluorescent Detection Kit’ (Product code PF0100).
Keywords: Drosophila melanogasterBackground
The intestine is subjected to many stresses such as feasting, fasting, chemicals, pathogens, injuries etc. The gut is able to overcome such stresses by maintaining its physiological equilibrium named homeostasis. To perceive the incoming stress and to yield an adapted answer to maintain gut functions, the intestine has developed robust and conserved mechanisms such as local innate immune defenses and tissue regeneration (Royet and Charroux, 2013; Bonfini et al., 2016). However, the maintenance of gut homeostasis can be compromised in certain cases. For example, during aging, there is an overall decline in tissue homeostasis maintenance with the presence of numerous immature or misdifferentiated cells (Jasper, 2015; Hu and Jasper, 2017). Another case where homeostasis can also be disrupted is upon exposure to xenobiotic or pathogens (such as opportunistic bacteria) that damage or kill cells impairing their functions (Bonfini et al., 2016). Hence, during the above cited examples, the digestive capacities of the gut are reduced. Moreover, during the process of tissue regeneration itself that produces many precursor cells the digestive capacities are also reduced (Loudhaief et al., 2017). Therefore, the assessment of the digestive capacities of the gut are of prime importance to evaluate the potential impact that can have an aggression on the gut physiology. Importantly, gut digestive function disruption may have both local and systemic metabolic consequences that will affect growth, immune defenses, reproduction, wellbeing, longevity…. Dietary proteins are essential for many (if not all) physiological functions (Soultoukis and Partridge, 2016). Imbalanced amino-acid absorption by the intestine can have dramatic consequences on growth for example. Protein digestion being essential to generate absorbable amino-acids by the enterocytes, the measurement of luminal protease activity appears a good readout to evaluate the physiological state of the intestine and its capacity to fulfill its digestive functions.
Materials and Reagents
- Drosophila rearing
- 6oz Drosophila stock bottles (Genesee Scientific, catalog number: 32-130 )
- Cotton balls for stock bottles (Genesee Scientific, catalog number: 51-102B )
- CantonS flies (Bloomington Drosophila Stock Center, catalog number: 64349 ) (flystocks.bio.indiana.edu)
- Agar (VWR, BDH®, catalog number: 20768-361 )
- Sugar (Carrefour or any other supermarket)
- Cornflour (AB, Celnat - NaturDis)
- Yeast (Biospringer, catalog number: BA10/0-PW )
- Tegosept (Apex, Fly Food preservative, Genesee Scientific, catalog number: 20-258 )
- Standard nutrient medium for Drosophila (see Recipes)
- 6oz Drosophila stock bottles (Genesee Scientific, catalog number: 32-130 )
- Bacterial culture
- Petri dishes
- Sterile tip
- 15 ml tubes (Corning, Falcon®, catalog number: 352096 )
- Graduated test tube
- Bacillus thuringiensis var. kurstaki (Btk) strain identified under the code 4D22 at the Bacillus Genetic Stock Center (http://www.bgsc.org/) and described by (Gonzalez et al., 1982)
- Erwinia carotovora carotovora (Ecc) was kindly provided by Bruno Lemaitre’s laboratory (École Polytechnique Fédérale, Lausanne, Switzerland)
- Escherichia coli (Ec) (One ShotTM TOP10 Chemically Competent E. coli) (Thermo Fisher Scientific, InvitrogenTM, catalog number: C404003 )
- Luria broth powder (Conda, catalog number: 1551 )
- Agar bacteriological (Euromedex, catalog number: 1330 )
- LB medium (see Recipes)
- LB-agar medium (see Recipes)
- Intoxication
- Cotton balls for narrow vials 25 mm (Genesee Scientific, catalog number: 51-101 )
- Spectrophotometry cuvettes (Ratiolab, catalog number: 2712120 )
- 2 ml microtubes (Paul Bottger, catalog number: 02-043 )
- 20 mm filter disks (Chromatography paper 3MM Chr) (GE Healthcare, catalog number: 3030-917 )
- 50 ml tube
- Drosophila narrow vials 25 mm (Genesee Scientific, catalog number: 32-109RL )
- Sucrose (Euromedex, catalog number: 200-301-B )
- 10% sucrose (see Recipes)
- Dissection
- 1.5 ml microtubes (Paul Bottger, catalog number: 02-063 )
- Graduated test tube
- Watch glass (Steriplan Petri dishes, DWK Life Sciences, catalog number: 237554008 )
- Ice
