- Submit a Protocol
- Receive Our Alerts
- Log in
- /
- Sign up
- My Bio Page
- Edit My Profile
- Change Password
- Log Out
- EN
- EN - English
- CN - 中文
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
- EN - English
- CN - 中文
- Home
- Protocols
- Articles and Issues
- For Authors
- About
- Become a Reviewer
Citrus Fruit Ascorbic Acid Extraction and Quantification by HPLC
Published: Vol 5, Iss 5, Mar 5, 2015 DOI: 10.21769/BioProtoc.1416 Views: 19629
Reviewed by: Samik BhattacharyaLaia ArmengotAnonymous reviewer(s)

Protocol Collections
Comprehensive collections of detailed, peer-reviewed protocols focusing on specific topics
Related protocols
Isolation of Intact Vacuoles from Arabidopsis Root Protoplasts and Elemental Analysis
Chuanfeng Ju [...] Zhenqian Zhang
Mar 5, 2023 2112 Views
High-Performance Liquid Chromatography Quantification of Glyphosate, Aminomethylphosphonic Acid, and Ascorbate in Culture Medium and Microalgal Cells
Juan Manuel Ostera [...] Gabriela Malanga
Apr 5, 2025 1227 Views
CAPS-Based SNP Genotyping for Nitrogen-Response Phenotypes in Maize Hybrids
Jannis Jacobs [...] Peter K. Lundquist
Dec 20, 2025 594 Views
Abstract
Citrus are among the most relevant sources of vitamin C (ascorbic acid + dehydroascorbic acid). Recent studies have revealed that it increases in the peel as fruit ripens and remains constant or even decreases in the pulp tissue. Moreover, important differences on ascorbic acid content exist among citrus varieties in both tissues. Here we describe a simple method for vitamin C analysis/quantification in the peel and pulp tissues of citrus fruit.
Keywords: CitrusMaterials and Reagents
- Citrus fruit tissue (ground frozen flavedo or pulp tissue)
- Metaphosphoric acid (MPA) (65%) (Sigma-Adrich, catalog number: 79615 )
- Orthophosphoric acid (Sigma-Adrich, catalog number: W290017 )
- Tris base (Sigma-Adrich, catalog number: T1503 )
- Methanol HPLC-grade (Sigma-Adrich, catalog number: 34860 )
- Ascorbic acid (Sigma-Adrich, catalog number: A902902 )
- C18 Sep Pak (Waters, catalog number: WAT091139 )
- DTT (Sigma-Aldrich, catalog number: D0632 )
- Sterile Mili-Q water
- 0.1% MPA (see Recipes)
- 2% MPA (see Recipes)
- 8.5% orthophosphoric acid (see Recipes)
- 400 mM Tris base (see Recipes)
- 200 mM DTT in 400 mM Tris base (see Recipes)
- Methanol:Milli-Q water (15:85, v/v) (see Recipes)
- Ascorbic acid stock solution I for standard curve (see Recipes)
Equipment
- Disposable 15 ml plastic tubes
- 0.45 μm nylon filter (25 mm diameter) (Análisis Vínicos, catalog number: E0036 )
- Refrigerated centrifuge (15 ml tubes)
- Polytron (Kinematica AG, model: PT-1035 GT; http://www.kinematica.ch/en.html)
- HPLC system with a photodiode array detector (PDA, Dionex)
- Ultrabase C18 column (100 x 4.6 mm, 2.5 μm)
Software
- Chromeleon version 6.80 (Dionex)
Procedure
- Ascorbic acid extraction
- Resuspend frozen ground citrus flavedo or pulp tissue (0.5 g) in 4 ml 0.1% MPA in a 15 ml disposable plastic tube and keep it on ice. The frozen tissue was previously ground using mortar and pestle chilled with liquid nitrogen.
- Homogenize the tissue for 1 min using a Polytron homogenizer at maximum speed (30,000 rpm).
- Centrifuge the homogenate for 10 min at 4,000 x g at 4 °C.
- Collect the supernatant and keep it in a fresh 15 ml disposable plastic tube on ice.
- Activate the Sep Pak C18 cartridges using 4 ml methanol and subsequently wash them with 4 ml Milli-Q water and 4 ml 2% MPA. Discard the solution from the washing steps.
