- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Axenic Culture of Sclerotinia sclerotiorum and Preparation of Sclerotinia Culture Filtrate Elicitor 1 (SCFE1)-containing Fractions, Triggering Immune Responses in Arabidopsis thaliana
Published: Vol 4, Iss 17, Sep 5, 2014 DOI: 10.21769/BioProtoc.1232 Views: 10406
Reviewed by: Ru ZhangAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols

An Assay to Study Botrytis cinerea-infected Grapevine Leaves Primed with Pseudomonas fluorescens
Charlotte Gruau [...] Aziz Aziz
Oct 5, 2016 12843 Views

Assessment of Wheat Resistance to Fusarium graminearum by Automated Image Analysis of Detached Leaves Assay
Alexandre Perochon and Fiona M. Doohan
Dec 20, 2016 12751 Views

Quantification of the Composition Dynamics of a Maize Root-associated Simplified Bacterial Community and Evaluation of Its Biological Control Effect
Ben Niu and Roberto Kolter
Jun 20, 2018 10502 Views
Abstract
The necrotrophic white mold fungus Sclerotinia sclerotiorum (S. sclerotiorum) is pathogenic to a broad range of plant species, including the Brassicaceae model plant Arabidopsis thaliana (Boland and Hall, 1994; Bolton et al., 2006). In Arabidopsis thaliana (A. thaliana), the semi-purified proteinaceous S. sclerotinorum elicitor SCFE1 (Sclerotinia culture filtrate elicitor 1) is sensed at the plasma membrane by the receptor-like protein RLP30 and triggers strong immune responses (Zhang et al., 2013), similar to the bacterial elicitor flagellin (Felix et al., 1999). Elicitation of plant defenses with SCFE1 is a tool to dissect the signaling pathway involving RLP30 and to study immunity to necrotrophic fungi. Here, we describe a simple protocol to axenically grow S. sclerotiorum. Further, we present a two-step liquid chromatography-based method for the partial purification of SCFE1 from culture filtrate (Figure 1A-B). Measurement by gas chromatography of the emission of the plant stress hormone ethylene is proposed as a bioassay to monitor elicitor activity in the fractions throughout the purification procedure (Figure 1C).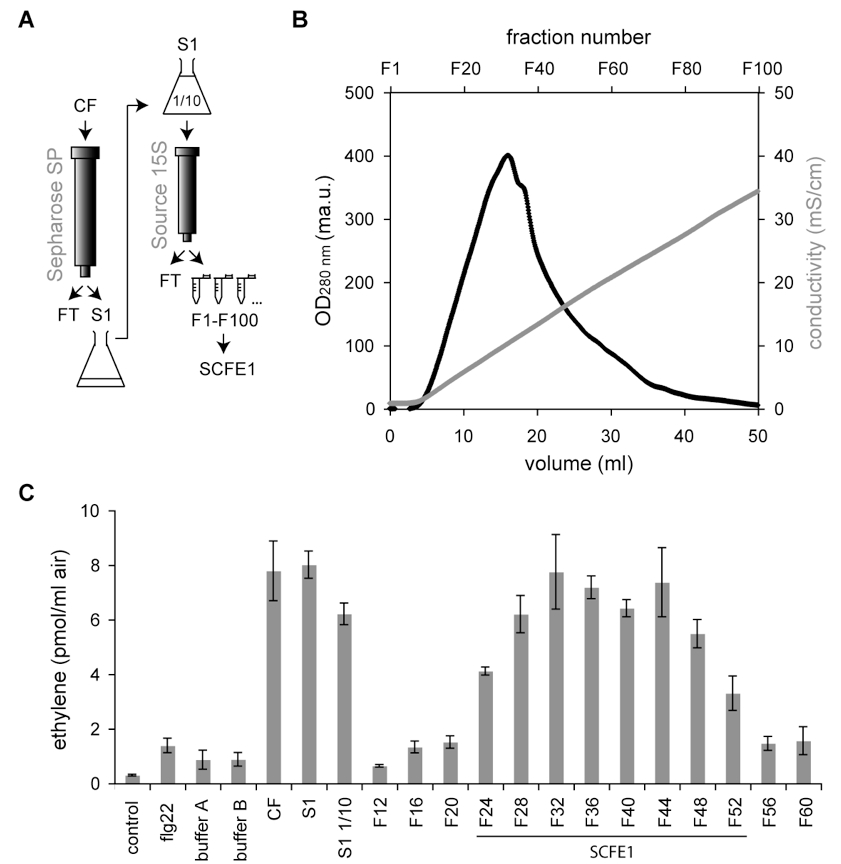
Figure 1. Two-step chromatographic fractionation of S. sclerotiorum culture filtrate to obtain semi-purified SCFE1. A. Purification scheme of SCFE1. Crude filtrate (CF) is loaded onto a Sepharose SP cation exchange chromatography column. The eluate (S1) is diluted 10-fold and loaded onto a Source 15S cation exchange chromatography column. Elution is performed with a linear gradient of 0 to 0.3 M KCl and elution fractions of 0.5 ml (F1 - F100) are collected. FT = flow-through. B. Chromatogram of the SCFE1-containing fractions eluted from a Source 15S cation exchange chromatography column. The black line represents the protein elution profile monitored with OD280 nm. The grey line shows the increasing conductivity of the elution buffer. ma.u. = milli-arbitrary