- Ethanol 70% (VWR, catalog number: 83801.360 )
- 10x PBS (Euromedex, catalog number: ET330 )
- 1x phosphate-buffered saline (PBS) (see Recipes)
- 1.5 ml microtubes (Paul Bottger, catalog number: 02-063 )
- Sample preparation
- Microtube pestle 1.5 ml (Argos Technologies, catalog number: P7339-901 )
- 0.5 ml microtubes (Paul Bottger, catalog number: 02-053 )
- 1x phosphate-buffered saline (PBS) (see Recipes)
- Assay
- 1.5 ml microtubes (Paul Bottger, catalog number: 02-063 )
- 96-well black microplates (Greiner Bio One International, catalog number: 655076 )
- 2 ml microtube
- 15 ml tube
- Aluminum foil
- Trypsin from bovine pancreas (Sigma-Aldrich, catalog number: T1005 )
- 1 mM HCl
- Casein Fluorescein IsoThioCyanate from bovine milk (Sigma-Aldrich, catalog number: C0528 )
- Distilled water
- cOmplete tablets EDTA-free (Roche Diagnostics, catalog number: 04693132001 )
- Trichloroacetic acid (TCA) (Sigma-Aldrich, catalog number: T6399 )
- Tris base
- Trypsin solution (see Recipes)
- Casein-FITC (see Recipes)
- 25x cOmplete (see Recipes)
- 10% TCA (see Recipes)
- 0.5 M Tris/HCl pH 8.5 (see Recipes)
Equipment
- Drosophila rearing
Refrigerated oven at constant temperature of 25 °C and with a 12 h/12 h light/dark cycle (Fisher Scientific, catalog number: 11857552). Humidity has to be maintained between 40% and 70%
Manufacturer: LMS, model: Model 240 . - Bacterial culture
- 100 ml flasks (Fisher Scientific, catalog number: 15409103 )
- 30 °C/37 °C shaking incubator (Infors, model: AK 82 )
- Intoxication
- Spectrophotometer (Aqualabo, Secomam, model: Prim Light & Aduanced)
- Droso-sleeper (Inject-Matic)
- Dissection
- Stereomicroscope (Leica Microsystems, model: Leica M60 )
- Dumont forceps #5 (Fine Science Tools, catalog numbers: 11251-20 and 11252-20 )
- Sample preparation
- Pestle motor (Heidolph Instruments, model: RZR 2100 )
- Refrigerated microfuge (Eppendorf, model: 5430 R )
- Assay
- Vortex (Scientific Industries, model: Vortex-Genie 2 )
- Automated pipette (Eppendorf, model: Multipette® plus)
- 37 °C solid-door incubator (Jouan)
- Fluorimeter (Agilent Technologies, model: Cary Eclipse )
Software
- Kyplot
- Excel
Procedure
- Drosophila rearing
- CantonS flies are reared on standard medium for Drosophila melanogaster (see Recipes) at 25 °C.
- Only virgin five-days-old females are used in our experiments. To obtain synchronized five-day-old females, we empty bottles from adult flies, then we wait for the emergence of new flies from pupae. Among the emergent flies, males are rapidly discarded and females are transferred in new bottles for five days at 25 °C.
- CantonS flies are reared on standard medium for Drosophila melanogaster (see Recipes) at 25 °C.
- Bacterial culture
In the experiments presented below, we use three different bacterial strains (Btk, Ecc and Ec, see Materials and Reagents) and water as negative control (Ctrl). Spread bacteria from the stocks on LB agar Petri dishes and let grow overnight at the required temperature (30 °C for 4D22 and Ecc and 37 °C for Ec).- Pick a single colony using a sterile tip and initiate a 10 ml starter culture of LB (see Recipes) in a 15 ml tube. Let grow for 8 h shaking (220 rpm) at 30 °C for 4D22 and Ecc and at 37 °C for Ec.
- Next, use the small starter culture to inoculate a second, larger culture: 50 µl of starter culture + 50 ml LB in a 100 ml flask and allow to grow overnight at the required temperature.
- Pick a single colony using a sterile tip and initiate a 10 ml starter culture of LB (see Recipes) in a 15 ml tube. Let grow for 8 h shaking (220 rpm) at 30 °C for 4D22 and Ecc and at 37 °C for Ec.
- Intoxication
- On the day of infection, measure the optic density (OD) of the culture at a dilution of 1/5 (200 µl culture + 800 µl LB). A reliable OD must be between 0.2 and 0.8.
- Depending on the cultured bacteria, the required ODs to intoxicate flies with 106 Colony Forming Unit (CFU) per Drosophila are presented in the Table 1:
Table 1. Required bacterial concentrations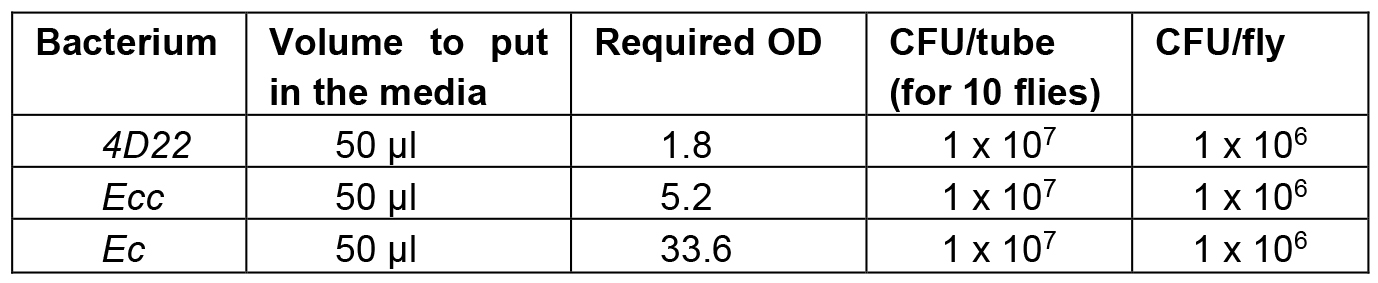
- Dilute the overnight bacterial culture with LB to obtain the required OD. For example: to obtain an OD 1.8 of 4D22 bacteria from an overnight culture at OD 9, you need to dilute the overnight culture by 5 (9/1.8 = 5). Use LB medium to make the dilution.