- For the ascorbic acid determination in flavedo take 1 ml of supernatant and mix it with 3 ml 0.1% MPA (1:4 dilution). For ascorbic acid determination in pulp do not dilute it, just continue with the protocol using the whole supernatant.
- Filter the solutions obtained in step A4 (pulp) or A6 (flavedo) through a previously activated C18 Sep Pak cartridge using a syringe. Collect the filtrate in a fresh 15 ml disposable plastic tube.
- Wash the C18 Sep Pak cartridge with 4 ml 2% MPA. Collect the eluate together with the solution from step A7.
- Filter the solution from step A8 through a 0.45 μm nylon filter using a syringe. This step is added to make sure the solution is clean of any particles that could damage the HPLC column.
- Wash the filter with 4 ml 2% MPA and collect it together with the filtrate solution from step A9.
- Measure the final volume and take it into account together with the initial dilution (in the case of flavedo samples) for the calculations of ascorbic acid concentration.
- Prepare an amber HPLC vial with 1 ml of the final extract (from step A10) for the ascorbic acid measurement.
- Resuspend frozen ground citrus flavedo or pulp tissue (0.5 g) in 4 ml 0.1% MPA in a 15 ml disposable plastic tube and keep it on ice. The frozen tissue was previously ground using mortar and pestle chilled with liquid nitrogen.
- Dehydroascorbic acid extraction
Dehydroascorbic acid is converted to ascorbic acid by the addition of DTT, then the amount of total ascorbic acid (dehydroascorbic acid + ascorbic acid) is measured and the dehydroascorbic acid concentration results from the substraction of ascorbic acid to total ascorbic acid (see step D3).- Transfer a 200 μl aliquot of the final extract (step A10) to an amber HPLC vial.
- Incubate it for 15 min at room temperature with 100 μl of 200 mM DTT in 400 mM Tris base (pH 8.5).
- Stop the reaction by acidification with 100 μl of 8.5 % orthophosphoric acid.
- Transfer a 200 μl aliquot of the final extract (step A10) to an amber HPLC vial.
- Ascorbic acid and dehydroascorbic acid HPLC measurements
- To quantify the ascorbic acid concentration inject 10 μl of the solution obtained from step A10 in a HPLC equipped with a photodiode array detector and an Ultrabase C18 column (100 x 4.6 mm, 2.5 μm). Set column temperature at 35 °C. Use an isocratic mobile phase of methanol: Milli-Q water (pH 2.5) (15:85, v/v, pH adjusted with MPA) at 0.2 ml/min flow.
- Quantify the area under the peak that displays a maximum at 248 nm (ascorbic acid absorbs between 210 and 300 nm with a maximum at 248 nm). This area is applied to the formula described in D2 to calculate the ascorbic acid concentration of the sample. Measure the spectrum between 200 and 450 nm with a PDA detector to confirm the quantification of pure ascorbic acid.
- To obtain the dehydroascorbic acid concentration inject 10 μl of the solution obtained from step B3 using the same equipment and conditions described in step C1.
- Quantify the area under the peak that displays a maximum at 248 nm. Apply this area to the formula described in D3 to calculate the dehydroascorbic acid concentration of the sample. Measure the spectrum between 200 and 450 nm with a PDA detector to confirm the quantification of pure ascorbic acid.
- To quantify the ascorbic acid concentration inject 10 μl of the solution obtained from step A10 in a HPLC equipped with a photodiode array detector and an Ultrabase C18 column (100 x 4.6 mm, 2.5 μm). Set column temperature at 35 °C. Use an isocratic mobile phase of methanol: Milli-Q water (pH 2.5) (15:85, v/v, pH adjusted with MPA) at 0.2 ml/min flow.
- Ascorbic acid and dehydroascorbic acid quantification
- Prepare a calibration curve using a series of ascorbic acid dilutions as described in Table 1 and measure the peak area following the same procedure as described in section C.