units. mS = milli-Siemens. C. Ethylene response in Arabidopsis Col-0 to SCFE1-containing fractions obtained by two-step cation exchange chromatography. Arabidopsis Col-0 leaf pieces are treated with 15 µl CF, undiluted and 10-fold diluted S1 as well as the Source 15S elution fractions (Fx). Treatment with 0.5 µM flg22 is used as a positive control for ethylene production. No treatment and treatment with 15 µl buffers A and B are used as negative controls. In this representative purification, SCFE1 is contained in fractions F24 to F52. Bars represent average values (n=2) ± S.D.
Materials and Reagents
- Arabidopsis thaliana Col-0 purchased from ‘Nottingham Arabidopsis Stock Centre’
- Sclerotinia sclerotiorum strain 1946 [purchased from the biological resource center ‘Deutsche Sammlung von Mikroorganismen und Zellkulturen’ (DSMZ) GmbH, shipped in a sterile culture tube on agar medium, stored at 15 °C in the dark]
- Malt extract (Carl Roth, catalog number: X976.2 )
- Peptone from casein (tryptic digest) (Carl Roth, catalog number: 8986.2 )
- Agar-agar (bacteriological) (Carl Roth, catalog number: 2266.3 )
- Ultrapure H2O (type 1) (filtered with MilliQ Reference purification system) (Merck KGaA)
- MES [2-(N-morpholino) ethanesulfonic acid] (Carl Roth, catalog number: 4256.3 )
- KCl (Carl Roth, catalog number: P017.1 ) (2 M solution with ultrapure H2O)
- Ethanol (Merck KGaA, catalog number: 100983 ) (diluted to 20% with ultrapure H2O)
- XK16/20 chromatography column (GE Healthcare, catalog number: 28-9889-37 )
- Sepharose SP Fast Flow (GE Healthcare, catalog number: 17-0729-01 ) (stored at 4 °C)
- Source 15S 4.6/100 PE column (GE Healthcare, catalog number: 17-5182-01 ) (stored at 4 °C)
- GS90 soil (CL Ton Kokos) (Gebrüder Patzer, catalog number: 10-00800-40 )
- Vermiculite for plant culture (BayWa AG or Gebrüder Patzer)
- Rubber stoppers (Carl Roth, catalog number EC93.1 )
- Flg22 peptide from Pseudomonas aeruginosa (Felix et al., 1999) (synthesized, 50 µM, stored at -20 °C)
- Malt-peptone-agar (see Recipes)
- Malt-peptone medium (see Recipes)
- Buffer A (see Recipes)
- Buffer B (see Recipes)
Equipment
- Petri dishes (94 x 16 mm, with vents) (Greiner Bio-One GmbH, catalog number: 633180 )
- Paraffin film (Parafilm M) (Sigma-Aldrich, catalog number: P7793 )
- Needle holder (Carl Roth, catalog number: 6189.1 )
- Lancets (Carl Roth, catalog number: 6181.1 )
- 1 L culture flasks (erlenmeyer-shaped, with straight neck and metal caps) (SCHOTT AG, DURAN®, catalog number: 21 771 54 )
- Laboratory funnel (minimum 15 cm diameter) (Carl Roth, catalog number: YA51.1 )
- Nylon mesh (60 µm pore size) (Carl Roth, catalog number: XA95.1 )
- 1 L laboratory bottles with screw-cap (SCHOTT AG, DURAN®, catalog number: 21 801 54 5 )
- Small silica gel bag
- 50 ml Falcon conical centrifuge tubes (VWR International, catalog number: 21008-940 )