- Then mix at 1:1 the diluted culture with 10% sucrose (see Recipes). For practical reasons mix 1 ml of diluted culture with 1 ml of 10% sucrose. This mixture will constitute the intoxication solution.
- 5-day-old virgin females are allowed to fast for 2 h at 25 °C: flies are placed in empty Drosophila bottles at 25 °C for 2 h.
- Starved flies are then transferred onto a Drosophila narrow vials containing fly medium covered with filter disks soaked with 50 µl of the intoxication solution (Figure 1).
Note: Before to deposit your flies onto the soaked filter disk in the vials, wait for a few minutes until the filter disk becomes dry, otherwise the flies will be wet and will probably remain stuck on the filter disk. - Flies are kept to feed on the contaminated media at 25 °C until dissection.
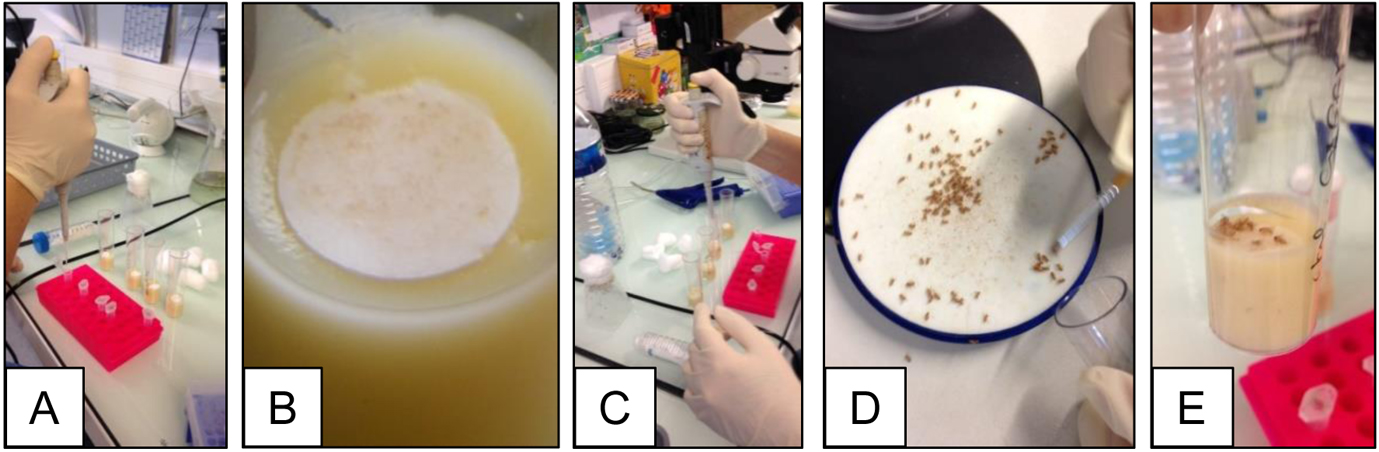
Figure1. Intoxication procedure. A. Annotate the Drosophila vials and place the filter disks inside on the top of the medium; B. Zoom up on a filter disk on the top of the medium; C. Add 50 µl of the intoxication solution in the vials; D. Sort by 10 the starved female flies using the Droso-sleeper; E. Deposit carefully 10 flies in each contaminated vial.
- On the day of infection, measure the optic density (OD) of the culture at a dilution of 1/5 (200 µl culture + 800 µl LB). A reliable OD must be between 0.2 and 0.8.
- Dissection
- Forceps and dissecting watch glasses have to be washed with a 70% ethanol solution.
- The flies (10 virgin females per condition) are anesthetized using the Droso-sleeper.
- Place one fly in a watch glass pre-filled with 1 ml 1x PBS (see Recipes) (Figure 2A).
- Using forceps, hold the fly by the head and pull gently on the posterior part of the abdomen to carefully detach the abdomen from the thorax (Figure 2B).
- Then, the intestine is carefully stretched (Figure 2C).
- Cut the head and untie very gently the intestine first from its anterior part and then from its posterior part (Figures 2D-2F).
Note: Here, we need only the midgut so we cut the foregut and the hindgut. We also removed Malpighian tubules that can still be attached at the midgut/hindgut boundary (Figures 2D-2F).
Figure 2. Dissection procedure. A. Place one fly in a watch glass pre-filled with 1 ml 1x PBS under a stereomicroscope; B. Hold the fly by the head and pull gently on the posterior part of the abdomen to carefully detach the abdomen from the thorax; C. Carefully stretch the intestine; D. Cut the head and untie very gently the intestine first from its anterior part and then from its posterior part. The whole intestine (foregut/midgut/hindgut) must be separated from the rest of the abdomen. E. Remove the crop; F. Remove the proventriculus, the hindgut and the Malpighian tubules. See also http://www.bio-protocol.org/e2079 (Chen et al., 2016) - The dissected midguts are then immediately placed in 1x PBS on ice waiting for the dissection of the other intestines.
- Only 8 guts (over the 10) are transferred into 50 µl of 1x PBS in a 1.5 ml microtube for sample preparation.
- Forceps and dissecting watch glasses have to be washed with a 70% ethanol solution.
- Sample preparation
- Recover gut content by pressing them with a microtube pestle fixed on a motor set to 1,000 rpm: put the motor on and perform 10 up and down movements with the microtube to allow the pestle to reach the bottom of the microtube (Video 1).
Note: Keep the tubes on ice to avoid any protein degradation. The goal is to recover most of lumen contents, so do not strongly crush the guts and do not perform more than 10 up and down movements.