- Adjust the linear function of ascorbic acid concentration vs. peak area and use the slope to calculate the concentration of the unknown samples as described in the formula:
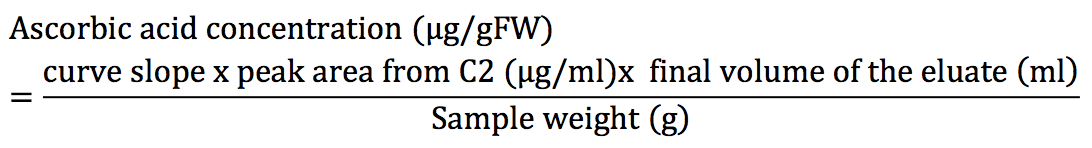
- Dehydorascorbic acid concentration:
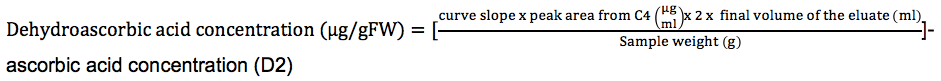
The x2 is included in the formula because it is the dilution factor due to the addition of 200 μls of extra volume in steps B2-3.
- Prepare a calibration curve using a series of ascorbic acid dilutions as described in Table 1 and measure the peak area following the same procedure as described in section C.
Recipes
- 0.1% MPA
1.5 g of MPA 65%
900 ml of Milli-Q water and after completely dissolved complete to 1,000 ml with Milli-Q water
Solution can be stored at room temperature indefinitely - 2% MPA
31 g of MPA 65%
900 ml of Milli-Q water and after completely dissolved complete to 1,000 ml with Milli-Q water
Solution can be stored at room temperature indefinitely - 8.5% orthophosphoric acid
10 ml of 85% orthophosphoric acid
90 ml of Milli-Q water and store at room temperature indefinitely - 400 mM Tris base (pH 9.0)
Dissolve 12.1 g of Tris base in 200 ml of Milli-Q water
Add Milli-Q water to a final volume of 250 ml, stored at 4 °C for three months - 200 mM DTT in 400 mM Tris base (pH 8.5)
Dissolve 0.031 g of DTT in 1 ml of 400 mM Tris base solution and vortex
Do not store, this solution must be prepared fresh daily - Methanol:Milli-Q water (pH 2.5) (15:85, v/v)
Mix 150 ml of methanol HPLC grade with 850 ml of Milli-Q water at pH 2.5 (pH is adjusted with 2% MPA) - Ascorbic acid stock solution I for standard curve (100 mg/ml in 2% MPA)
Table 1. Calibration curve preparationAscorbic acid solution Final concentration Composition Stock solution I 100 mg/ml 100 mg ascorbic acid + 1 ml 2% MPA Stock solution II 1 mg/ml 990 μl MPA 2% + 10 μl stock solution I A 100 μg/ml 900 μl MPA 2% + 100 μl stock solution II B 50 μg/ml 500 μl MPA 2% + 500 μl solution A C 25 μg/ml 500 μl MPA 2% + 500 μl solution B D 12.5 μg/ml 500 μl MPA 2% + 500 μl solution C E 6.25 μg/ml 500 μl MPA 2% + 500 μl solution D F 3.125 μg/ml 500 μl MPA 2% + 500 μl solution E G 1.5625 μg/ml 500 μl MPA 2% + 500 μl solution F H 0 μg/ml 500 μl MPA 2%
Acknowledgments
This work was supported by research grants FP7-PEOPLE-2011-CIG-2011-303652 (Marie Curie Actions, European Union), Proyectos de I + D para Grupos de Investigación Emergentes GV/2012/036 (Generalitat Valenciana, Spain). Enriqueta Alós was recipient of a JAE-Doc (CSIC) post-doctoral contract which is co-funded by Fondo Social Europeo de Desarrollo Regional (FEDER). This protocol is based on the methodology used in the manuscript Alós et at. (2014).
References
- Alós, E., Rodrigo, M. J. and Zacarías, L. (2014). Differential transcriptional regulation of L-ascorbic acid content in peel and pulp of citrus fruits during development and maturation. Planta 239(5): 1113-1128.
Article Information
Copyright
© 2015 The Authors; exclusive licensee Bio-protocol LLC.
How to cite
Alós, E., Lado, J., Rodrigo, M. J. and Zacarías, L. (2015). Citrus Fruit Ascorbic Acid Extraction and Quantification by HPLC . Bio-protocol 5(5): e1416. DOI: 10.21769/BioProtoc.1416.
Category
Plant Science > Plant biochemistry > Other compound
Plant Science > Plant physiology > Nutrition
Biochemistry > Other compound > Acid
Do you have any questions about this protocol?
Post your question to gather feedback from the community. We will also invite the authors of this article to respond.
Share
Bluesky
X
Copy link