- 0.2 µm bottle-top filters for 500 ml (Carl Roth, catalog number: AC20.1 )
- Paper scissors
- 6 ml thick-walled glass test tubes (Carl Roth, catalog number: HA75.1 )
- 1 ml syringe (B. Braun Melsungen AG, Omnifix-F Tuberculin, catalog number: 9161406V )
- Syringe needle (27 Ga. x ¾ in., size 20) (B. Braun Melsungen AG, Sterican, catalog number: 4657705 )
- Spatula (Carl Roth, catalog number: YK51.1 )
- 1.5 ml reaction tubes with caps (VWR International, catalog number: 700-5239 )
- MilliQ Reference purification system (Merck KGaA, catalog number: Z00QSV001 )
- Laminar flow clean bench
- 15 °C incubator
- Front-loading lyophilizer of at least 10 L capacity with basins (e.g. L10, WKF-Gesellschaft für elektrophysikalischen Apparatebau, not produced anymore)
- Floor-standing high-speed centrifuge (e.g. Sorvall RC-5B Plus Superspeed centrifuge and Sorvall SLA-1500 Superlite rotor with adapters for 50 ml Falcon conical centrifuge tubes, Thermo Fisher Scientific Inc., not produced anymore)
- Vacuum pump (Gardner Denver, Welch, catalog number: 2522C-02 )
- 4 °C cold lab chamber or cold room
- ÄKTA Explorer 10 liquid chromatography system (GE Healthcare, catalog number: 18-1300-00 ) equipped with:
- Pump P-900
- Sample pump P-960 (GE Healthcare, catalog number: 18-6727-00 )
- Monitor UV-900 (GE Healthcare, catalog number: 18-1108-35 )
- Monitor pH/C-900 (GE Healthcare, catalog number: 18-1129-74 )
- Fraction collector Frac-950 (GE Healthcare, catalog number: 18-6083-00 )
- Rack A for 18/30 mm tubes (GE Healthcare, catalog number: 18-6083-11 )
- Rack B for 12 mm tubes (GE Healthcare, catalog number: 18-6083-12 )
- Unicorn control software (version 4.12 or more)
- Pump P-900
- Plant growth chamber (controlled light, temperature and humidity)
- Gas chromatograph with Al2O3-column and flame-ionization detector (e.g. GC-14A with analysis unit C-R4AX Chromatopac, Shimadzu Deutschland GmbH, not produced anymore)
Procedure
- Axenic culture of Sclerotinia sclerotiorum
- Work under sterile conditions when handling S. sclerotiorum cultures.
- Prepare fungal stock plate:
- Cut 2 agar plugs (0.5 x 0.5 cm) from the initial S. sclerotiorum-containing culture tube using a lancet.
- Place agar plugs in a Petri dish on malt-peptone-agar.
- Seal the plate with paraffin film.
- Let the fungus grow for 3 days at room temperature (RT) in the dark (Figure 2).
Note: The mycelium should cover almost the whole surface of the plate. - Store plate at 15 °C in the dark and reuse it for up to 3 months.
- Cut 2 agar plugs (0.5 x 0.5 cm) from the initial S. sclerotiorum-containing culture tube using a lancet.
- Prepare fungal liquid culture:
- Prepare 1 L culture flasks containing 400 ml malt-peptone medium (25 flasks for 10 L culture).
- Cut agar plugs (0.5 x 0.5 cm) of fresh mycelium from the stock plate and inoculate each flask with 2 of them.