Video 1. Crushing intestines. The video describes the procedure to gently crush intestines using a microtube pestle. Note at the end of the video that to avoid to waste gut content, we take off the pestle and press it against the edge of the microtube - Centrifuge for 5 min at 10,000 x g and 4 °C.
- Dilute at 1:10 a part of the supernatant: take 20 µl of the supernatant and transfer it in a 1.5 ml microtube containing 180 µl of 1x PBS.
- Take another 20 µl of the supernatant in a 0.5 ml microtube and place it at 4 °C waiting for protein assay if one wants to normalize the results per mg of protein.
- Recover gut content by pressing them with a microtube pestle fixed on a motor set to 1,000 rpm: put the motor on and perform 10 up and down movements with the microtube to allow the pestle to reach the bottom of the microtube (Video 1).
- Assay
Note: For each sample, we assay 3 volumes to be sure to be in the fluorimeter’s measurement range and to have reproducible results. As negative control, we use the maximal sample volume (i.e., 20 µl) complemented with a cocktail of protease inhibitors (25x cOmplete). As positive control, we use trypsin. The Table 2 gives an example of a 96-well plate’s scheme according to these rules. This is a plane of an independent experiment with two technical replicates (one from columns 1 to 4 lanes B to E and one from columns 6 to 9 lanes B to E).
Table 2. 96-well plate’s plane. Volume (µl) for: 10x PBS/Distilled water/Sample/Trypsin/25x cOmplete.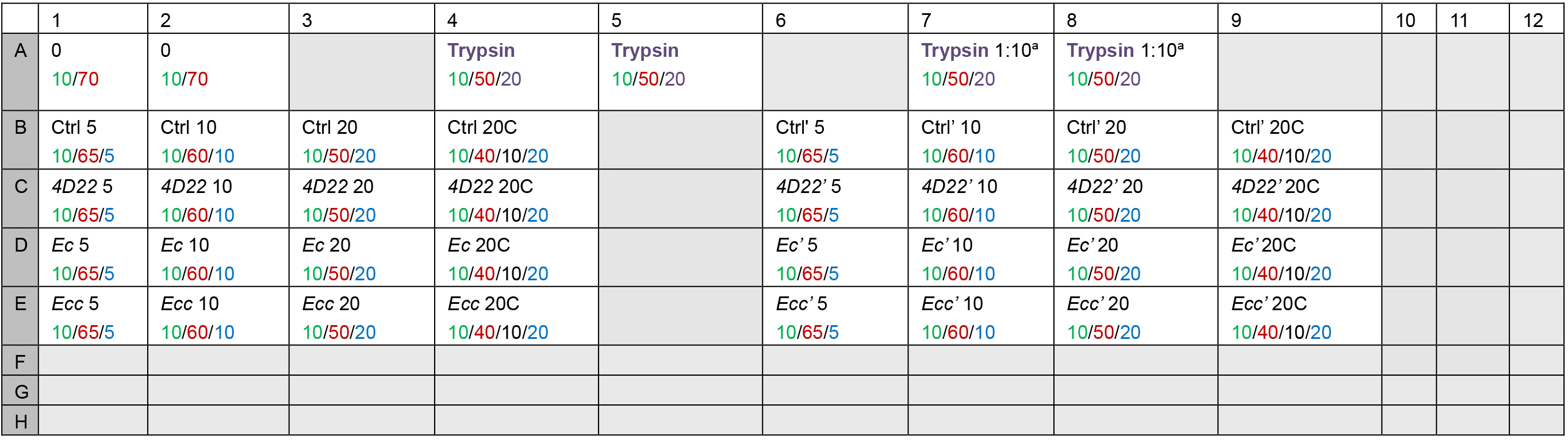
aTo be sure to stay in the reading range of the spectrophotometer, carry out two assays with a dilution of trypsin to 1:1- Prepare the trypsin solution (see Recipes) as described below.
- Annotate 1.5 ml microtubes with the letter and the number corresponding to their position in the 96-well plate.
- Add the indicated volume of distilled water in each tube.
- Add the 10x PBS.
- Finally add the indicated volume of sample or trypsin.
- When necessary add the protease inhibitors (25x cOmplete) (see Recipes).
Note: From this point, it is important to work with the least of light as possible. - Prepare the Casein-FITC (see Recipes) as described below.
- Add 20 µl Casein-FITC in each tube and vortex few seconds.
Note: Be careful, do not vortex too strongly to avoid fluorescence background. - Incubate for 1.5 h in a 37 °C solid-door incubator (samples have to be in the dark).
Note: Samples can be incubated until 24 h to increase the signal. Beyond one day, Casein-FITC can be degraded leading to high fluorescence background. - Add 300 µl 10% TCA (see Recipes) and vortex for a few seconds.
- Incubate for 0.5 h in a 37 °C solid-door incubator.
- Centrifuge for 10 min at 10,000 x g, 4 °C.
Note: Whole proteins and big fragments, which underwent a few or no proteolytic cleavage, precipitate with the TCA and are centrifuged down in the pellet. Casein-FITC small fragments that underwent a lot of proteolytic cleavage are in the supernatant. That’s what will be measured. - In a black 96-well plate, put 50 µl of supernatant according to the Table 2.
- Add 150 µl 0.5 M Tris/HCl (see Recipes) pH 8.5.