- Let fungus grow without shaking for 2-3 weeks at RT in the dark until the liquid surface is completely covered with mycelium.
- Prepare 1 L culture flasks containing 400 ml malt-peptone medium (25 flasks for 10 L culture).
Figure 2. S. sclerotiorum growing on a malt-peptone-agar plate - Work under sterile conditions when handling S. sclerotiorum cultures.
- Freeze-drying of S. sclerotiorum culture filtrate
- Put a nylon mesh into a laboratory funnel. Filter the culture medium through the mesh and collect it.
- Discard the mycelium.
- Poor the culture filtrate into basins and freeze-dry it in a front-loading lyophilizer for 3 to 4 days.
- Store freeze-dried material at RT in a hermetically closed 1 L laboratory bottle. Add a silica gel bag to keep the material dry.
- Put a nylon mesh into a laboratory funnel. Filter the culture medium through the mesh and collect it.
- Re-suspension of the culture filtrate
- Re-suspend freeze-dried culture filtrate (e.g. 15 g dry weight) in a minimal volume (approx. 6 ml/g) of ice-cold buffer A in a 50 ml Falcon conical centrifuge tube.
- Centrifuge sample for 20 min at 10,000 x g and 4 °C.
- Collect the supernatant and keep on ice.
- Repeat steps C9-10 if necessary to remove all insoluble particles.
- Keep an aliquot of culture filtrate for subsequent activity assays in plants.
- Re-suspended culture filtrate may be stored at -20 °C.
Note: Frozen concentrated culture filtrate is stable for at least one year. Repeated freeze-thaw cycles do not damage the sample.
- Re-suspend freeze-dried culture filtrate (e.g. 15 g dry weight) in a minimal volume (approx. 6 ml/g) of ice-cold buffer A in a 50 ml Falcon conical centrifuge tube.
- Isolation of SCFE1: Rough fractionation by cation exchange chromatography
- For the isolation of SCFE1 from re-suspended culture filtrate, work with the ÄKTA Explorer liquid chromatography system cooled at 4 °C (in a cold lab chamber or cold room).
- All solutions for the chromatography should be prepared with ultrapure H2O, filtered through a bottle-top filter into clean, dust-free bottles and cooled at 4 °C.
- Pack a XK16/20 chromatography column from GE Healthcare with Sepharose SP Fast Flow matrix from GE Healthcare for cation exchange chromatography to around 15 ml column volume (CV).
Note: For a small-scale purification (less than 5 g dry weight), 1 or 5 ml pre-packed Sepharose SP Fast Flow columns from GE Healthcare may be used. - Wash column with 5 CV ultrapure H2O at a flow rate of 3 ml/min.
- Equilibrate column with 5 CV buffer A at a flow rate of 3 ml/min.
- Load re-suspended culture filtrate via the P-960 sample pump at a flow rate of 3 ml/min.
- Register flow, pressure, pH, conductivity, OD280 nm and OD215 nm during the run.
- Collect flow-through once OD280 nm increases and until loading is completed.
- Wash column with buffer A until OD280 nm and OD215 nm are back to the base line and stable.
- Elute in one step with 100% buffer B.
- Collect eluate into a 50 ml Falcon conical centrifuge tube using rack A for the fraction collector. Start collecting once OD280 nm exceeds 300 ma.u. (milli-arbitrary units) and stop when the end of the elution peak is reached.
- Wash column subsequently with 5 CV 2 M KCl, 5 CV ultrapure H2O, then 2 CV 20% ethanol for storage.
- Before proceeding to the second purification step, test activity of culture filtrate (CF), flow-through (FT) and eluate (S1) with the ethylene assay in Arabidopsis (see step F).
Note: S1 should have the highest activity and contain most of SCFE1. - Store S1 at 4 °C until next day or at -20 °C for more than 1 year. The flow-through may be discarded if the activity is successfully recovered in S1.
- For the isolation of SCFE1 from re-suspended culture filtrate, work with the ÄKTA Explorer liquid chromatography system cooled at 4 °C (in a cold lab chamber or cold room).
- Isolation of SCFE1: Refined fractionation by cation exchange chromatography
- Dilute S1 10-fold with buffer A.
- Install a pre-packed Source 15S 4.6/100 PE column from GE Healthcare for cation exchange chromatography.
- Wash column with 5 CV ultrapure H2O at a flow rate of 1ml/min.
- Equilibrate column with 5 CV buffer A at a flow rate of 1 ml/min.
- Load total volume of diluted sample via the P-960 sample pump at a flow rate of 1 ml/min.
Note: The pressure on the column increases with time. The flow rate may be reduced progressively. - Register flow, pressure, pH, conductivity, OD280 nm and OD215 nm during the run.
- Collect flow-through once OD280 nm increases and until loading is completed.
- Wash column with buffer A until OD280 nm and OD215 nm are back to the base line and stable.
- Elute with a linear gradient from 0% to 60% buffer B in 40 CV at a flow rate of 0.5 to 1 ml/min (see Figure 1B for a typical elution profile).
- Collect 0.5 ml fractions in 1.5 ml reaction tubes using rack B for the fraction collector (approx. 100 fractions, labeled F1-F100). Create a program for automated fractionation with the method wizard of the Unicorn software.
Note: SCFE1 should elute at a salt conductivity value between 8 and 16 mS/cm. - Wash column with 5 CV 100% buffer B at a flow rate of 1 ml/min.
Note: A small elution peak may appear. It does not need to be collected, as it does not contain any activity. - Wash column with 5 CV water, then 2 CV 20% ethanol for storage.
Note: If the pressure on the column remains higher than initially, it means that it was slightly clogged during sample loading and requires additional washing before reuse. In this case, follow the cleaning-in-place (CIP) procedure for Source 15S columns as described by the manufacturer. - Test activity of flow-through (FT) and elution fractions (F1-F100) with the ethylene assay in Arabidopsis (see step F).
Note: It is sufficient to test 1 fraction out of 5 for those with low OD280 nm and 1 fraction out of 2 for those corresponding to the elution peak. - Store active fractions containing semi-purified SCFE1 at 4 °C for maximum 1 day or at -20 °C for more than 1 year. The flow-through may be discarded if the activity is successfully recovered in the elution fractions.
- Dilute S1 10-fold with buffer A.
- Ethylene assay
- Grow Arabidopsis Col-0 plants on soil (GS90/vermiculite mixture 3.5: 1) for 5 to 6 weeks in a growth chamber: 8 h light/16 h dark, 23 °C, 40–60% relative humidity, 100 µmol m-2 s-1 light intensity (short day conditions).
- Choose the biggest leaves from the plants. Cut each leaf into 6 rectangular pieces using paper scissors. Omit the edges, tip and stalk. Float leaf pieces on dH2O in a Petri dish (Figure 3).
Note: Prepare the leaf pieces 12 to 20 h before elicitation. Leaf pieces left for a longer time will not respond well to elicitation anymore. - Leave leaf pieces over night in the closed Petri dish at RT or in an air-conditioned room.
- Pipet 0.5 ml dH2O into glass test tubes. Use 2 or 3 tubes per sample to test (for duplicate or triplicate measurements).
- Put 3 leaf pieces into each test tube using a spatula.
Note: Leaf pieces may rest for a few hours in the test tubes or may be elicited immediately. - Add 5 to 15 µl of the samples to test (CF, FT, S1 and/or selected fractions of F1-F100). Include an untreated control and a positive control (5 µl of 50 µM flg22 = 0.5 µM final concentration).
- Close test tubes with rubber stopper and carefully shake tube rack to mix.
Note: Leaves should stay in the water or on the surface but not stick to the glass. - Incubate for 3 to 4 h at RT.
- Measure the ethylene content in each tube:
- Prick through the rubber stopper of a tube using a 1 ml syringe with a needle.
- Mix the air inside the tube by moving up and down several times.
- Withdraw 1 ml air and inject it into the gas chromatograph.
- Record retention time and peak area to determine the amount of ethylene in pmol/ml air (see Figure 1C for representative data sets).