- Read the fluorescence on a spectrofluorimeter set on 485 nm excitation wavelength and 535 nm emission wavelength.
- Prepare the trypsin solution (see Recipes) as described below.
Data analysis
- FITC-intensity measurement analysis
- We systematically perform at least 3 independent experiments. We defined ‘independent experiments’ when they are performed on different days.
- Each experiment is performed in duplicate meaning that for a given day for one condition of intoxication, there are two different pool of 10 Drosophila contaminated by the same batch bacteria.
- Below we present results for 4 conditions of intoxication (Ctrl, 4D22, Ecc and Ec). For each condition there are 3 independent experiments and for each experiment two replicates.
- For each replicate, we have assayed 3 different volumes and a negative control (5 µl, 10 µl or 20 µl of samples and 20 µl of samples + 25x cOmplete).
- In each replicate, 8 intestines were used, therefore we have in total 48 intestines/each condition.
- We have calculated the mean of results obtained in the 3 independent experiments (i.e., six replicates) for each condition and for each volume.
- Table 3 and the Figure 3 give an example of raw data obtained after FITC fluorescence measurement.
Note: Below are presented all the raw data without excluding any value. However, the experimenter can remove some replicates if he has performed more than 3 independent experiments and if he judges that one of the replicates failed.
Table 3. FITC intensity measurements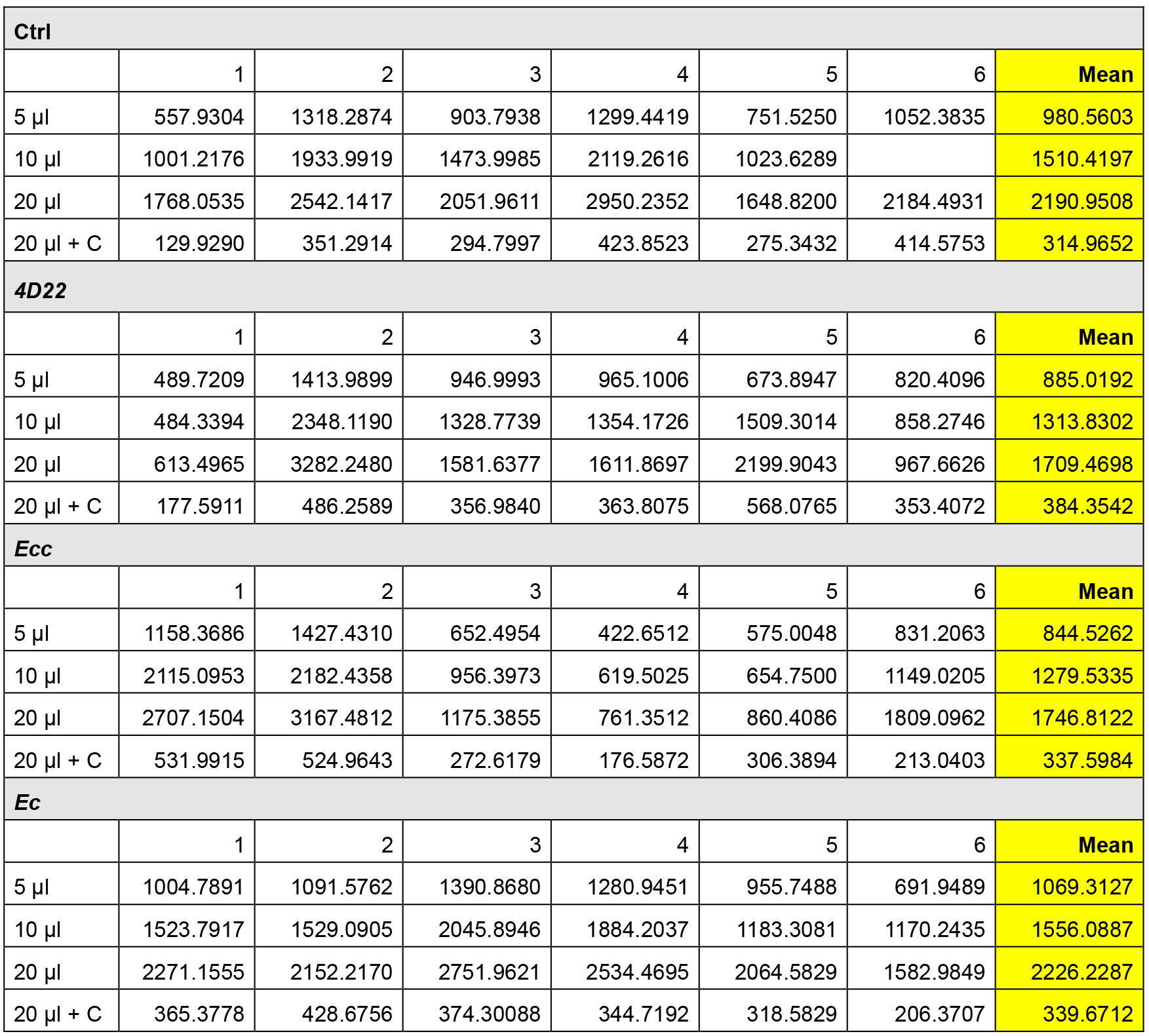
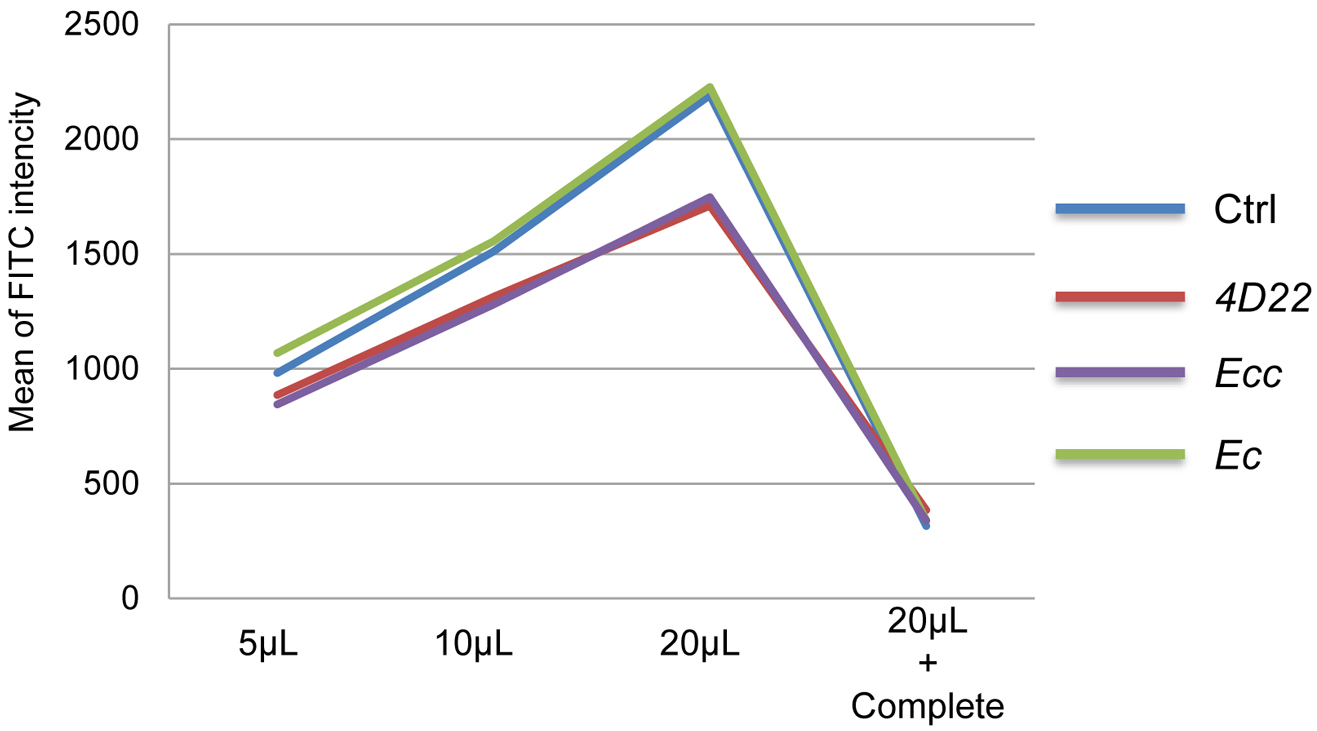
Figure 3. Graph of average FITC-intensity. The data of the Table 3 are reported in this graph. Only the means were used to draw the graph. - For result presentation and statistical analyses, we selected the data obtained with the volume 20 µl of samples (more reliable and reproducible).
- Results are presented in % of FITC intensity (reflecting protease activity) where Ctrl is considered at 100% (Table 4).
- We used SEM (Standard Error of the Mean) to present error bars in the graph (Figure 4).A. FITC-intensity measurement analysis