Note: For the untreated control, an average value of 0.60 +/- 0.21 pmol/ml air is expected. The mean values for buffer A and B are 0.81 +/- 0.23 and 0.81 +/- 0.23, respectively. With 1.30 +/- 0.20 pmol/ml, the ethylene production in the flg22 control is expected to be around 2x higher than in the non-treated and 1.5x higher than in the buffer controls. The values for culture filtrate (CF) can range from 2.5 to 15 pmol/ml. As culture filtrate yields a higher ethylene response than flg22, an active culture filtrate sample from a previous purification (stored at -20 °C) may be used as additional positive control.
- Prick through the rubber stopper of a tube using a 1 ml syringe with a needle.

Figure 3. Cutting and floating of A. thaliana leaf pieces - Grow Arabidopsis Col-0 plants on soil (GS90/vermiculite mixture 3.5: 1) for 5 to 6 weeks in a growth chamber: 8 h light/16 h dark, 23 °C, 40–60% relative humidity, 100 µmol m-2 s-1 light intensity (short day conditions).
Notes
- The ethylene assay is a very reliable and fast method to detect and quantify the immune-eliciting activity of the SCFE1-containing fractions. It also allows testing a large number of samples in a row. Other immune assays may however be used, such as the luminol-based oxidative burst assay to measure the production of reactive oxygen species (requires dialysis of the SCFE1-containing fractions to remove buffer), immunoblotting to detect activated MAP kinases or qRT-PCR for transcriptional profiling of defense-related genes (Zhang et al., 2013).
- To confirm that in the selected chromatographic fractions truly contain SCFE1 and no other or additional elicitor, the fractions may be tested on the Arabidopsis RLP30 knock-out mutants rlp30-1, -2 or -4. These mutants are completely insensitive to treatment with SCFE1-containing fractions, but react normally to flg22 challenge. Alternatively proteinase K sensitivity and heat stability of the immune-eliciting activity are good indicators for the identity of SCFE1 (Zhang et al., 2013).
Recipes
- Malt-peptone-agar (autoclaved)
10 g malt extract
2.5 g peptone from casein, tryptic digest
15 g agar-agar
dH2O qs. to 1 L - Malt-peptone medium (autoclaved)
10 g malt extract
2.5 g peptone from casein, tryptic digest
dH2O qs. to 1 L - Buffer A
0.1 M MES
Ultrapure H2O
Adjust pH to 5.4
Sored at 4 °C - Buffer B
0.1 M MES
0.5 M KCl
Ultrapure H2O
Adjust pH to 5.4
Stored at 4 °C
Acknowledgments
We acknowledge Prof. Georg Felix and his group at the ZMBP for having established gas chromatography and the ethylene assay protocol in the research department.
References
- Boland, G. and Hall, R. (1994). Index of plant hosts of Sclerotinia sclerotiorum. Canadian J Plant Pathol 16(2): 93-108.
- Bolton, M. D., Thomma, B. P. and Nelson, B. D. (2006). Sclerotinia sclerotiorum (Lib.) de Bary: biology and molecular traits of a cosmopolitan pathogen. Mol Plant Pathol 7(1): 1-16.
- Felix, G., Duran, J. D., Volko, S. and Boller, T. (1999). Plants have a sensitive perception system for the most conserved domain of bacterial flagellin. Plant J 18(3): 265-276.
- Zhang, W., Fraiture, M., Kolb, D., Löffelhardt, B., Desaki, Y., Boutrot, F. F., Tor, M., Zipfel, C., Gust, A. A. and Brunner, F. (2013). Arabidopsis receptor-like protein30 and receptor-like kinase suppressor of BIR1-1/EVERSHED mediate innate immunity to necrotrophic fungi. Plant Cell 25(10): 4227-4241.
Article Information
Copyright
© 2014 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Fraiture, M. and Brunner, F. (2014). Axenic Culture of Sclerotinia sclerotiorum and Preparation of Sclerotinia Culture Filtrate Elicitor 1 (SCFE1)-containing Fractions, Triggering Immune Responses in Arabidopsis thaliana. Bio-protocol 4(17): e1232. DOI: 10.21769/BioProtoc.1232.
Category
Plant Science > Plant immunity > Perception and signaling
Microbiology > Microbe-host interactions > In vivo model > Plant
Biochemistry > Protein > Isolation and purification
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link