Table 4. Mean and percentage of FITC-intensity reflecting the protease activity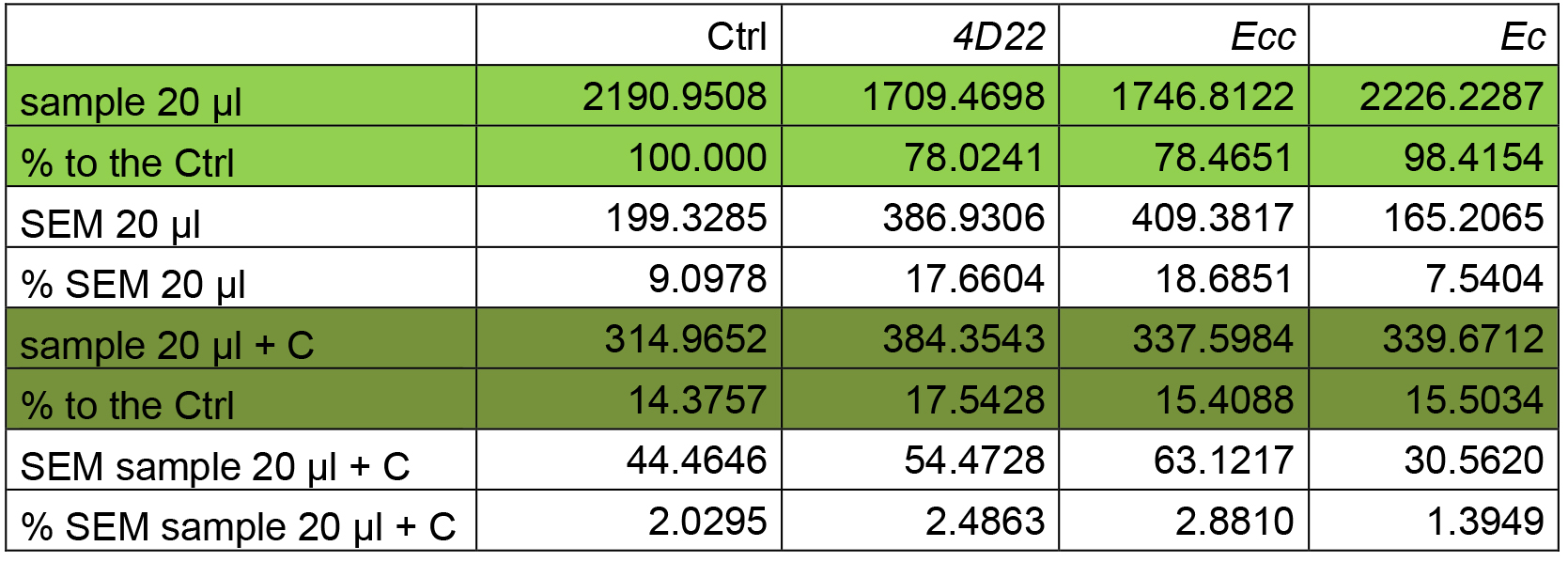
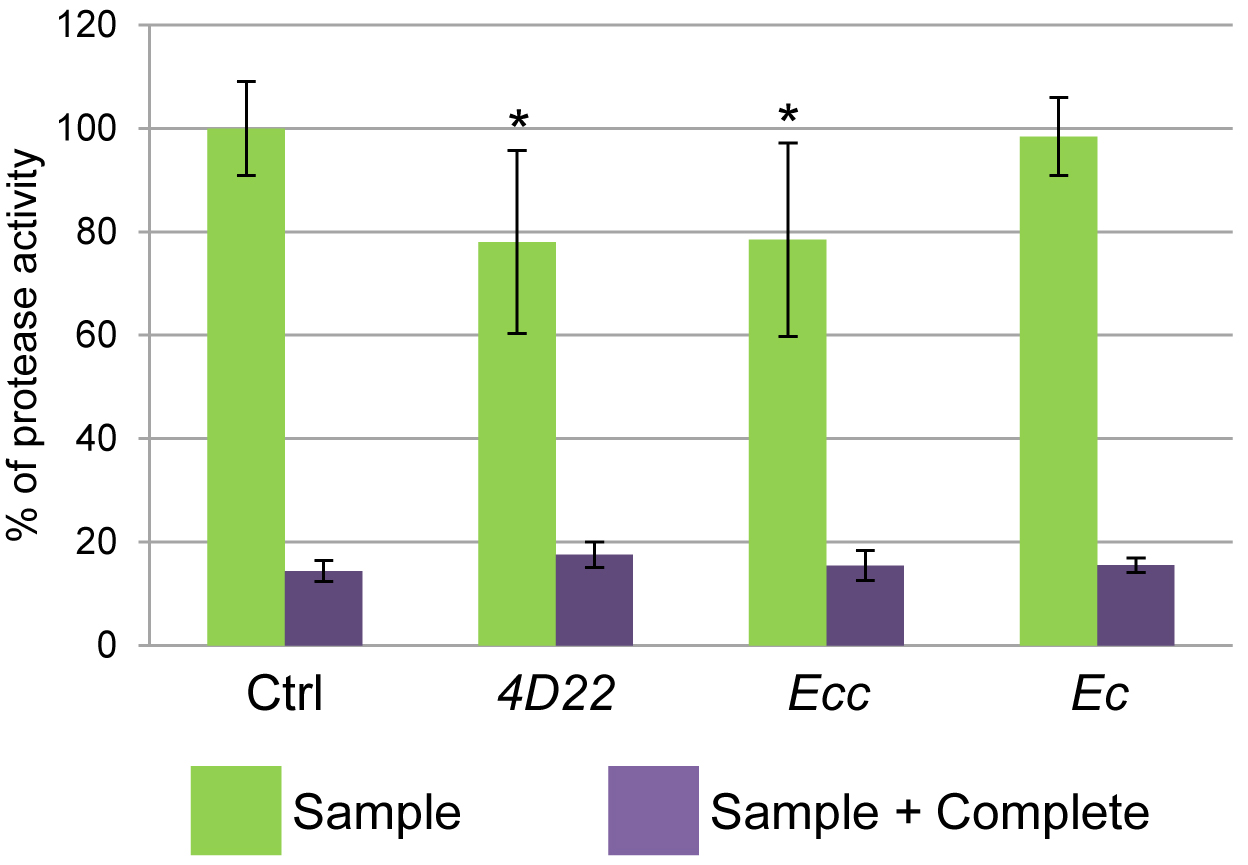
Figure 4. Percentage of protease activity. Protease activity (proportional to FITC intensity) shows a reduction of about 20% following 4D22 or Ecc treatment. No difference is observed between the Ctrl and Ec ingestion. *P ≤ 0.05. - Results are presented in % of FITC intensity (reflecting protease activity) where Ctrl is considered at 100% (Table 4).
- Statistical analysis
- Effects of treatments are analyzed using a pair wise comparison test (Tukey’s test).
- Samples are compared to the control.
- Differences are considered significant when P < 0.05 (*P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001) (Figure 4).
- The used software is Kyplot.
- Effects of treatments are analyzed using a pair wise comparison test (Tukey’s test).
Notes
The protocol described here can easily be transposed to Drosophila larval midgut and even to any other insect midguts.
Recipes
- Standard nutrient medium for Drosophila melanogaster
Note: All the reagents are mixed in distilled water.
8 g/L agar
25 g/L sugar
80 g/L cornflour
20 g/L yeast
6 g/L tegosept (stock solution at 100 g/L in 95% ethanol. Store at 4 °C) - LB medium
- Weigh out 25 g of Luria broth medium powder
- Adjust to 1 L with distilled water in a graduated test tube
- Adjust pH to 7.2 if necessary
- Autoclave
- Weigh out 25 g of Luria broth medium powder
- LB-agar medium
- Weigh out 25 g of Luria broth medium powder
- Adjust to 1 L with distilled water in a graduated test tube
- Adjust pH to 7.2 if necessary
- Add 15 g agar powder
- Autoclave
- Weigh out 25 g of Luria broth medium powder
- 10% sucrose
- Weigh out 2 g of sucrose in a 50 ml tube
- Add 20 ml distilled water and vortex
- Weigh out 2 g of sucrose in a 50 ml tube
- 1x phosphate-buffered saline (PBS)
- Add 100 ml of 10x PBS solution to 900 ml of distilled water in a graduated test tube
- Filter and store at 4 °C if you want to keep this solution
- Add 100 ml of 10x PBS solution to 900 ml of distilled water in a graduated test tube
- Trypsin solution
Note: This solution has to be prepared immediately before use, do not keep it because of trypsin self-digest.- Weigh out 10 mg of trypsin powder in a 2 ml microtube and add 1 ml 1 mM HCl
- Vortex to dissolve powder and keep the tube on ice
- Weigh out 10 mg of trypsin powder in a 2 ml microtube and add 1 ml 1 mM HCl
- Casein-FITC
- Weigh out 10 mg of Casein Fluorescein IsoThioCyanate powder in a 15 ml tube
- Add 10 ml of distilled water
- Wrap the tube with aluminum foil and vortex thoroughly until total dissolution
- Keep Casein-FITC on ice and in the dark until use
- Weigh out 10 mg of Casein Fluorescein IsoThioCyanate powder in a 15 ml tube
- 25x cOmplete
- Dissolve one tablet of cOmplete tablets EDTA free in 2 ml of distilled water in a 5 ml tube
- Vortex strongly
- Divide it into two 1.5 ml microtubes and store one at -20 °C
Note: Keep the other one tube containing cOmplete on ice until use.
- Dissolve one tablet of cOmplete tablets EDTA free in 2 ml of distilled water in a 5 ml tube
- 10% TCA
- Make a stock solution with 100 mg of trichloroacetic acid powder in 100 ml of distilled water
- Dilute at 1:10 this stock solution by adding 5 ml of TCA stock solution to 45 ml of distilled water
Note: Do not keep the diluted solution. - Make a stock solution with 100 mg of trichloroacetic acid powder in 100 ml of distilled water
- 0.5 M Tris/HCl pH 8.5
- Weigh out 60.57 g of Tris base powder for 1 L of distilled water
- Adjust pH to 8.5 with 12 N HCl
- Filter if you want to store this solution
- Weigh out 60.57 g of Tris base powder for 1 L of distilled water
Acknowledgments
We would like thank David Pauron for his help at the beginning of the protocol setup. MPNE was supported by INRA. RL was supported by the Fondation pour la Recherche Médicale (FRM) and Université Côte d'Azur (UCA) and AG was supported by the CNRS.
References
- Bonfini, A., Liu, X. and Buchon, N. (2016). From pathogens to microbiota: How Drosophila intestinal stem cells react to gut microbes. Dev Comp Immunol 64: 22-38.
- Chen, J., Li, J., Huang, H. and Xi, R. (2016). Gene expression analysis of sorted cells by RNA-seq in Drosophila intestine. Bio Protoc 6(24): e2079.
- Gonzalez, J. M., Jr., Brown, B. J. and Carlton, B. C. (1982). Transfer of Bacillus thuringiensis plasmids coding for delta-endotoxin among strains of B. thuringiensis and B. cereus. Proc Natl Acad Sci U S A 79(22): 6951-6955.
- Hu, D. J. and Jasper, H. (2017). Epithelia: Understanding the cell biology of intestinal barrier dysfunction. Curr Biol 27(5): R185-R187.
- Jasper, H. (2015). Exploring the physiology and pathology of aging in the intestine of Drosophila melanogaster. Invertebr Reprod Dev 59(sup1): 51-58.
- Loudhaief, R., Brun-Barale, A., Benguettat, O., Nawrot-Esposito, M. P., Pauron, D., Amichot, M. and Gallet, A. (2017). Apoptosis restores cellular density by eliminating a physiologically or genetically induced excess of enterocytes in the Drosophila midgut. Development 144(5): 808-819.
- Royet, J. and Charroux, B. (2013). Mechanisms and consequence of bacteria detection by the Drosophila gut epithelium. Gut Microbes 4(3): 259-263.
- Soultoukis, G. A. and Partridge, L. (2016). Dietary protein, metabolism, and aging. Annu Rev Biochem 85: 5-34.
Article Information
Copyright
© 2017 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Nawrot-Esposito, M., Loudhaief, R. and Gallet, A. (2017). Protease Activity Assay in Fly Intestines. Bio-protocol 7(18): e2560. DOI: 10.21769/BioProtoc.2560.
Category
Biochemistry > Protein > Activity
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link










